|
Exohedral Fullerenes
Exohedral fullerenes, also called exofullerenes, are fullerenes that have additional atoms, ions, or clusters attached their outer spheres, such as C50Cl10 and C60H8. or fullerene ligands. See also * Fullerene ligands *Endohedral fullerene Endohedral fullerenes, also called endofullerenes, are fullerenes that have additional atoms, ions, or clusters enclosed within their inner spheres. The first lanthanum C60 complex called La@C60 was synthesized in 1985. The @ (at sign) in the ... References {{reflist, 2 Fullerenes Supramolecular chemistry ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fullerene
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ellipsoid, tube, or many other shapes and sizes. Graphene (isolated atomic layers of graphite), which is a flat mesh of regular hexagonal rings, can be seen as an extreme member of the family. Fullerenes with a closed mesh topology are informally denoted by their empirical formula C''n'', often written C''n'', where ''n'' is the number of carbon atoms. However, for some values of ''n'' there may be more than one isomer. The family is named after buckminsterfullerene (C60), the most famous member, which in turn is named after Buckminster Fuller. The closed fullerenes, especially C60, are also informally called buckyballs for their resemblance to the standard ball of association football ("soccer"). Nested closed fullerenes have been named ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are extremely small, typically around 100 picometers across. They are so small that accurately predicting their behavior using classical physics, as if they were tennis balls for example, is not possible due to quantum effects. More than 99.94% of an atom's mass is in the nucleus. The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. If the number of protons and electrons are equal, then the atom is electrically neutral. If an atom has more or fewer electrons than protons, then it has an overall negative or positive charge, respectively – such atoms are called ions. The electrons of an atom are a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cluster (physics)
In physics, the term clusters denotes small, polyatomic particles. As a rule of thumb, any particle made of between 3×100 and 3×107 atoms is considered a cluster. The term can also refer to the organization of protons and neutrons within an atomic nucleus, e.g. the alpha particle (also known as "α-cluster"), consisting of two protons and two neutrons (as in a helium nucleus). Overview Although first reports of cluster species date back to the 1940s, cluster science emerged as a separate direction of research in the 1980s, One purpose of the research was to study the gradual development of collective phenomena which characterize a bulk solid. For example, these are the color of a body, its electrical conductivity, its ability to absorb or reflect light, and magnetic phenomena such as ferro-, ferri-, or antiferromagnetism. These are typical collective phenomena which only develop in an aggregate of a large number of atoms. It was found that collective phenomena break down for v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
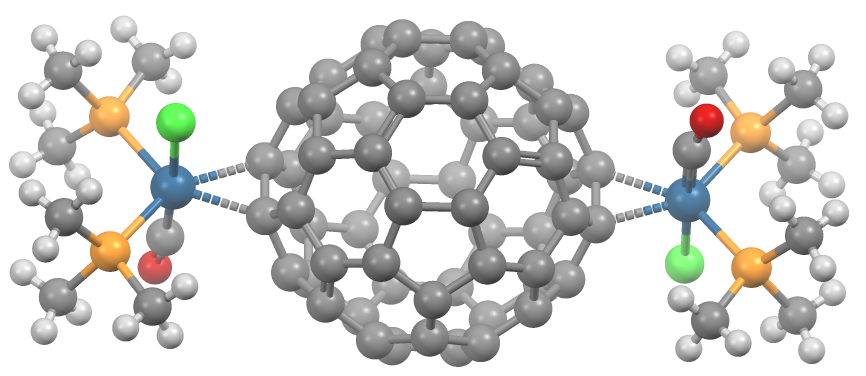
Fullerene Ligand
A transition metal fullerene complex is a coordination complex wherein fullerene serves as a ligand. Fullerenes are typically spheroidal carbon compounds, the most prevalent being buckminsterfullerene, C60. One year after it was prepared in milligram quantities in 1990, C60 was shown to function as a ligand in the complex h3Psub>2Pt(η2-C60). Since this report, a variety of transition metals and binding modes were demonstrated. Most transition metal fullerene complex are derived from C60, although other fullerenes also coordinate to metals as seen with C70Rh(H)(CO)(PPh3)2. Binding modes As ligands, fullerenes behave similarly to electron-deficient alkenes such as tetracyanoethylene. Thus, their complexes are a subset of metal-alkene complexes. They almost always coordinate in a dihapto fashion and prefer electron-rich metal centers.Spessard, p. 162 This binding occurs on the junction of two 6-membered rings. Hexahapto and pentahapto bonding is rarely observed.Spessard, p. 165 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fullerene Ligand
A transition metal fullerene complex is a coordination complex wherein fullerene serves as a ligand. Fullerenes are typically spheroidal carbon compounds, the most prevalent being buckminsterfullerene, C60. One year after it was prepared in milligram quantities in 1990, C60 was shown to function as a ligand in the complex h3Psub>2Pt(η2-C60). Since this report, a variety of transition metals and binding modes were demonstrated. Most transition metal fullerene complex are derived from C60, although other fullerenes also coordinate to metals as seen with C70Rh(H)(CO)(PPh3)2. Binding modes As ligands, fullerenes behave similarly to electron-deficient alkenes such as tetracyanoethylene. Thus, their complexes are a subset of metal-alkene complexes. They almost always coordinate in a dihapto fashion and prefer electron-rich metal centers.Spessard, p. 162 This binding occurs on the junction of two 6-membered rings. Hexahapto and pentahapto bonding is rarely observed.Spessard, p. 165 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Endohedral Fullerene
Endohedral fullerenes, also called endofullerenes, are fullerenes that have additional atoms, ions, or clusters enclosed within their inner spheres. The first lanthanum C60 complex called La@C60 was synthesized in 1985. The @ (at sign) in the name reflects the notion of a small molecule trapped inside a shell. Two types of endohedral complexes exist: endohedral metallofullerenes and non-metal doped fullerenes. Notation In a traditional chemical formula notation, a buckminsterfullerene (C60) with an atom (M) was simply represented as MC60 regardless of whether M was inside or outside the fullerene. In order to allow for more detailed discussions with minimal loss of information, a more explicit notation was proposed in 1991, where the atoms listed to the left of the @ sign are situated inside the network composed of the atoms listed to the right. The example above would then be denoted M@C60 if M were inside the carbon network. A more complex example is K2(K@C59B), which denotes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fullerenes
A fullerene is an allotrope of carbon whose molecule consists of carbon atoms connected by single and double bonds so as to form a closed or partially closed mesh, with fused rings of five to seven atoms. The molecule may be a hollow sphere, ellipsoid, tube, or many other shapes and sizes. Graphene (isolated atomic layers of graphite), which is a flat mesh of regular hexagonal rings, can be seen as an extreme member of the family. Fullerenes with a closed mesh topology are informally denoted by their empirical formula C''n'', often written C''n'', where ''n'' is the number of carbon atoms. However, for some values of ''n'' there may be more than one isomer. The family is named after buckminsterfullerene (C60), the most famous member, which in turn is named after Buckminster Fuller. The closed fullerenes, especially C60, are also informally called buckyballs for their resemblance to the standard ball of association football ("soccer"). Nested closed fullerenes have been named ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |