|
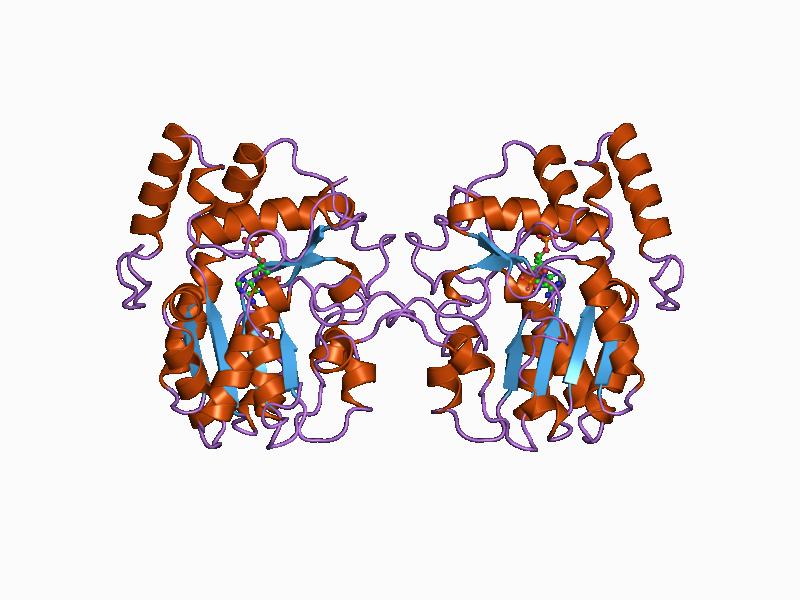
Guanylate Kinase
In enzymology, a guanylate kinase () is an enzyme that catalyzes the chemical reaction : ATP + GMP \rightleftharpoons ADP + GDP Thus, the two substrates of this enzyme are ATP and GMP, whereas its two product (chemistry), products are adenosine diphosphate, ADP and GDP Gross domestic product (GDP) is a monetary measure of the market value of all the final goods and services produced and sold (not resold) in a specific time period by countries. Due to its complex and subjective nature this measure is ofte .... This enzyme belongs to the family of transferases, specifically those transferring phosphorus-containing groups (phosphotransferases) with a phosphate group as acceptor. This enzyme participates in purine metabolism. Guanylate kinase catalyzes the ATP-dependent phosphorylation of GMP into GDP. It is essential for recycling GMP and indirectly, Cyclic guanosine monophosphate, cGMP. In prokaryotes (such as ''Escherichia coli''), lower eukaryotes (such as yeast) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzymology
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transferase
A transferase is any one of a class of enzymes that catalyse the transfer of specific functional groups (e.g. a methyl or glycosyl group) from one molecule (called the donor) to another (called the acceptor). They are involved in hundreds of different biochemical pathways throughout biology, and are integral to some of life's most important processes. Transferases are involved in myriad reactions in the cell. Three examples of these reactions are the activity of coenzyme A (CoA) transferase, which transfers thiol esters, the action of N-acetyltransferase, which is part of the pathway that metabolizes tryptophan, and the regulation of pyruvate dehydrogenase (PDH), which converts pyruvate to acetyl CoA. Transferases are also utilized during translation. In this case, an amino acid chain is the functional group transferred by a peptidyl transferase. The transfer involves the removal of the growing amino acid chain from the tRNA molecule in the A-site of the ribosome and its subse ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
List Of Enzymes
This article lists enzymes by their classification in the International Union of Biochemistry and Molecular Biology's Enzyme Commission (EC) numbering system. * List of EC numbers (EC 5) * List of EC numbers (EC 6) :Oxidoreductases (EC 1) (Oxidoreductase) *Dehydrogenase * Luciferase *DMSO reductase :EC 1.1 (act on the CH-OH group of donors) * :EC 1.1.1 (with NAD+ or NADP+ as acceptor) ** Alcohol dehydrogenase (NAD) ** Alcohol dehydrogenase (NADP) **Homoserine dehydrogenase ** Aminopropanol oxidoreductase **Diacetyl reductase **Glycerol dehydrogenase **Propanediol-phosphate dehydrogenase ** glycerol-3-phosphate dehydrogenase (NAD+) ** D-xylulose reductase **L-xylulose reductase **Lactate dehydrogenase **Malate dehydrogenase **Isocitrate dehydrogenase ** HMG-CoA reductase * :EC 1.1.2 (with a cytochrome as acceptor) * :EC 1.1.3 (with oxygen as acceptor) **Glucose oxidase **L-gulonolactone oxidase **Thiamine oxidase **Xanthine oxidase * :EC 1.1.4 (with a disul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) is a strain of coronavirus that causes COVID-19 (coronavirus disease 2019), the respiratory illness responsible for the ongoing COVID-19 pandemic. The virus previously had a provisional name, 2019 novel coronavirus (2019-nCoV), and has also been called the human coronavirus 2019 (HCoV-19 or hCoV-19). First identified in the city of Wuhan, Hubei, China, the World Health Organization declared the outbreak a public health emergency of international concern on January 30, 2020, and a pandemic on March 11, 2020. SARS‑CoV‑2 is a positive-sense single-stranded RNA virus that is contagious in humans. SARS‑CoV‑2 is a virus of the species ''severe acute respiratory syndrome–related coronavirus'' (SARSr-CoV), related to the SARS-CoV-1 virus that caused the 2002–2004 SARS outbreak. Despite its close relation to SARS-CoV-1, its closest known relatives, with which it forms a sister group, are the derived SARS ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Andreas Dräger
Andreas Dräger (; born 20 September 1980) is a German bioinformatician who leads the research group for Computational Systems Biology of Infections and Antimicrobial-Resistant Pathogens at the University of Tübingen. Education In high school, Dräger was fascinated by computer science and recent advancements in genetics and biotechnology in the late 1990s. When he learned about a new degree program that allowed combining those technologies, he was immediately inspired. So, Dräger studied bioinformatics at the Martin Luther University of Halle-Wittenberg in Halle (Saale) from 2000 to 2006. He worked as an intern for genome sequencing at the Max Planck Institute for Molecular Genetics, Berlin. He prepared his thesis about heavy-metal-resistant bacteria at the microbiology department of the University of Illinois at Chicago. Dräger earned his doctorate at the Center for Bioinformatics in Tübingen (ZBIT), focusing on the dynamic simulation of metabolic networks in a virtual ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Systems Biology
Systems biology is the computational modeling, computational and mathematical analysis and modeling of complex biological systems. It is a biology-based interdisciplinary field of study that focuses on complex interactions within biological systems, using a holistic approach (holism instead of the more traditional reductionist, reductionism) to biological research. Particularly from the year 2000 onwards, the concept has been used widely in biology in a variety of contexts. The Human Genome Project is an example of applied systems thinking in biology which has led to new, collaborative ways of working on problems in the biological field of genetics. One of the aims of systems biology is to model and discover emergent property, emergent properties, properties of cell (biology), cells, tissue (biology), tissues and organisms functioning as a system whose theoretical description is only possible using techniques of systems biology. These typically involve metabolic networks or cell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vaccinia
''Vaccinia virus'' (VACV or VV) is a large, complex, enveloped virus belonging to the poxvirus family. It has a linear, double-stranded DNA genome approximately 190 kbp in length, which encodes approximately 250 genes. The dimensions of the virion are roughly 360 × 270 × 250 nm, with a mass of approximately 5–10 fg. The vaccinia virus is the source of the modern smallpox vaccine, which the World Health Organisation used to eradicate smallpox in a global vaccination campaign in 1958–1977. Although ''smallpox'' no longer exists in the wild, ''vaccinia'' virus is still studied widely by scientists as a tool for gene therapy and genetic engineering. Smallpox had been an endemic human disease that had a 30% fatality rate. In 1796, the British doctor Edward Jenner proved that an infection with the relatively mild cowpox virus would also confer immunity to the deadly smallpox. Jenner referred to cowpox as ''variolae vaccinae'' (smallpox of the cow). However, the origins ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclic Guanosine Monophosphate
Cyclic guanosine monophosphate (cGMP) is a cyclic nucleotide derived from guanosine triphosphate (GTP). cGMP acts as a second messenger much like cyclic AMP. Its most likely mechanism of action is activation of intracellular protein kinases in response to the binding of membrane-impermeable peptide hormones to the external cell surface. Synthesis Guanylate cyclase (GC) catalyzes cGMP synthesis. This enzyme converts GTP to cGMP. Peptide hormones such as the atrial natriuretic factor activate membrane-bound GC, while soluble GC (sGC) is typically activated by nitric oxide to stimulate cGMP synthesis. sGC can be inhibited by ODQ (1H-,2,4xadiazolo ,3-auinoxalin-1-one). Functions cGMP is a common regulator of ion channel conductance, glycogenolysis, and cellular apoptosis. It also relaxes smooth muscle tissues. In blood vessels, relaxation of vascular smooth muscles leads to vasodilation and increased blood flow. At presynaptic terminals in the striatum, cGMP controls the e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Purine Metabolism
Purine metabolism refers to the metabolic pathways to synthesize and break down purines that are present in many organisms. Biosynthesis Purines are biologically synthesized as nucleotides and in particular as ribotides, i.e. bases attached to ribose 5-phosphate. Both adenine and guanine are derived from the nucleotide inosine monophosphate (IMP), which is the first compound in the pathway to have a completely formed purine ring system. IMP Inosine monophosphate is synthesized on a pre-existing ribose-phosphate through a complex pathway (as shown in the figure on the right). The source of the carbon and nitrogen atoms of the purine ring, 5 and 4 respectively, come from multiple sources. The amino acid glycine contributes all its carbon (2) and nitrogen (1) atoms, with additional nitrogen atoms from glutamine (2) and aspartic acid (1), and additional carbon atoms from formyl groups (2), which are transferred from the coenzyme tetrahydrofolate as 10-formyltetrahydrofolate, and a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphotransferase
Phosphotransferases are a category of enzymes ( EC number 2.7) that catalyze phosphorylation reactions. The general form of the reactions they catalyze is: :A-P + B \rightleftharpoons B-P + A Where ''P'' is a phosphate group and A and B are the donating and accepting molecules, respectively. Classification Phosphotransferases are generally classified according to the acceptor molecule. , Classification in this article follows the rules of Enzyme Nomenclature of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology (NC-IUBMB). *EC 2.7.1 Phosphotransferases with an as acceptor *EC 2.7.2 Phosphotransferases with a |
Adenosine Diphosphate
Adenosine diphosphate (ADP), also known as adenosine pyrophosphate (APP), is an important organic compound in metabolism and is essential to the flow of energy in living cells. ADP consists of three important structural components: a sugar backbone attached to adenine and two phosphate groups bonded to the 5 carbon atom of ribose. The diphosphate group of ADP is attached to the 5’ carbon of the sugar backbone, while the adenine attaches to the 1’ carbon. ADP can be interconverted to adenosine triphosphate (ATP) and adenosine monophosphate (AMP). ATP contains one more phosphate group than does ADP. AMP contains one fewer phosphate group. Energy transfer used by all living things is a result of dephosphorylation of ATP by enzymes known as ATPases. The cleavage of a phosphate group from ATP results in the coupling of energy to metabolic reactions and a by-product of ADP. ATP is continually reformed from lower-energy species ADP and AMP. The biosynthesis of ATP is achieved through ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |