|
Iridium(IV) Oxide
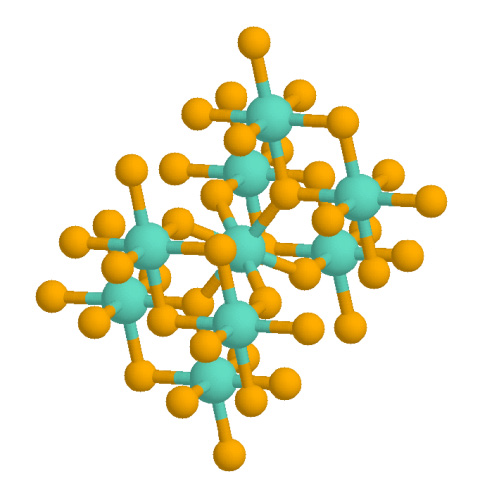
Iridium(IV) oxide, IrO2, is the only well-characterised oxide of iridium. It is a blue-black solid. The compound adopts the TiO2 rutile structure, featuring six coordinate iridium and three coordinate oxygen. It is used with other rare oxides in the coating of anode-electrodes for industrial electrolysis and in microelectrodes for electrophysiology research. As described by its discoverers, it can be formed by treating the green form of iridium trichloride Iridium(III) chloride is the inorganic compound with the formula IrCl3. The anhydrous compound is relatively rare, but the related hydrate is useful for preparing other iridium compounds. The anhydrous salt is a dark green crystalline solid. ... with oxygen at high temperatures: :2 IrCl3 + 2 O2 → 2 IrO2 + 3 Cl2 A hydrated form is also known. Application Iridium dioxide can be used as an anode electrode for industrial electrolysis and as a microelectrode for electrophysiological studies. Iridium dioxide can be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer Polymorphism (materials science), polymorphs of TiO2 are known, including anatase, akaogiite, and brookite. Rutile has one of the highest refractive index, refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion (optics), dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially Polarization (waves), polarization optics, for longer light, visible and infrared, infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum. Rutile derives its name from the Latin ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner. Occurrence Rutile is a common accessory ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iridium(IV) Fluoride
Iridium(IV) fluoride is a chemical compound of iridium and fluorine, with the chemical formula IrF4 and is a dark brown solid. Early reports of IrF4 prior to 1965 are questionable and appear to describe the compound iridium(V) fluoride, IrF5. The solid can be prepared by reduction of IrF5 with iridium black or reduction with H2 in aqueous HF. The crystal structure of the solid is notable as it was the first example of a three-dimensional lattice structure found for a metal tetrafluoride and subsequently RhF4, PdF4 and PtF4 have been found to have the same structure. Wells A.F. (1984) ''Structural Inorganic Chemistry'' 5th edition Oxford Science Publications The structure has 6 coordinate, octahedral, iridium where two edges of the octahedra are shared and the two unshared fluorine atoms are ''cis'' to one another. References {{Fluorides Iridium compounds Fluorides Platinum group halides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iridium Disulfide
Iridium disulfide is the binary inorganic compound with the formula IrS2. Prepared by the direct reaction of the elements, the compound adopts the Pyrite#Crystallography, pyrite crystal structure at high pressure. At normal atmospheric pressures, an Orthorhombic crystal system, orthorhombic polymorph is observed.. The high- and low-pressure forms both feature octahedral Ir centers, but the S–S distances are pressure dependent. Although not practical, IrS2 is a highly active catalyst for hydrodesulfurization. References } {{Sulfides Disulfides Iridium compounds Transition metal dichalcogenides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhodium Dioxide
Rhodium(IV) oxide (or rhodium dioxide) is the chemical compound with the formula Rh O2. Chemical properties RhO2 is highly insoluble even in hot aqua regia. Structure RhO2 has the tetragonal rutile structure. Physical properties RhO2 has metallic resistivity with values <10−4 Ohm·cm. It transforms in air to Rh2O3 at 850 °C and then to metal and oxygen at 1050 °C. See also *Rhodium
Rhodium is a chemical element with the symbol Rh and atomic number 45. It is a very rare, silvery-white, hard, corrosion-resistant transition metal. It is a noble metal and a member of the platinum group. It has only one naturally occurring isoto ...
Ref ...
|
Osmium Dioxide
Osmium dioxide is an inorganic compound with the formula OsO2. It exists as brown to black crystalline powder, but single crystals are golden and exhibit metallic conductivity. The compound crystallizes in the rutile structural motif, i.e. the connectivity is very similar to that in the mineral rutile. Preparation OsO2 can be obtained by the reaction of osmium with a variety of oxidizing agents, including, sodium chlorate, osmium tetroxide, and nitric oxide at about 600 °C. Using chemical transport, one can obtain large crystals of OsO2, sized up to 7x5x3 mm3. Single crystals show metallic resistivity of ~15 μΩ cm. A typical transport agent is via the reversible formation of volatile OsO4: : OsO2 + O2 OsO4 Reactions OsO2 does not dissolve in water, but is attacked by dilute hydrochloric acid. The crystals have rutile structure. Unlike osmium tetroxide Osmium tetroxide (also osmium(VIII) oxide) is the chemical compound with the formula OsO4. The compound is notewort ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum Dioxide
Adams' catalyst, also known as platinum dioxide, is usually represented as platinum(IV) oxide hydrate, PtO2•H2O. It is a catalyst for hydrogenation and hydrogenolysis in organic synthesis. This dark brown powder is commercially available. The oxide itself is not an active catalyst, but it becomes active after exposure to hydrogen whereupon it converts to platinum black, which is responsible for reactions. Preparation Adams' catalyst is prepared from chloroplatinic acid H2PtCl6 or ammonium chloroplatinate, (NH4)2PtCl6, by fusion with sodium nitrate. The first published preparation was reported by V. Voorhees and Roger Adams. The procedure involves first preparing a platinum nitrate which is then heated to expel nitrogen oxides. :H2PtCl6 + 6 NaNO3 → Pt(NO3)4 + 6 NaCl (aq) + 2 HNO3 :Pt(NO3)4 → PtO2 + 4 NO2 + O2 The resulting brown cake is washed with water to free it from nitrates. The catalyst can either be used as is or dried and stored in a desiccator for later use. Pl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rutile Structure
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite. Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum. Rutile derives its name from the Latin ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner. Occurrence Rutile is a common accessory mineral in high-temperature and high-pressure metamorphic rocks and in igneous rocks. Thermodynamically, ru ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Microelectrode
A microelectrode is an electrode used in electrophysiology either for recording neural signals or for the electrical stimulation of nervous tissue (they were first developed by Ida Hyde in 1921). Pulled glass pipettes with tip diameters of 0.5 μm or less are usually filled with 3 molars potassium chloride solution as the electrical conductor. When the tip penetrates a cell membrane the lipids in the membrane seal onto the glass, providing an excellent electrical connection between the tip and the interior of the cell, which is apparent because the microelectrode becomes electrically negative compared to the extracellular solution. There are also microelectrodes made with insulated metal wires, made from inert metals with high Young modulus such as tungsten, stainless steel, or platinum-iridium alloy and coated with glass or polymer insulator with exposed conductive tips. These are mostly used for recording from the external side of the cell membrane. More recent advances i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophysiology
Electrophysiology (from Greek , ''ēlektron'', "amber" etymology of "electron"">Electron#Etymology">etymology of "electron" , ''physis'', "nature, origin"; and , '' -logia'') is the branch of physiology that studies the electrical properties of biological cells and tissues. It involves measurements of voltage changes or electric current or manipulations on a wide variety of scales from single ion channel proteins to whole organs like the heart. In neuroscience, it includes measurements of the electrical activity of neurons, and, in particular, action potential activity. Recordings of large-scale electric signals from the nervous system, such as electroencephalography, may also be referred to as electrophysiological recordings. They are useful for electrodiagnosis and monitoring. Definition and scope Classical electrophysiological techniques Principle and mechanisms Electrophysiology is the branch of physiology that pertains broadly to the flow of ions (ion current) in biologi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iridium Trichloride
Iridium(III) chloride is the inorganic compound with the formula IrCl3. The anhydrous compound is relatively rare, but the related hydrate is useful for preparing other iridium compounds. The anhydrous salt is a dark green crystalline solid. More commonly encountered is the trihydrate IrCl3(H2O)3. Preparation Iridium is separated from the other platinum group metals as crystalline ammonium hexachloroiridate, (NH4)2 rCl6 which can be reduced to iridium metal in a stream of hydrogen. The spongy Ir thus produced reacts with chlorine at 300–400 °C to give iridium(III) chloride. Hydrated iridium trichloride is obtained by heating hydrated iridium(III) oxide with hydrochloric acid. Structure Like the related rhodium compound, IrCl3 adopts the structure seen for aluminium chloride. This is the monoclinic α polymorph. A rhombohedral β polymorph also exists. Both polymorphs have effectively the same anion lattice but differ in the octahedral interstices the iridium ions o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iridium Compounds
Iridium compounds are compounds containing the element iridium (Ir). Iridium forms compounds in oxidation states between −3 and +9, but the most common oxidation states are +1, +3, and +4. Well-characterized compounds containing iridium in the +6 oxidation state include and the oxides and . iridium(VIII) oxide () was generated under matrix isolation conditions at 6 K in argon. The highest oxidation state (+9), which is also the highest recorded for ''any'' element, is found in gaseous . Oxides Only one binary oxide is well-characterized: Iridium dioxide, . It is a blue-black solid. The compound adopts the TiO2 rutile structure, featuring six coordinate iridium and three coordinate oxygen. It adopts the fluorite structure. A sesquioxide, , has been described as a blue-black powder, which is oxidized to by . The corresponding disulfides, diselenides, sesquisulfides, and sesquiselenides are known, as well as . Another oxide, iridium tetroxide, is also known, with iridium ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

x.jpg)