|
Hydrogen Ozonide
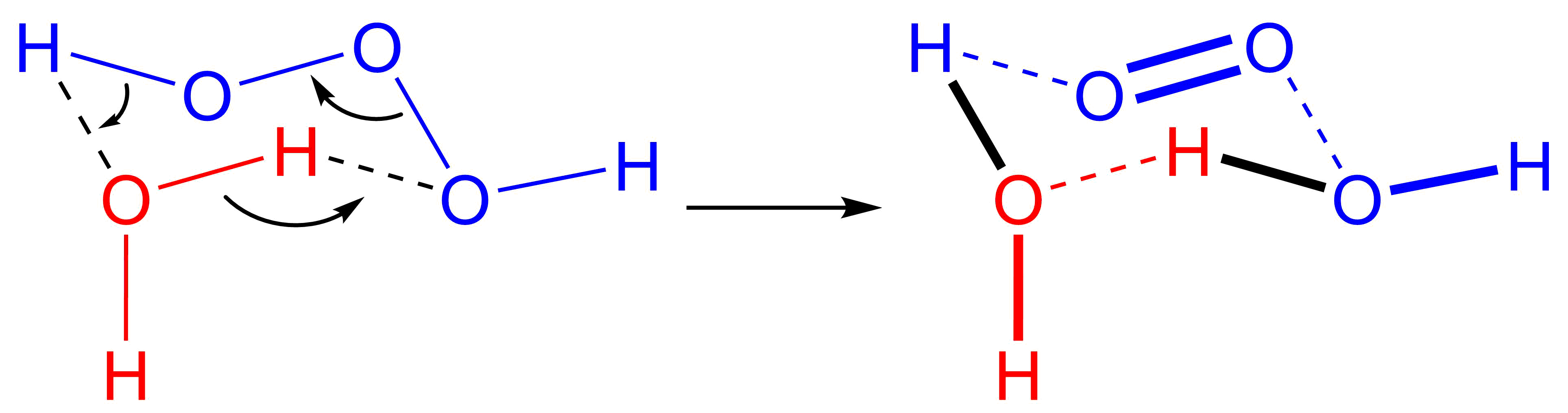
Hydrogen ozonide () is a radical molecule consisting of a hydrogen atom covalently bonded to an ozonide unit. It is possibly produced in the reaction of the hydroxyl radical with dioxygen: OH• + O2 → HO3•. It has been detected in a mass spectrometer Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a '' mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is us ... experiment using HO3+ ( protonated ozone) as precursor. References Extra reading * * {{inorganic-compound-stub Hydrogen compounds Ozonides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Trioxide
Trioxidane (systematically named μ-trioxidanediidodihydrogen), also called dihydrogen trioxide, is an inorganic compound with the chemical formula (can be written as or ). It is one of the unstable hydrogen polyoxides. In aqueous solutions, trioxidane decomposes to form water and singlet oxygen: The reverse reaction, the addition of singlet oxygen to water, typically does not occur in part due to the scarcity of singlet oxygen. In biological systems, however, ozone is known to be generated from singlet oxygen, and the presumed mechanism is an antibody-catalyzed production of trioxidane from singlet oxygen. Preparation Trioxidane can be obtained in small, but detectable, amounts in reactions of ozone and hydrogen peroxide, or by the electrolysis of water. Larger quantities have been prepared by the reaction of ozone with organic reducing agents at low temperatures in a variety of organic solvents, such as the anthraquinone process. It is also formed during the decomposition ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Superoxide
The hydroperoxyl radical, also known as the hydrogen superoxide, is the protonated form of superoxide with the chemical formula HO2. This species plays an important role in the atmosphere and as a reactive oxygen species in cell biology. Structure and reactions The molecule has a bent structure. The superoxide anion, , and the hydroperoxyl radical exist in equilibrium in aqueous solution: : + H2O HO2 + OH− The p''K''a of HO2 is 4.88. Therefore, about 0.3% of any superoxide present in the cytosol of a typical cell is in the protonated form. It oxidizes nitric oxide to nitrogen dioxide: :NO + HO2 → NO2 + HO Reactive oxygen species in biology Together with its conjugate base superoxide, hydroperoxyl is an important reactive oxygen species. Unlike , which has reducing properties, HO2 can act as an oxidant in a number of biologically important reactions, such as the abstraction of hydrogen atoms from tocopherol and polyunstaturated fatty acids in the lipid bilay ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Polyoxide
Hydrogen polyoxides (also known as oxidanes, oxohydrogens, or oxyhydrogens) are chemical compounds that consist only of hydrogen and oxygen atoms, are bonded exclusively by single bonds (i.e., they are saturated), and are acyclic (have molecular structures containing no cycles or loops). They can, therefore, be classed as hydrogen chalcogenides. The simplest possible stable hydrogen polyoxide (the parent molecule) is water, H2O. The general structure of the class of molecules is some number of oxygen atoms single-bonded to each other in a chain. The oxygen atom at each end of this oxygen skeleton is attached to a hydrogen atom. Thus, these compounds form a homologous series with chemical formula in which the members differ by a constant relative molecular mass of 16 (the mass of each additional oxygen atom). The number of oxygen atoms is used to define the size of the hydrogen polyoxide (e.g., hydrogen pentoxide contains a five-oxygen backbone). An oxidanyl group is a fun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protonated Ozone
Protonated ozone is a hydrogen polyoxide having the molecular formula (also written ). It is a cationic structure consisting of an ozone unit with a hydrogen atom attached to one end. This substance is proposed to exist as an intermediate in several interstellar, atmospheric,and synthetic chemical processes. It has been synthesized in mass spectrometer experiments by protonation of ozone using various strong acids. Related experiments have used it as the precursor for generating hydrogen ozonide Hydrogen ozonide () is a radical molecule consisting of a hydrogen atom covalently bonded to an ozonide unit. It is possibly produced in the reaction of the hydroxyl radical with dioxygen: OH• + O2 → HO3•. It has been detected in a mass .... References {{inorganic-compound-stub Hydrogen compounds Oxycations ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical (chemistry)
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes. A notable example of a radical is the hydroxyl radical (HO·), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (꞉) which have two unpaired electrons. Radicals may be generated in a number of ways, but typical methods involve redox reactions. Ionizing radiation, heat, electrical discharges, and electrolysis are known to produce radicals. Radicals are intermediates in many chemical reactions, more so than is apparent from the balanced equations. Radicals are important in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. A majority of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Covalently Bonded
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding. For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full valence shell, corresponding to a stable electronic configuration. In organic chemistry, covalent bonding is much more common than ionic bonding. Covalent bonding also includes many kinds of interactions, including σ-bonding, π-bonding, metal-to-metal bonding, agostic interactions, bent bonds, three-center two-electron bonds and three-center four-electron bonds. The term ''covalent bond'' dates from 1939. The prefix ''co-'' means ''jointly, associated in action, partnered to a lesser degree, '' etc.; thus a "co-valent bond", in essence, means that the atoms share " valence", such a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ozonide
Ozonide is the polyatomic anion . Cyclic organic compounds formed by the addition of ozone () to an alkene are also called ozonides. Ionic ozonides Inorganic ozonides are dark red salts. The anion has the bent shape of the ozone molecule. Inorganic ozonides are formed by burning potassium, rubidium, or caesium in ozone, or by treating the alkali metal hydroxide with ozone; this yields potassium ozonide, rubidium ozonide, and caesium ozonide respectively. They are very sensitive explosives that have to be handled at low temperatures in an atmosphere consisting of an inert gas. Lithium and sodium ozonide are extremely labile and must be prepared by low-temperature ion exchange starting from . Sodium ozonide, , which is prone to decomposition into NaOH and , was previously thought to be impossible to obtain in pure form. However, with the help of cryptands and methylamine, pure sodium ozonide may be obtained as red crystals isostructural to . Ionic ozonides are being investigate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyl Radical
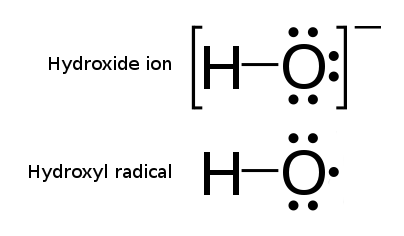
The hydroxyl radical is the diatomic molecule . The hydroxyl radical is very stable as a dilute gas, but it decays very rapidly in the condensed phase. It is pervasive in some situations. Most notably the hydroxyl radicals are produced from the decomposition of hydroperoxides (ROOH) or, in atmospheric chemistry, by the reaction of excited atomic oxygen with water. It is also important in the field of radiation chemistry, since it leads to the formation of hydrogen peroxide and oxygen, which can enhance corrosion and SCC in coolant systems subjected to radioactive environments. In organic synthesis, hydroxyl radicals are most commonly generated by photolysis of 1-hydroxy-2(1''H'')-pyridinethione. Notation The unpaired electron of the hydroxyl radical is officially represented by a middle dot, •, beside the O. Biology Hydroxyl radicals can occasionally be produced as a byproduct of immune action. Macrophages and microglia most frequently generate this compound when exp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dioxygen
There are several known allotropes of oxygen. The most familiar is molecular oxygen (O2), present at significant levels in Earth's atmosphere and also known as dioxygen or triplet oxygen. Another is the highly reactive ozone (O3). Others are: *Atomic oxygen (O1), a free radical. *Singlet oxygen (O2*), one of two metastable states of molecular oxygen. *Tetraoxygen (O4), another metastable form. *Solid oxygen, existing in six variously colored phases, of which one is and another one metallic. Atomic oxygen Atomic oxygen, denoted O(3P) or O(3P), is very reactive, as the single atoms of oxygen tend to quickly bond with nearby molecules. On Earth's surface, it exists naturally for a very short time. In outer space, the presence of ample ultraviolet radiation results in a low Earth orbit atmosphere in which 96% of the oxygen occurs in atomic form. Ryan D. McCulla, Saint Louis University (2010). /acswebcontent.acs.org/prfar/2010/reports/P11141.html "Atomic Oxygen O(3P): Photog ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass Spectrometer
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used in many different fields and is applied to pure samples as well as complex mixtures. A mass spectrum is a type of plot of the ion signal as a function of the mass-to-charge ratio. These spectra are used to determine the elemental or isotopic signature of a sample, the masses of particles and of molecules, and to elucidate the chemical identity or structure of molecules and other chemical compounds. In a typical MS procedure, a sample, which may be solid, liquid, or gaseous, is ionized, for example by bombarding it with a beam of electrons. This may cause some of the sample's molecules to break up into positively charged fragments or simply become positively charged without fragmenting. These ions (fragments) are then separated according ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protonated Ozone
Protonated ozone is a hydrogen polyoxide having the molecular formula (also written ). It is a cationic structure consisting of an ozone unit with a hydrogen atom attached to one end. This substance is proposed to exist as an intermediate in several interstellar, atmospheric,and synthetic chemical processes. It has been synthesized in mass spectrometer experiments by protonation of ozone using various strong acids. Related experiments have used it as the precursor for generating hydrogen ozonide Hydrogen ozonide () is a radical molecule consisting of a hydrogen atom covalently bonded to an ozonide unit. It is possibly produced in the reaction of the hydroxyl radical with dioxygen: OH• + O2 → HO3•. It has been detected in a mass .... References {{inorganic-compound-stub Hydrogen compounds Oxycations ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Compounds
Hydrogen compounds are compounds containg the element hydrogen. In these compounds, hydrogen can form in the +1 and -1 oxidation states. Hydrogen can form compounds both ionically and in covalent substances. It is a part of many organic compounds such as hydrocarbons as well as water and other organic substances. The ion is often called a proton because it has one proton and no electrons, although the proton does not move freely. Brønsted–Lowry acids are capable of donating ions to bases. Covalent and organic compounds While is not very reactive under standard conditions, it does form compounds with most elements. Hydrogen can form compounds with elements that are more electronegative, such as halogens (F, Cl, Br, I), or oxygen; in these compounds hydrogen takes on a partial positive charge. When bonded to a more electronegative element, particularly fluorine, oxygen, or nitrogen, hydrogen can participate in a form of medium-strength noncovalent bonding with another elect ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

.jpg)
