|
Heat Transfer Agent
A coolant is a substance, typically liquid, that is used to reduce or regulate the temperature of a system. An ideal coolant has high thermal capacity, low viscosity, is low-cost, non-toxic, chemically inert and neither causes nor promotes corrosion of the cooling system. Some applications also require the coolant to be an electrical insulator. While the term "coolant" is commonly used in automotive and HVAC applications, in industrial processing heat-transfer fluid is one technical term more often used in high temperature as well as low-temperature manufacturing applications. The term also covers cutting fluids. Industrial cutting fluid has broadly been classified as water-soluble coolant and neat cutting fluid. Water-soluble coolant is oil in water emulsion. It has varying oil content from nil oil (synthetic coolant). This coolant can either keep its phase and stay liquid or gaseous, or can undergo a phase transition, with the latent heat adding to the cooling efficiency. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
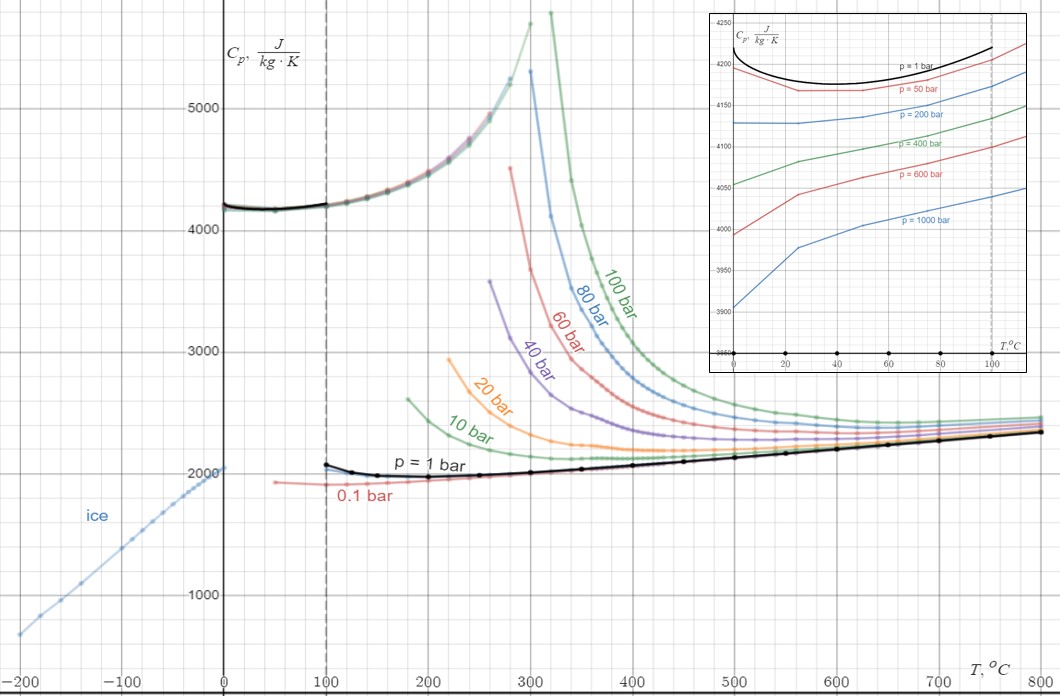
Thermal Capacity
Heat capacity or thermal capacity is a physical property of matter, defined as the amount of heat to be supplied to an object to produce a unit change in its temperature. The SI unit of heat capacity is joule per kelvin (J/K). Heat capacity is an extensive property. The corresponding intensive property is the specific heat capacity, found by dividing the heat capacity of an object by its mass. Dividing the heat capacity by the amount of substance in moles yields its molar heat capacity. The volumetric heat capacity measures the heat capacity per volume. In architecture and civil engineering, the heat capacity of a building is often referred to as its thermal mass. Definition Basic definition The heat capacity of an object, denoted by C, is the limit : C = \lim_\frac, where \Delta Q is the amount of heat that must be added to the object (of mass ''M'') in order to raise its temperature by \Delta T. The value of this parameter usually varies considerably depending on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy. Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons. In the early universe, the formation of protons, the nuclei of hydrogen, occurred during the first second after the Big Bang. The emergence of neutral hydrogen atoms throughout the universe occurred about 370,000 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transparent to visible light but absorbs infrared radiation, acting as a greenhouse gas. It is a trace gas in Earth's atmosphere at 421 parts per million (ppm), or about 0.04% by volume (as of May 2022), having risen from pre-industrial levels of 280 ppm. Burning fossil fuels is the primary cause of these increased CO2 concentrations and also the primary cause of climate change.IPCC (2022Summary for policy makersiClimate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA Carbon dioxide is soluble in water and is found in groundwater, lakes, ice caps, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron Activation
Neutron activation is the process in which neutron radiation induces radioactivity in materials, and occurs when atomic nuclei capture free neutrons, becoming heavier and entering excited states. The excited nucleus decays immediately by emitting gamma rays, or particles such as beta particles, alpha particles, fission products, and neutrons (in nuclear fission). Thus, the process of neutron capture, even after any intermediate decay, often results in the formation of an unstable activation product. Such radioactive nuclei can exhibit half-lives ranging from small fractions of a second to many years. Neutron activation is the only common way that a stable material can be induced into becoming intrinsically radioactive. All naturally occurring materials, including air, water, and soil, can be induced (activated) by neutron capture into some amount of radioactivity in varying degrees, as a result of the production of neutron-rich radioisotopes. Some atoms require more than one neu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron Capture
Neutron capture is a nuclear reaction in which an atomic nucleus and one or more neutrons collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged protons, which are repelled electrostatically. Neutron capture plays a significant role in the cosmic nucleosynthesis of heavy elements. In stars it can proceed in two ways: as a rapid process (r-process) or a slow process (s-process). Nuclei of masses greater than 56 cannot be formed by thermonuclear reactions (i.e., by nuclear fusion) but can be formed by neutron capture. Neutron capture on protons yields a line at 2.223 MeV predicted and commonly observed in solar flares. Neutron capture at small neutron flux At small neutron flux, as in a nuclear reactor, a single neutron is captured by a nucleus. For example, when natural gold (197Au) is irradiated by neutrons (n), the isotope 198Au is formed in a highly excited state, and quickly deca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
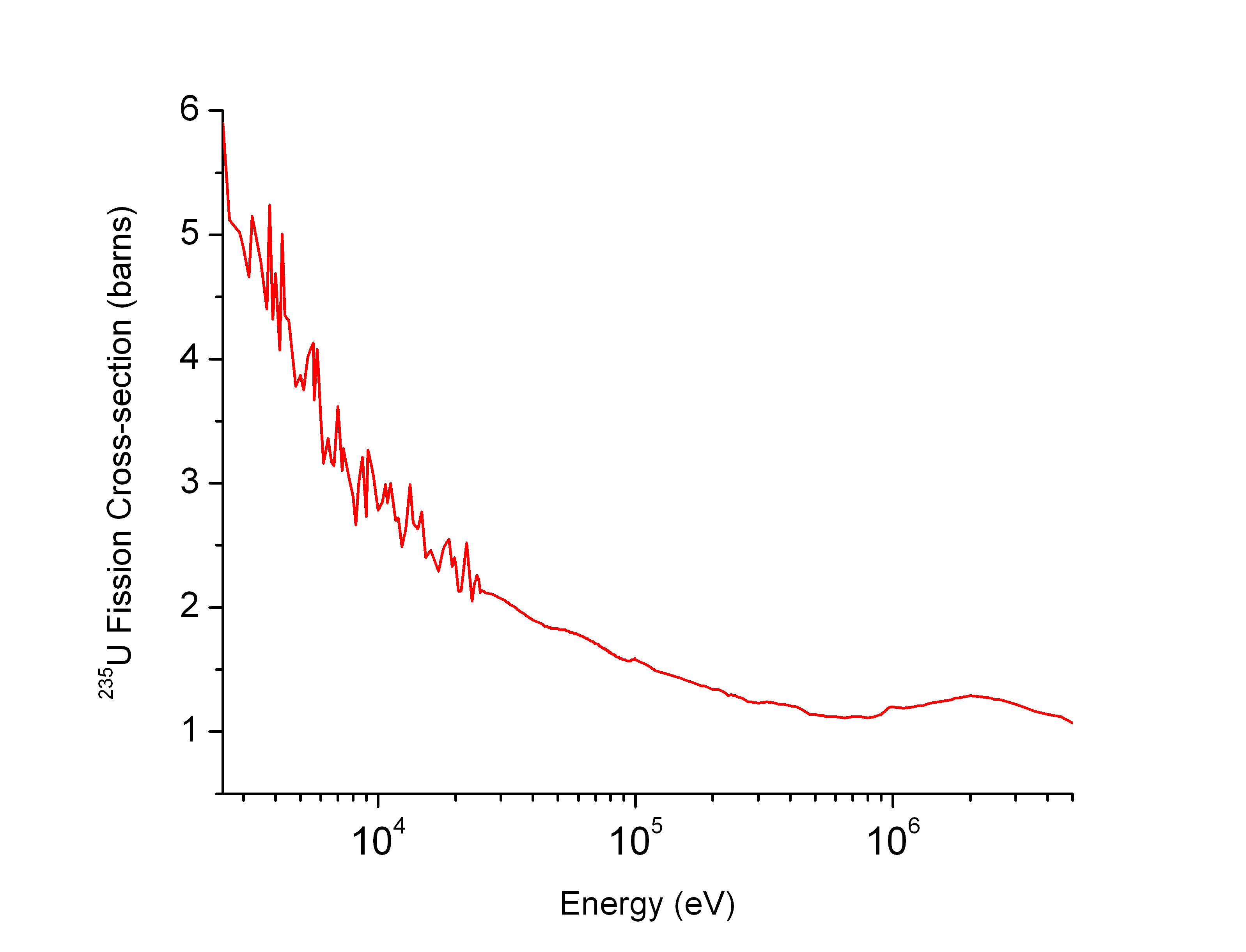
Neutron Cross Section
In nuclear physics, the concept of a neutron cross section is used to express the likelihood of interaction between an incident neutron and a target nucleus. The neutron cross section σ can be defined as the area in cm2 for which the number of neutron-nuclei reactions taking place is equal to the product of the number of incident neutrons that would pass through the area and the number of target nuclei. In conjunction with the neutron flux, it enables the calculation of the reaction rate, for example to derive the thermal power of a nuclear power plant. The standard unit for measuring the cross section is the barn, which is equal to 10−28 m2 or 10−24 cm2. The larger the neutron cross section, the more likely a neutron will react with the nucleus. An isotope (or nuclide) can be classified according to its neutron cross section and how it reacts to an incident neutron. Nuclides that tend to absorb a neutron and either decay or keep the neutron in its nucleus are neutron absor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium
Helium (from el, ἥλιος, helios, lit=sun) is a chemical element with the symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas and the first in the noble gas group in the periodic table. Its boiling and melting point are the lowest among all the elements. It is the second lightest and second most abundant element in the observable universe (hydrogen is the lightest and most abundant). It is present at about 24% of the total elemental mass, which is more than 12 times the mass of all the heavier elements combined. Its abundance is similar to this in both the Sun and in Jupiter, due to the very high nuclear binding energy (per nucleon) of helium-4, with respect to the next three elements after helium. This helium-4 binding energy also accounts for why it is a product of both nuclear fusion and radioactive decay. The most common isotope of helium in the universe is helium-4, the vast majority of which was formed during t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gas-cooled Reactor
A gas-cooled reactor (GCR) is a nuclear reactor that uses graphite as a neutron moderator and a gas (carbon dioxide or helium in extant designs) as coolant. Although there are many other types of reactor cooled by gas, the terms ''GCR'' and to a lesser extent ''gas cooled reactor'' are particularly used to refer to this type of reactor. The GCR was able to use natural uranium as fuel, enabling the countries that developed them to fabricate their own fuel without relying on other countries for supplies of enriched uranium, which was at the time of their development in the 1950s only available from the United States or the Soviet Union. The Canadian CANDU reactor, using heavy water as a moderator, was designed with the same goal of using natural uranium fuel for similar reasons. Design considerations Historically thermal spectrum graphite moderated gas cooled reactors mostly competed with light water reactors, ultimately losing out to them after having seen some deployment in Brit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Reactor Coolant
A nuclear reactor coolant is a coolant in a nuclear reactor used to remove heat from the nuclear reactor core and transfer it to electrical generators and the Environment (systems), environment. Frequently, a chain of two coolant loops are used because the primary coolant loop takes on short-term radioactivity from the reactor. Water Almost all currently operating nuclear power plants are light water reactors using ordinary water under high pressure as coolant and neutron moderator. About 1/3 are boiling water reactors where the primary coolant undergoes phase transition to steam inside the reactor. About 2/3 are pressurized water reactors at even higher pressure. Current reactors stay under the critical point (thermodynamics), critical point at around 374 °C and 218 Bar (unit), bar where the distinction between liquid and gas disappears, which limits thermal efficiency, but the proposed supercritical water reactor would operate above this point. Heavy water reactors use de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inert Gas
An inert gas is a gas that does not readily undergo chemical reactions with other chemical substances and therefore does not readily form chemical compounds. The noble gases often do not react with many substances and were historically referred to as the inert gases. Inert gases are used generally to avoid unwanted chemical reactions degrading a sample. These undesirable chemical reactions are often oxidation and hydrolysis reactions with the oxygen and moisture in air. The term ''inert gas'' is context-dependent because several of the noble gases can be made to react under certain conditions. Purified argon gas is the most commonly used inert gas due to its high natural abundance (78.3% N2, 1% Ar in air) and low relative cost. Unlike noble gases, an inert gas is not necessarily elemental and is often a compound gas. Like the noble gases, the tendency for non-reactivity is due to the valence, the outermost electron shell, being complete in all the inert gases. This is a tendency, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen-cooled Turbogenerator
A hydrogen-cooled turbo generator is a turbo generator with gaseous hydrogen as a coolant. Hydrogen-cooled turbo generators are designed to provide a low- drag atmosphere and cooling for single-shaft and combined-cycle applications in combination with steam turbines. Because of the high thermal conductivity and other favorable properties of hydrogen gas, this is the most common type in its field today. History Based on the air-cooled turbo generator, gaseous hydrogen first went into service as the coolant in a hydrogen-cooled turbo generator in October 1937, at the Dayton Power & Light Co. in Dayton, Ohio. Design The use of gaseous hydrogen as a coolant is based on its properties, namely low density, high specific heat, and the highest thermal conductivity (at 0.168 W/(m·K)) of all gases; it is 7 to 10 times better at cooling than air. Another advantage of hydrogen is its easy detection by hydrogen sensors. A hydrogen-cooled generator can be significantly smaller, and therefor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Windage
Windage is a term used in aerodynamics, firearms ballistics, and automobiles. Usage Aerodynamics Windage is a force created on an object by friction when there is relative movement between air and the object. Windage loss is the reduction in efficiency due to windage forces. For example, electric motors are affected by friction between the rotor and air. Large alternators have significant losses due to windage. To reduce losses, hydrogen gas may be used, since it is less dense. There are two causes of windage: # The object is moving and being slowed by resistance from the air. # A wind is blowing, producing a force on the object. The term can refer to: * The effect of the force, for example the deflection of a missile or an aircraft by a cross wind. * The area and shape of the object that make it susceptible to friction, for example those parts of a boat that are exposed to the wind. Aerodynamic streamlining can be used to reduce windage. There is a hydrodynamic effect si ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |