|
Glycoside Hydrolase Family 31
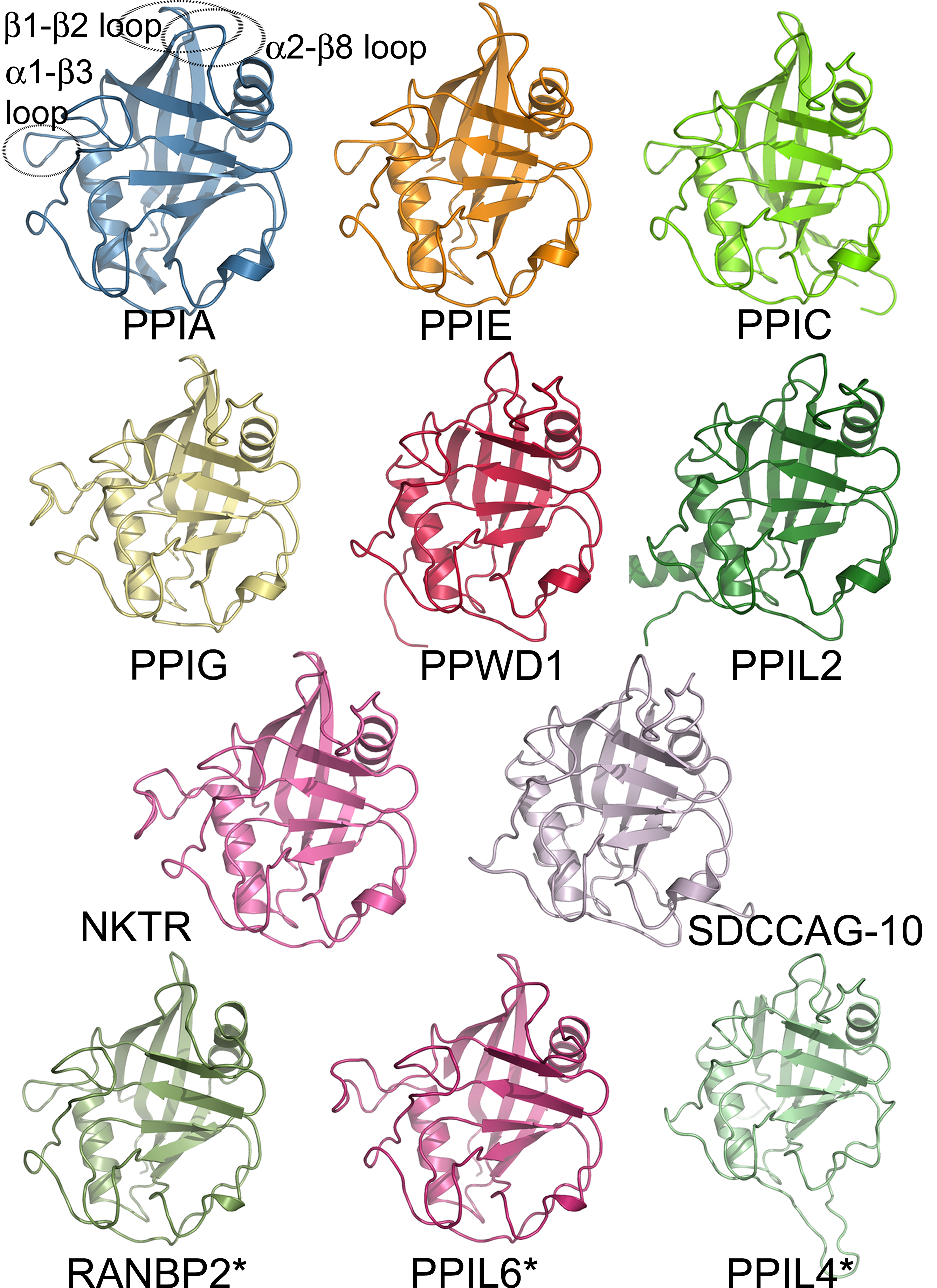
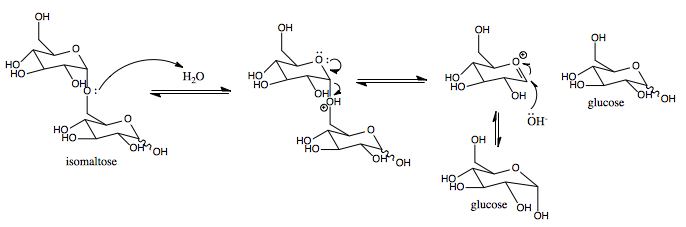
In molecular biology, glycoside hydrolase family 31 is a family of glycoside hydrolases. Glycoside hydrolases are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A classification system for glycoside hydrolases, based on sequence similarity, has led to the definition of >100 different families. This classification is available on the CAZy web site, and also discussed at CAZypedia, an online encyclopedia of carbohydrate active enzymes. Glycoside hydrolase family 3CAZY GH_31comprises enzymes with several known activities; alpha-glucosidase (), alpha-galactosidase (); glucoamylase (), sucrase-isomaltase () (); alpha-xylosidase (); alpha-glucan lyase (). Glycoside hydrolase family 31 groups a number of glycosyl hydrolases on the basis of sequence similarities An aspartic acid has been implicated in the catalytic activity of sucrase, isomaltase, and lysosomal alpha-glucosidas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Family
A protein family is a group of evolutionarily related proteins. In many cases, a protein family has a corresponding gene family, in which each gene encodes a corresponding protein with a 1:1 relationship. The term "protein family" should not be confused with Family (biology), family as it is used in taxonomy. Proteins in a family descend from a common ancestor and typically have similar protein structure, three-dimensional structures, functions, and significant Sequence homology, sequence similarity. The most important of these is sequence similarity (usually amino-acid sequence), since it is the strictest indicator of homology and therefore the clearest indicator of common ancestry. A fairly well developed framework exists for evaluating the significance of similarity between a group of sequences using sequence alignment methods. Proteins that do not share a common ancestor are very unlikely to show statistically significant sequence similarity, making sequence alignment a powerf ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycoside Hydrolase
Glycoside hydrolases (also called glycosidases or glycosyl hydrolases) catalyze the hydrolysis of glycosidic bonds in complex sugars. They are extremely common enzymes with roles in nature including degradation of biomass such as cellulose (cellulase), hemicellulose, and starch (amylase), in anti-bacterial defense strategies (e.g., lysozyme), in pathogenesis mechanisms (e.g., viral neuraminidases) and in normal cellular function (e.g., trimming mannosidases involved in N-linked glycoprotein biosynthesis). Together with glycosyltransferases, glycosidases form the major catalytic machinery for the synthesis and breakage of glycosidic bonds. Occurrence and importance Glycoside hydrolases are found in essentially all domains of life. In prokaryotes, they are found both as intracellular and extracellular enzymes that are largely involved in nutrient acquisition. One of the important occurrences of glycoside hydrolases in bacteria is the enzyme beta-galactosidase (LacZ), which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycoside Hydrolases
Glycoside hydrolases (also called glycosidases or glycosyl hydrolases) catalyze the hydrolysis of glycosidic bonds in complex sugars. They are extremely common enzymes with roles in nature including degradation of biomass such as cellulose (cellulase), hemicellulose, and starch (amylase), in anti-bacterial defense strategies (e.g., lysozyme), in pathogenesis mechanisms (e.g., viral neuraminidases) and in normal cellular function (e.g., trimming mannosidases involved in N-linked glycoprotein biosynthesis). Together with glycosyltransferases, glycosidases form the major catalytic machinery for the synthesis and breakage of glycosidic bonds. Occurrence and importance Glycoside hydrolases are found in essentially all domains of life. In prokaryotes, they are found both as intracellular and extracellular enzymes that are largely involved in nutrient acquisition. One of the important occurrences of glycoside hydrolases in bacteria is the enzyme beta-galactosidase (LacZ), which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolyse
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and water molecule to split into two parts. In s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycosidic Bond
A glycosidic bond or glycosidic linkage is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate. A glycosidic bond is formed between the hemiacetal or hemiketal group of a saccharide (or a molecule derived from a saccharide) and the hydroxyl group of some compound such as an alcohol. A substance containing a glycosidic bond is a glycoside. The term 'glycoside' is now extended to also cover compounds with bonds formed between hemiacetal (or hemiketal) groups of sugars and several chemical groups other than hydroxyls, such as -SR (thioglycosides), -SeR (selenoglycosides), -NR1R2 (N-glycosides), or even -CR1R2R3 (C-glycosides). Particularly in naturally occurring glycosides, the compound ROH from which the carbohydrate residue has been removed is often termed the aglycone, and the carbohydrate residue itself is sometimes referred to as the 'glycone'. S-, N-, C-, and O-glycosidic bonds Glycosidi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CAZy
CAZy is a database of Carbohydrate-Active enZYmes (CAZymes). The database contains a classification and associated information about enzymes involved in the synthesis, metabolism, and recognition of complex carbohydrates, i.e. disaccharides, oligosaccharides, polysaccharides, and glycoconjugates. Included in the database are families of glycoside hydrolases, glycosyltransferases, polysaccharide lyases, carbohydrate esterases, and non-catalytic carbohydrate-binding modules. The CAZy database also includes a classification of Auxiliary Activity redox enzymes involved in the breakdown of lignocellulose. CAZy was established in 1999 in order to provide online and constantly updated access to the protein sequence-based family classification of CAZymes, which was originally developed in early 1990s to classify the glycoside hydrolases. New entries are added shortly after they appear in the daily releases of GenBank. The rapid evolution of high-throughput DNA sequencing has resulted i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aspartic Acid
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the protonated –NH form under physiological conditions, while its α-carboxylic acid group is deprotonated −COO− under physiological conditions. Aspartic acid has an acidic side chain (CH2COOH) which reacts with other amino acids, enzymes and proteins in the body. Under physiological conditions (pH 7.4) in proteins the side chain usually occurs as the negatively charged aspartate form, −COO−. It is a non-essential amino acid in humans, meaning the body can synthesize it as needed. It is encoded by the codons GAU and GAC. D-Aspartate is one of two D-amino acids commonly found in mammals. .html" ;"title="/sup>">/sup> In proteins aspartate sidechains are often hydrogen bonded to form asx turns or asx motifs, which frequently occur at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sucrase
Sucrase is a digestive enzyme that catalyzes the hydrolysis of sucrose to its subunits fructose and glucose. One form, sucrase-isomaltase, is secreted in the small intestine on the brush border. The sucrase enzyme invertase, which occurs more commonly in plants, also hydrolyzes sucrose but by a different mechanism. Types * is isomaltase * is invertase * is sucrose alpha-glucosidase Physiology Sucrose intolerance (also known as congenital sucrase-isomaltase deficiency (CSID), genetic sucrase-isomaltase deficiency (GSID), or sucrase-isomaltase deficiency) occurs when sucrase is not being secreted in the small intestine. With sucrose intolerance, the result of consuming sucrose is excess gas production and often diarrhea and malabsorption. Lactose intolerance is a related disorder that reflects an individual's inability to hydrolyze the disaccharide lactose. Sucrase is secreted by the tips of the villi of the epithelium in the small intestine. Its levels are reduced in response ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomaltase
Isomaltase () is an enzyme that breaks the bonds linking saccharides, which cannot be broken by amylase or maltase. It digests polysaccharides at the alpha 1-6 linkages. Its substrate, alpha-limit dextrin, is a product of amylopectin digestion that retains its 1-6 linkage (its alpha 1-4 linkages having already been broken down by amylase). The product of the enzymatic digestion of alpha-limit dextrin by isomaltase is maltose. Isomaltase helps amylase to digest alpha-limit dextrin to produce maltose. The human sucrase-isomaltase is a dual-function enzyme with two GH31 domains, one serving as the isomaltase, the other as a sucrose alpha-glucosidase. Nomenclature The systematic name of sucrase-isomaltase is oligosaccharide 6-alpha-glucohydrolase. This enzyme is also known as: * Sucrase-alpha-dextrinase * oligo-1,6-glucosidase, * limit dextrin, * so maltase, * exo-oligo-1,6-glucosidase, * dextrin 6alpha-glucanohydrolase, * alpha-limit dextrin, * dextrin 6-glucanohydrolase, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lysosomal
A lysosome () is a membrane-bound organelle found in many animal Cell (biology), cells. They are spherical Vesicle (biology and chemistry), vesicles that contain Hydrolysis, hydrolytic enzymes that can break down many kinds of biomolecules. A lysosome has a specific composition, of both its membrane proteins, and its lumen (anatomy), lumenal proteins. The lumen's pH (~4.5–5.0) is optimal for the enzymes involved in hydrolysis, analogous to the activity of the stomach. Besides degradation of polymers, the lysosome is involved in various cell processes, including secretion, plasma membrane repair, apoptosis, cell signaling, and energy metabolism. Lysosomes act as the waste disposal system of the cell by digesting used materials in the cytoplasm, from both inside and outside the cell. Material from outside the cell is taken up through endocytosis, while material from the inside of the cell is digested through autophagy. The sizes of the organelles vary greatly—the larger ones ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha-glucosidase
α-Glucosidase (EC 3.2.1.20, maltase, glucoinvertase, glucosidosucrase, maltase-glucoamylase, α-glucopyranosidase, glucosidoinvertase, α-D-glucosidase, α-glucoside hydrolase, α-1,4-glucosidase, α-D-glucoside glucohydrolase; systematic name α-D-glucoside glucohydrolase) is a glucosidase located in the brush border of the small intestine that acts upon α(1→4) bonds: : Hydrolysis of terminal, non-reducing (1→4)-linked α-D-glucose residues with release of D-glucose This is in contrast to β-glucosidase. α-Glucosidase breaks down starch and disaccharides to glucose. Other glucosidases include: * Cellulase * Beta-glucosidase * Debranching enzyme Mechanism α-Glucosidase hydrolyzes terminal non-reducing (1→4)-linked α-glucose residues to release a single α-glucose molecule. α-Glucosidase is a carbohydrate-hydrolase that releases α-glucose as opposed to β-glucose. β-Glucose residues can be released by glucoamylase, a functionally similar enzyme. The substr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |