|
Enolate Anion
In organic chemistry, enolates are organic anions derived from the deprotonation of carbonyl () compounds. Rarely isolated, they are widely used as reagents in the synthesis of organic compounds. Bonding and structure Enolate anions are electronically related to allyl anions. The anionic charge is delocalized over the oxygen and the two carbon sites. Thus they have the character of both an alkoxide and a carbanion. Although they are often drawn as being simple salts, in fact they adopt complicated structures often featuring aggregates. Preparation Deprotonation of enolizable ketones, aromatic alcohols, aldehydes, and esters gives enolates. With strong bases, the deprotonation is quantitative. Typically enolates are generated from using lithium diisopropylamide (LDA). Often, as in conventional Claisen condensations, Mannich reactions, and aldol condensations, enolates are generated in low concentrations with alkoxide bases. Under such conditions, they exist in low conc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enolate Resonance
In organic chemistry, enolates are organic anions derived from the deprotonation of carbonyl () compounds. Rarely isolated, they are widely used as reagents in the synthesis of organic compounds. Bonding and structure Enolate anions are electronically related to allyl anions. The anionic charge is delocalized over the oxygen and the two carbon sites. Thus they have the character of both an alkoxide and a carbanion. Although they are often drawn as being simple salts, in fact they adopt complicated structures often featuring aggregates. Preparation Deprotonation of enolizable ketones, aromatic alcohols, aldehydes, and esters gives enolates. With strong bases, the deprotonation is quantitative. Typically enolates are generated from using lithium diisopropylamide (LDA). Often, as in conventional Claisen condensations, Mannich reactions, and aldol condensations, enolates are generated in low concentrations with alkoxide bases. Under such conditions, they exist in low concent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
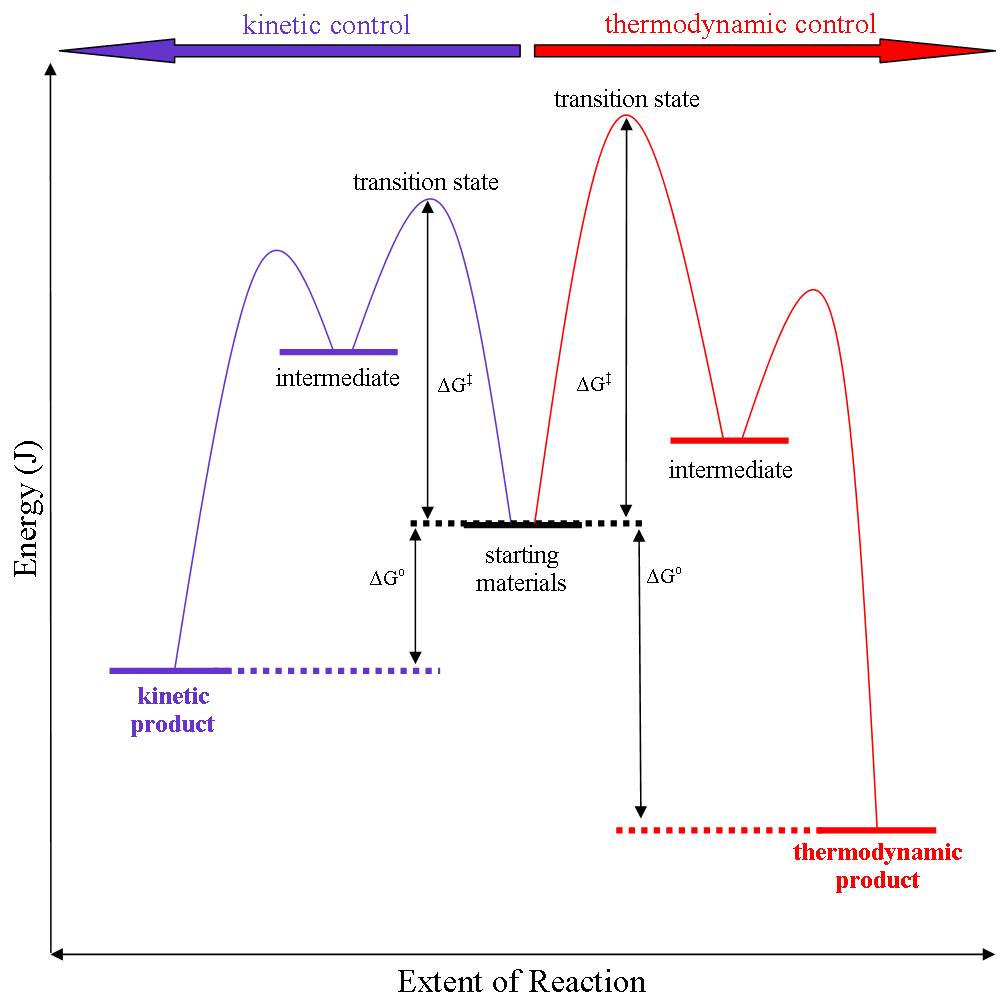
Kinetic Versus Thermodynamic Reaction Control
Thermodynamic reaction control or kinetic reaction control in a chemical reaction can decide the composition in a reaction product mixture when competing pathways lead to different products and the reaction conditions influence the selectivity or stereoselectivity. The distinction is relevant when product A forms faster than product B because the activation energy for product A is lower than that for product B, yet product B is more stable. In such a case A is the kinetic product and is favoured under kinetic control and B is the thermodynamic product and is favoured under thermodynamic control.Introduction to Organic Chemistry I, Seth Robert Elsheimer, Blackwell Publishing, 2000 The conditions of the reaction, such as temperature, pressure, or solvent, affect which reaction pathway may be favored: either the kinetically controlled or the thermodynamically controlled one. Note this is only true if the activation energy of the two pathways differ, with one pathway having a lower '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry. Discovered independently by the Russian chemist Alexander Borodin in 1869 and by the French chemist Charles-Adolphe Wurtz in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as ''aldols'', from the ''ald''ehyde + alcoh''ol'', a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic. For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritol and the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt). The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the Alpha carbon, α- and β-carbon of the aldol adduct, mar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Self-condensation
Self-condensation is an organic reaction in which a chemical compound containing a carbonyl group acts both as the electrophile and the nucleophile in an aldol condensation. It is also called a symmetrical aldol condensation as opposed to a mixed aldol condensation in which the electrophile and nucleophile are different species. For example, two molecules of acetone condense to a single compound mesityl oxide in the presence of an ion-exchange resin: :2 CH3COCH3 → (CH3)2C=CH(CO)CH3 + H2O For synthetic uses, this is generally an undesirable, but spontaneous and favored side-reaction of mixed aldol condensation, and special precautions are needed to prevent it. Preventing self-condensation In many cases, self-condensation is an unwanted side-reaction. Therefore, chemists have adopted many ways to prevent this from occurring when performing a crossed aldol reaction. The use of a more reactive electrophile, and a non-enolizable partner If acetophenone and benzaldehyde are put toge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aza Enolate Formation2
Aza or AZA may refer to: Places * Aza, Azerbaijan, a village and municipality * Azadkənd, Nakhchivan or Lower Aza, Azerbaijan *Aza, medieval name of Haza, Province of Burgos, Spain *Aźa, a Tibetan name for the Tuyuhun kingdom *Aza, a Hebrew romanization for Gaza City or Gaza Strip People * Aza (given name) *Aza of Mannea, king, reigned c. 710–700 BC *Alejandro De Aza (born 1984), Dominican baseball outfielder * Vital Aza (1851–1912), Spanish author, playwright, poet and satirist Other uses *Aza (Kanji: 字), a village or town section in the Japanese addressing system * Azelanic acid, an organic acid used for medical treatment of acne and as whitening agent Abbreviations or initialisms * Azathioprine, an immunosuppressant medication * Association of Zoos and Aquariums (formerly named "American Zoo and Aquarium Association") * Aleph Zadik Aleph, a Jewish youth group that is part of BBYO * ISO 639-3 aza for Azha language, a language of China * ICAO code for Alitalia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Diisopropylamide
Lithium diisopropylamide (commonly abbreviated LDA) is a chemical compound with the molecular formula . It is used as a strong base and has been widely utilized due to its good solubility in non-polar organic solvents and non-nucleophilic nature. It is a colorless solid, but is usually generated and observed only in solution. It was first prepared by Hamell and Levine in 1950 along with several other hindered lithium diorganylamides to effect the deprotonation of esters at the α position without attack of the carbonyl group. Preparation and structure LDA is commonly formed by treating a cooled (0 to −78 °C) mixture of tetrahydrofuran and diisopropylamine with ''n''-butyllithium. When dissociated, the diisopropylamide anion can become protonated to form diisopropylamine. Diisopropylamine has a p''K''a value of 36. Therefore, its conjugate base is suitable for the deprotonation of compounds with greater acidity, importantly, such weakly acidic compounds (carbon acids) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Li Enolate 2
Li, li, or LI may refer to: Businesses and organizations * Landscape Institute, a British professional body for landscape architects * Leadership Institute, a non-profit organization located in Arlington, Virginia, US, that teaches "political technology." * Li Auto (Nasdaq: LI), a Chinese manufacturer of electric vehicles * Liberal International, a political federation for liberal parties * Linux International, an international non-profit organization * Lyndon Institute, an independent high school in the U.S. state of Vermont * The Light Infantry, a British Army infantry regiment Names * Li (surname), including: ** List of people with surname Li ** Li (surname 李), one of the most common surnames in the world ** Li (surname 黎), the 84th most common surname in China ** Li (surname 栗), the 249th most common surname in China ** Li (surname 利), the 299th most common surname in China ** Li (surname 厉), a Chinese surname ** Li (surname 郦), a Chinese surname ** Li (surname ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Michael Acceptor
In organic chemistry, the Michael reaction or Michael addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon. It belongs to the larger class of conjugate additions and is widely used for the mild formation of carbon-carbon bonds. The Michael addition is an important atom-economical method for diastereoselective and enantioselective C–C bond formation, and many asymmetric variants exist : In this general Michael addition scheme, either or both of R and R' on the nucleophile (the Michael donor) represent electron-withdrawing substituents such as acyl, cyano, nitro, or sulfone groups, which make the adjacent methylene hydrogen acidic enough to form a carbanion when reacted with the base, ''B:''. For the alkene (the Michael acceptor), the R" substituent is usually a carbonyl, which makes the compound ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Counterion
160px, Polystyrene sulfonate, a cation-exchange resin, is typically supplied with as the counterion.">cation-exchange_resin.html" ;"title="Polystyrene sulfonate, a cation-exchange resin">Polystyrene sulfonate, a cation-exchange resin, is typically supplied with as the counterion. In chemistry, a counterion (sometimes written as "counter ion", pronounced as such) is the ion that accompanies an Ionic compound, ionic species in order to maintain Electric charge, electric neutrality. In table salt (NaCl, also known as sodium chloride) the sodium ion (positively charged) is the counterion for the chloride ion (negatively charged) and vice versa. A counterion will be more commonly referred to as an anion or a cation, depending on whether it is negatively or positively charged. Thus, the counterion to an anion will be a cation, and vice versa. In biochemistry, counterions are generally vaguely defined. Depending on their charge, proteins are associated with a variety of smaller ani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mukaiyama Aldol-Übersichtsreaktion1
was a Japanese organic chemist. One of the most prolific chemists of the 20th century in the field of organic synthesis, Mukaiyama helped establish the field of organic chemistry in Japan after World War II. Education Mukaiyama studied chemistry at the Tokyo Institute of Technology, earning his BSc in synthetic organic chemistry in 1948. He became assistant professor at Gakushuin University in 1953, where he stayed until he received his Ph.D. in synthetic organic chemistry from the University of Tokyo in 1957. Research and career Early work Mukaiyama became an assistant professor at the Tokyo Institute of Technology in 1958 and earned his full professorship in 1963. During this time, his main focus was on organophosphorus chemistry. While examining deoxygenation reactions involving phosphines, Mukaiyama found that the mercury(II) acetate employed as a catalyst would react with phosphorus(III) compounds to produce acetic anhydride. This initial example expanded into the concept o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mukaiyama Aldol Reaction
The Mukaiyama aldol addition is an organic reaction and a type of aldol reaction between a silyl enol ether and an aldehyde or formate. The reaction was discovered by Teruaki Mukaiyama (1927–2018) in 1973. His choice of reactants allows for a crossed aldol reaction between an aldehyde and a ketone or a different aldehyde without self-condensation of the aldehyde. For this reason the reaction is used extensively in organic synthesis. General reaction scheme The Mukaiyama aldol addition is a Lewis acid mediated addition of enol silanes to carbonyl compounds. In this reaction compounds with various organic groups can be used (see educts). A basic version ( = H) without the presence of chiral catalysts is shown below. A racemic mix of enantiomers is built. If Z- or E-enol silanes are used in this reaction a mixture of four products occurs, yielding two racemates. Whether the ''anti''-diastereomer or the ''syn''-diastereomer is built depends largely on reaction conditions, subs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silyl Enol Ether
Silyl enol ethers in organic chemistry are a class of organic compounds that share a common functional group composed of an enolate bonded through its oxygen end to an organosilicon group. They are important intermediates in organic synthesis. Synthesis Silyl enol ethers are generally prepared by reacting an enolizable Carbonyl group, carbonyl compound with a silyl electrophile and a Base (chemistry), base, or just reacting an enolate with a silyl electrophile.Clayden, J., Greeves, N., & Warren, S. (2012). Silyl enol ethers. In ''Organic chemistry'' (Second ed., pp. 466-467). Oxford University Press. Since silyl electrophiles are HSAB theory, hard and silicon-oxygen bonds are very strong, the oxygen (of the carbonyl compound or enolate) acts as the nucleophile to form a Si-O single bond. The most commonly used silyl electrophile is trimethylsilyl chloride. To increase the rate of reaction, trimethylsilyl triflate may also be used in the place of trimethylsilyl chloride as a more el ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
=CMe2(Litmeda)_dimer_(VITLUG).png)