|
Dithiolene
Dithiolene metal complexes are complexes containing 1,2-dithiolene ligands. 1,2-Dithiolene ligands, a particular case of 1,2-dichalcogenolene species, are unsaturated bidentate ligand wherein the two donor atoms are sulfur. 1,2-Dithiolene metal complexes are often referred to as "metal dithiolenes", "metallodithiolenes" or "dithiolene complexes". Most molybdenum- and tungsten-containing proteins have dithiolene-like moieties at their active sites, which feature the so-called molybdopterin cofactor bound to the Mo or W. Dithiolene metal complexes have been studied since the 1960s when they were first popularized by Gerhard N. Schrauzer and Volker P. Mayweg, who prepared nickel bis(stilbene-1,2-dithiolate) (Ni(S2C2Ph2)2) by the reaction of nickel sulfide and diphenylacetylene. The structural, spectroscopic, and electrochemical properties of many related complexes have been described. Structure and bonding Dithiolene metal complexes can be found in coordination compounds where t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Bis(stilbenedithiolate)
Nickel bis(stilbenedithiolate) or bis(dithiobenzil)nickel is a coordination complex with the formula Ni(S2C2Ph2)2 (where Ph = phenyl). It exists as a black solid that gives green solutions in toluene due to a strong absorption at 855 nm. The complex is a prototype of a large family of bis(dithiolene) complexes or the formula Ni(S2C2R2)2 (R = H, alkyl, aryl). These complexes have attracted much attention as dyes. They are of academic interest because the dithiolenes are noninnocent ligands. The lengths of the C-S and C-C bonds in the backbone, respectively 1.71 and 1.39 Å, are intermediate between double and single bonds. : The complex was prepared originally by treating nickel sulfide with diphenylacetylene. High yielding syntheses involve treating nickel salts with sulfiding, sulfided benzoin (organic compound), benzoin. The complex reacts with ligands to form monodithiolene complexes of the type Ni(S2C2Ph2)L2. References {{Reflist Coordination complexes Chelating age ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Non-innocent Ligand
In chemistry, a (redox) non-innocent ligand is a ligand in a metal complex where the oxidation state is not clear. Typically, complexes containing non-innocent ligands are redox active at mild potentials. The concept assumes that redox reactions in metal complexes are either metal or ligand localized, which is a simplification, albeit a useful one. C.K. Jørgensen first described ligands as "innocent" and "suspect": "Ligands are innocent when they allow oxidation states of the central atoms to be defined. The simplest case of a suspect ligand is NO..." Redox reactions of complexes of innocent vs. non-innocent ligands Conventionally, redox reactions of coordination complexes are assumed to be metal-centered. The reduction of MnO4− to MnO42− is described by the change in oxidation state of manganese from 7+ to 6+. The oxide ligands do not change in oxidation state, remaining 2-. Oxide is an innocent ligand. Another example of conventional metal-centered redox couple is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdopterin
Molybdopterins are a class of cofactors found in most molybdenum-containing and all tungsten-containing enzymes. Synonyms for molybdopterin are: MPT and pyranopterin-dithiolate. The nomenclature for this biomolecule can be confusing: Molybdopterin itself contains no molybdenum; rather, this is the name of the ligand (a ''pterin'') that will bind the active metal. After molybdopterin is eventually complexed with molybdenum, the complete ligand is usually called molybdenum cofactor. Molybdopterin consists of a pyranopterin, a complex heterocycle featuring a pyran fused to a pterin ring. In addition, the pyran ring features two thiolates, which serve as ligands in molybdo- and tungstoenzymes. In some cases, the alkyl phosphate group is replaced by an alkyl diphosphate nucleotide. Enzymes that contain the molybdopterin cofactor include xanthine oxidase, DMSO reductase, sulfite oxidase, and nitrate reductase. The only molybdenum-containing enzymes that do not feature molybdopteri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Maleonitriledithiolate
Sodium maleonitriledithiolate is the chemical compound described by the formula Na2S2C2(CN)2. The name refers to the cis compound, structurally related to maleonitrile ((CHCN)2). Maleonitriledithiolate is often abbreviated mnt. It is a "dithiolene", i.e. a chelating alkene-1,2-dithiolate. It is a prototypical non-innocent ligand in coordination chemistry. Several complexes are known, such as i(mnt)2sup>2−. The salt is synthesized by treating carbon disulfide with sodium cyanide to give the cyanodithioformate salt, which eliminates elemental sulfur in aqueous solution: :2 NaCN + 2 CS2 → Na2S2C2(CN)2 + 1/4 S8 The compound was first described by Bähr and Schleitzer 1958. {{cite journal , author = G. Bähr and G. Schleitzer , title = Beiträge zur Chemie des Schwefelkohlenstoffs und Selenkohlenstoffs, II. Die Kondensierende Spontan-Entschwefelung von Salzen und Estern der Cyan-Dithioameisensäure. Freie Cyan-Dithioameisensäure , journal = Chemische Berichte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium 1,3-dithiole-2-thione-4,5-dithiolate
Sodium 1,3-dithiole-2-thione-4,5-dithiolate is the organosulfur compound with the formula Na2C3S5, abbreviated Na2dmit. It is the sodium salt of the conjugate base of the 1,3-dithiole-2-thione-4,5-dithiol. The salt is a precursor to dithiolene complexes and tetrathiafulvalenes. Reduction of carbon disulfide with sodium affords sodium 1,3-dithiole-2-thione-4,5-dithiolate together with sodium trithiocarbonate: : 4 Na + 4 CS2 → Na2C3S5 + Na2CS3 Before the characterization of dmit2-, reduction of CS2 was thought to give tetrathiooxalate (Na2C2S4). The dianion C3S52- is purified as the tetraethylammonium salt of the zincate complex n(C3S5)2sup>2-. This salt converts to the bis(thioester) upon treatment with benzoyl chloride: : (C2H5)4sub>2 n(C3S5)2 + 4 C6H5COCl → 2 C3S3(SC(O)C6H5)2 + (C2H5)4sub>2 nCl4Cleavage of the thioester with sodium methoxide Sodium methoxide is the simplest sodium alkoxide. With the formula , it is a white solid, which is formed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation States
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. Conceptually, the oxidation state may be positive, negative or zero. While fully ionic bonds are not found in nature, many bonds exhibit strong ionicity, making oxidation state a useful predictor of charge. The oxidation state of an atom does not represent the "real" formal charge on that atom, or any other actual atomic property. This is particularly true of high oxidation states, where the ionization energy required to produce a multiply positive ion is far greater than the energies available in chemical reactions. Additionally, the oxidation states of atoms in a given compound may vary depending on the choice of electronegativity scale used in their calculation. Thus, the oxidation state of an atom in a compound is purely a formalism. It ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation State
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. Conceptually, the oxidation state may be positive, negative or zero. While fully ionic bonds are not found in nature, many bonds exhibit strong ionicity, making oxidation state a useful predictor of charge. The oxidation state of an atom does not represent the "real" formal charge on that atom, or any other actual atomic property. This is particularly true of high oxidation states, where the ionization energy required to produce a multiply positive ion is far greater than the energies available in chemical reactions. Additionally, the oxidation states of atoms in a given compound may vary depending on the choice of electronegativity scale used in their calculation. Thus, the oxidation state of an atom in a compound is purely a formalism. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dithiete
Dithiete is an unsaturated heterocyclic compound that contains two adjacent sulfur atoms and two sp2-hybridized carbon centers. Derivatives are known collectively as dithietes or 1,2-dithietes. With 6 π electrons, 1,2-dithietes are examples of aromatic organosulfur compounds. A few 1,2-dithietes have been isolated. Unsubstituted 1,2-dithiete has been generated in thermolytic reactions and was characterized by microwave spectroscopy, ultraviolet photoelectron spectroscopy and infrared spectroscopy in a low temperature matrix. The open ring isomer, dithioglyoxal, HC(S)C(S)H, is less stable than the 1,2-dithiete. The dithione can be prepared (as ''trans''-dithioglyoxal) by low temperature photolysis of 1,3-dithiol-2-one. Quantum chemical calculations reproduce the observed greater stability of 1,2-dithiete only if large basis-sets with polarization functions are used. : See also * Dithietane - the corresponding saturated ring * Thiete Thiete is a heterocyclic compound contai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
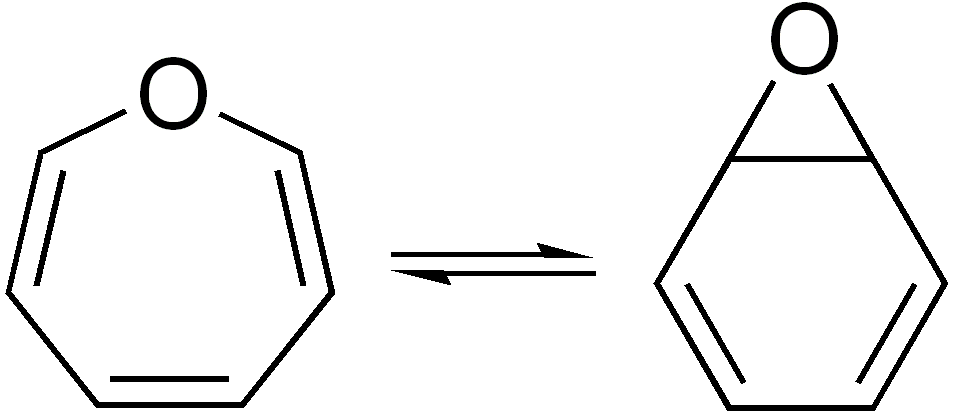
Valence Isomer
In organic chemistry, two molecules are valence isomers when they are constitutional isomers that can interconvert through pericyclic reactions. Benzene There are many valence isomers one can draw for the C6H6 formula benzene. Some were originally proposed for benzene itself before the actual structure of benzene was known. Others were later synthesized in lab. Some have been observed to isomerize to benzene, whereas others tend to undergo other reactions instead, or isomerize by ways other than pericyclic reactions. Image:Benzene-2D-flat.png, Benzene Image:Historic Benzene Formulae Dewar(1867) V.1.svg, Dewar benzene Image:Prisman2.svg, Prismane Image:Benzvalene.png, Benzvalene Image:Bicycloprop-2-enyl.svg, Bicyclopropenyl Cyclooctatetraene The valence isomers are not restricted to isomers of benzene. Valence isomers are also seen in the series (CH)8. Due to the larger number of units, the number of possible valence isomers is also greater and at least 21: Image:Cyclooctatet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Pentasulfide
Phosphorus pentasulfide is the inorganic compound with the formula (monomer) or (dimer). This yellow solid is the one of two phosphorus sulfides of commercial value. Samples often appear greenish-gray due to impurities. It is soluble in carbon disulfide but reacts with many other solvents such as alcohols, DMSO, and DMF. Structure and synthesis Its tetrahedral molecular structure is similar to that of adamantane and almost identical to the structure of phosphorus pentoxide. Phosphorus pentasulfide is obtained by the reaction of liquid white phosphorus () with sulfur above 300 °C. The first synthesis of by Berzelius in 1843 was by this method. Alternatively, can be formed by reacting elemental sulfur or pyrite, , with ferrophosphorus, a crude form of (a byproduct of white phosphorus () production from phosphate rock): : : Applications Approximately 150,000 tons of are produced annually. The compound is mainly converted to other derivatives for use as lubrication ad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acyloin
Acyloins or α-hydroxy ketones are a class of organic compounds which all possess a hydroxy group adjacent to a ketone group. The name acyloin is derived from the fact that they are formally derived from reductive coupling of carboxylic acyl groups. Synthesis Classic organic reactions exist for the synthesis of acyloins. * The acyloin condensation is a reductive coupling of esters * The benzoin condensation is condensation reaction between aldehydes catalyzed by a nucleophile * Oxidation of carbonyls is possible with molecular oxygen but not selective * Better alternative is oxidation of corresponding silyl enol ethers with ''m''CPBA in the Rubottom oxidation * MoOPH oxidation of carbonyls is a system with molybdenum peroxide, pyridine and hexamethylphosphoramide. Enolate oxidation by sulfonyloxaziridines Enolates can be oxidized by sulfonyloxaziridines. The enolate reacts by nucleophilic displacement at the electron deficient oxygen of the oxaziridine ring. : This rea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2Ni(mnt)2.jpg)