|
Dimethyltubocurarine
Dimethyltubocurarinium chloride (INN; also known as metocurine chloride (USAN) and dimethyltubocurarine chloride) is a non-depolarizing nicotinic acetylcholine receptor antagonist used as a muscle relaxant A muscle relaxant is a drug that affects skeletal muscle function and decreases the muscle tone. It may be used to alleviate symptoms such as muscle spasms, pain, and hyperreflexia. The term "muscle relaxant" is used to refer to two major therapeu .... References Quaternary ammonium compounds Nicotinic antagonists Norsalsolinol ethers Pyrogallol ethers Macrocycles Cyclophanes {{musculoskeletal-drug-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
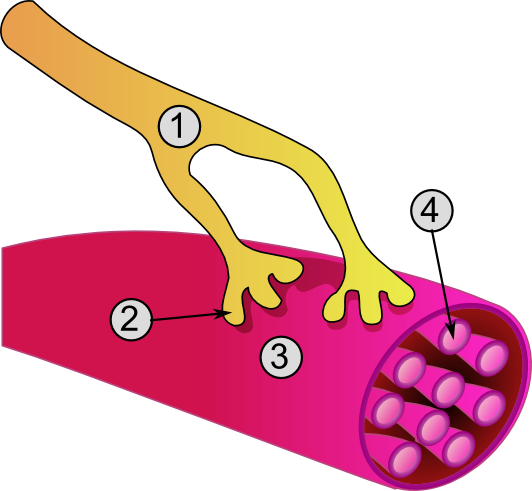
Neuromuscular-blocking Drugs
Neuromuscular-blocking drugs block neuromuscular transmission at the neuromuscular junction, causing paralysis of the affected skeletal muscles. This is accomplished via their action on the post-synaptic acetylcholine (Nm) receptors. In clinical use, neuromuscular block is used adjunctively to anesthesia to produce paralysis, firstly to paralyze the vocal cords, and permit intubation of the trachea, and secondly to optimize the surgical field by inhibiting spontaneous ventilation, and causing relaxation of skeletal muscles. Because the appropriate dose of neuromuscular-blocking drug may paralyze muscles required for breathing (i.e., the diaphragm), mechanical ventilation should be available to maintain adequate respiration. Patients are still aware of pain even after full conduction block has occurred; hence, general anesthetics and/or analgesics must also be given to prevent anesthesia awareness. Nomenclature Neuromuscular blocking drugs are often classified into two broad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
United States Adopted Name
A United States Adopted Name (USAN) is a unique nonproprietary name assigned to a medication marketed in the United States. Each name is assigned by the USAN Council, which is co-sponsored by the American Medical Association (AMA), the United States Pharmacopeial Convention (USP), and the American Pharmacists Association (APhA). The USAN Program states that its goal is to select simple, informative, and unique nonproprietary names (also called generic names) for drugs by establishing logical nomenclature classifications based on pharmacological or chemical relationships. In addition to drugs, the USAN Council names agents for gene therapy and cell therapy, List of soft contact lens materials, contact lens polymers, surgical materials, diagnostics, carriers, and substances used as an excipient. The USAN Council works in conjunction with the World Health Organization (WHO) international nonproprietary name (INN) Expert Committee and national nomenclature groups to standardize drug ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinic Acetylcholine Receptor
Nicotinic acetylcholine receptors, or nAChRs, are receptor polypeptides that respond to the neurotransmitter acetylcholine. Nicotinic receptors also respond to drugs such as the agonist nicotine. They are found in the central and peripheral nervous system, muscle, and many other tissues of many organisms. At the neuromuscular junction they are the primary receptor in muscle for motor nerve-muscle communication that controls muscle contraction. In the peripheral nervous system: (1) they transmit outgoing signals from the presynaptic to the postsynaptic cells within the sympathetic and parasympathetic nervous system, and (2) they are the receptors found on skeletal muscle that receive acetylcholine released to signal for muscular contraction. In the immune system, nAChRs regulate inflammatory processes and signal through distinct intracellular pathways. In insects, the cholinergic system is limited to the central nervous system. The nicotinic receptors are considered cholinergi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinic Antagonist
A nicotinic antagonist is a type of anticholinergic drug that inhibits the action of acetylcholine (ACh) at nicotinic acetylcholine receptors. These compounds are mainly used for peripheral muscle paralysis in surgery, the classical agent of this type being tubocurarine,P. Taylor (1990). In ''Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th Ed.'', (A. G. Gilman et al., Eds.), pp. 166-186, New York: Pergamon Press. but some centrally acting compounds such as bupropion, mecamylamine, and 18-methoxycoronaridine block nicotinic acetylcholine receptors in the brain and have been proposed for treating nicotine addiction. *Note: Succinylcholine is a nicotinic agonist. See neuromuscular blocking agents page for details on the mechanism of action. See also * Nicotinic acetylcholine receptor * Nicotinic agonist * Muscarinic acetylcholine receptor * Muscarinic agonist * Muscarinic antagonist A muscarinic receptor antagonist (MRA) is a type of anticholinergic agen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quaternary Ammonium Compounds
In chemistry, quaternary ammonium cations, also known as quats, are positively charged polyatomic ions of the structure , R being an alkyl group or an aryl group. Unlike the ammonium ion () and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently charged, independent of the pH of their solution. Quaternary ammonium salts or quaternary ammonium compounds (called quaternary amines in oilfield parlance) are salts of quaternary ammonium cations. Polyquats are a variety of engineered polymer forms which provide multiple quat molecules within a larger molecule. Quats are used in consumer applications including as antimicrobials (such as detergents and disinfectants), fabric softeners, and hair conditioners. As an antimicrobial, they are able to inactivate enveloped viruses (such as SARS-CoV-2). Quats tend to be gentler on surfaces than bleach-based disinfectants, and are generally fabric-safe. Synthesis Quaternary ammonium compou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinic Antagonists
A nicotinic antagonist is a type of anticholinergic drug that inhibits the action of acetylcholine (ACh) at nicotinic acetylcholine receptors. These compounds are mainly used for peripheral muscle paralysis in surgery, the classical agent of this type being tubocurarine,P. Taylor (1990). In ''Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th Ed.'', (A. G. Gilman et al., Eds.), pp. 166-186, New York: Pergamon Press. but some centrally acting compounds such as bupropion, mecamylamine, and 18-methoxycoronaridine block nicotinic acetylcholine receptors in the brain and have been proposed for treating nicotine addiction. *Note: Succinylcholine is a nicotinic agonist. See neuromuscular blocking agents page for details on the mechanism of action. See also * Nicotinic acetylcholine receptor * Nicotinic agonist * Muscarinic acetylcholine receptor * Muscarinic agonist * Muscarinic antagonist A muscarinic receptor antagonist (MRA) is a type of anticholinergic agent t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Norsalsolinol Ethers
Norsalsolinol is a chemical compound that is produced naturally in the body through metabolism of dopamine. It has been shown to be a selective dopaminergic neurotoxin, and has been suggested as a possible cause of neurodegenerative conditions such as Parkinson's disease and the brain damage associated with alcoholism, although evidence for a causal relationship is unclear. (R)-Salsolinol which has been shown to be a product of ethanol metabolism, stereospecifically induces behavioral sensitization and leads to excessive alcohol intake in rats See also * 6-Hydroxydopamine * MPTP * Rotenone Rotenone is an odorless, colorless, crystalline isoflavone used as a broad-spectrum insecticide, piscicide, and pesticide. It occurs naturally in the seeds and stems of several plants, such as the jicama vine plant, and the roots of several member ... References Tetrahydroisoquinolines Catechols {{nervous-system-drug-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrogallol Ethers
Pyrogallol is an organic compound with the formula C6H3(OH)3. It is a water-soluble, white solid although samples are typically brownish because of its sensitivity toward oxygen. It is one of three isomers of benzenetriols. Production and reactions It is produced in the manner first reported by Scheele in 1786: heating gallic acid to induce decarboxylation. Gallic acid is also obtained from tannin. Many alternative routes have been devised. One preparation involves treating ''para''-chlorophenoldisulfonic acid with potassium hydroxide, a variant on the time-honored route to phenols from sulfonic acids. When in alkaline solution, pyrogallol undergoes deprotonation of one or more phenolic groups. Such solutions absorb oxygen from the air, turning brown. This conversion can be used to determine the amount of oxygen in a gas sample, notably by the use of the Orsat apparatus. Polyhydroxybenzenes are relatively electron-rich. One manifestation is the easy C-acetylation o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Macrocycles
Macrocycles are often described as molecules and ions containing a ring of twelve or more atoms. Classical examples include the crown ethers, calixarenes, porphyrins, and cyclodextrins. Macrocycles describe a large, mature area of chemistry. Synthesis The formation of macrocycles by ring-closure is called macrocylization. Pioneering work was reported for studies on terpenoid macrocycles. The central challenge to macrocyclization is that ring-closing reactions do not favor the formation of large rings. Instead, small rings or polymers tend to form. This kinetic problem can be addressed by using high-dilution reactions, whereby intramolecular processes are favored relative to polymerizations. Some macrocyclizations are favored using template reactions. Templates are ions, molecules, surfaces etc. that bind and pre-organize compounds, guiding them toward formation of a particular ring size. The crown ethers are often generated in the presence of an alkali metal cation, whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |