|
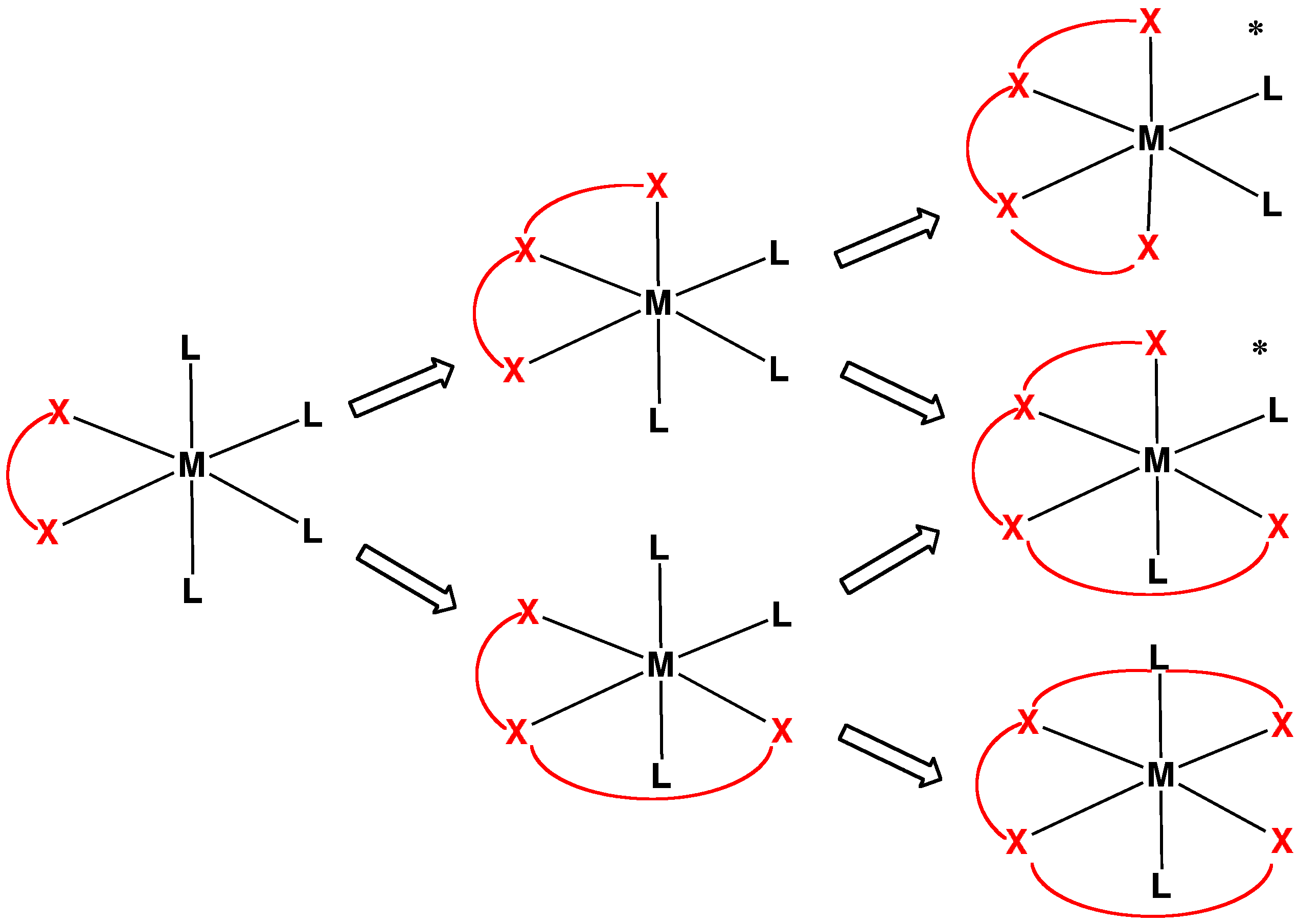
Denticity
In coordination chemistry, denticity () refers to the number of donor groups in a given ligand that bind to the central metal atom in a coordination complex. In many cases, only one atom in the ligand binds to the metal, so the denticity equals one, and the ligand is said to be monodentate (sometimes called unidentate). Ligands with more than one bonded atom are called polydentate or multidentate. The denticity of a ligand is described with the Greek letter κ ('kappa'). For example, κ6-EDTA describes an EDTA ligand that coordinates through 6 non-contiguous atoms. Denticity is different from hapticity because hapticity refers exclusively to ligands where the coordinating atoms are contiguous. In these cases the η ('eta') notation is used. Bridging ligands use the μ ('mu') notation. Classes Polydentate ligands are chelating agents and classified by their denticity. Some atoms cannot form the maximum possible number of bonds a ligand could make. In that case one or mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hapticity
In coordination chemistry, hapticity is the coordination of a ligand to a metal center via an uninterrupted and contiguous series of atoms. The hapticity of a ligand is described with the Greek letter η ('eta'). For example, η2 describes a ligand that coordinates through 2 contiguous atoms. In general the η-notation only applies when multiple atoms are coordinated (otherwise the κ-notation is used). In addition, if the ligand coordinates through multiple atoms that are not contiguous then this is considered denticity (not hapticity), and the κ-notation is used once again. When naming complexes care should be taken not to confuse η with μ ('mu'), which relates to bridging ligands. History The need for additional nomenclature for organometallic compounds became apparent in the mid-1950s when Dunitz, Orgel, and Rich described the structure of the " sandwich complex" ferrocene by X-ray crystallography where an iron atom is ''"sandwiched"'' between two parallel cyclopent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand
In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetradentate Ligand
In chemistry, tetradentate ligands are ligands that bind four donor atoms to a central atom to form a coordination complex. This number of donor atoms that bind is called denticity and is a method of classifying ligands. Tetradentate ligands are common in nature in the form of chlorophyll, which has a core ligand called chlorin, and heme, which has a core ligand called porphyrin. They are responsible for the colour observed in plants and humans. Phthalocyanine is an artificial macrocyclic tetradentate ligand that is used to make blue and green pigments. Shape Tetradentate ligands can be classified by the topology of the connections between donor atoms. Common forms are linear (also called sequential), ring or tripodal. A tetrapodal ligand that is also tetradentate has four legs with donor atoms and a bridgehead that is not a donor. Upon binding with a central atom, there are several arrangements possible (known as geometric isomers). Linear ligands A linear tetradentate ligand ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylenediamine
Ethylenediamine (abbreviated as en when a ligand) is the organic compound with the formula C2H4(NH2)2. This colorless liquid with an ammonia-like odor is a basic amine. It is a widely used building block in chemical synthesis, with approximately 500,000 tonnes produced in 1998.Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic" in Ullmann's Encyclopedia of Industrial Chemistry, 2005 Wiley-VCH Verlag, Weinheim. Ethylenediamine is the first member of the so-called polyethylene amines. Synthesis Ethylenediamine is produced industrially by treating 1,2-dichloroethane with ammonia under pressure at 180 °C in an aqueous medium:Hans-Jürgen Arpe, Industrielle Organische Chemie, 6. Auflage (2007), Seite 245, Wiley VCH : In this reaction hydrogen chloride is generated, which forms a salt with the amine. The amine is liberated by addition of sodium hydroxide and can then be recovered by . Diethylenetriamine (DETA) and triethylenetetramine (TET ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
EDTA
Ethylenediaminetetraacetic acid (EDTA) is an aminopolycarboxylic acid with the formula H2N(CH2CO2H)2sub>2. This white, water-soluble solid is widely used to bind to iron (Fe2+/Fe3+) and calcium ions (Ca2+), forming water-soluble complexes even at neutral pH. It is thus used to dissolve Fe- and Ca-containing scale as well as to deliver iron ions under conditions where its oxides are insoluble. EDTA is available as several salts, notably disodium EDTA, sodium calcium edetate, and tetrasodium EDTA, but these all function similarly. Uses Textile industry In industry, EDTA is mainly used to sequester (bind or confine) metal ions in aqueous solution. In the textile industry, it prevents metal ion impurities from modifying colours of dyed products. In the pulp and paper industry, EDTA inhibits the ability of metal ions, especially Mn2+, from catalysing the disproportionation of hydrogen peroxide, which is used in chlorine-free bleaching. In a similar manner, EDTA is added ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexadentate Ligand
A hexadentate ligand in coordination chemistry is a ligand that combines with a central metal atom with six bonds. One example of a hexadentate ligand that can form complexes with soft metal ions is TPEN. A commercially important hexadentate ligand is EDTA. The denticity In coordination chemistry, denticity () refers to the number of donor groups in a given ligand that bind to the central metal atom in a coordination complex. In many cases, only one atom in the ligand binds to the metal, so the denticity equals ... of hexadentate ligands is often denoted with the prefix κ6. Topology The way the donor atoms are joined together in the molecule is its topology. Some topologies are simple, such as the linear or ring shapes. The molecule can also be branched, either at a donor atom, or at a non-donor atom. Example shapes are the tripod, and amplector, with a bifurcation at each end. Rigid molecules can be used to force unusual coordination such as trigonal prism. F. Lions ident ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylenediaminetetraacetic Acid
Ethylenediaminetetraacetic acid (EDTA) is an aminopolycarboxylic acid with the formula H2N(CH2CO2H)2sub>2. This white, water-soluble solid is widely used to bind to iron (Fe2+/Fe3+) and calcium ions (Ca2+), forming water-soluble complexes even at neutral pH. It is thus used to dissolve Fe- and Ca-containing scale as well as to deliver iron ions under conditions where its oxides are insoluble. EDTA is available as several salts, notably disodium EDTA, sodium calcium edetate, and tetrasodium EDTA, but these all function similarly. Uses Textile industry In industry, EDTA is mainly used to sequester (bind or confine) metal ions in aqueous solution. In the textile industry, it prevents metal ion impurities from modifying colours of dyed products. In the pulp and paper industry, EDTA inhibits the ability of metal ions, especially Mn2+, from catalysing the disproportionation of hydrogen peroxide, which is used in chlorine-free bleaching. In a similar manner, EDTA is added ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kappa (letter)
Kappa (uppercase Κ, lowercase κ or cursive ; el, κάππα, ''káppa'') is the 10th letter of the Greek alphabet, representing the voiceless velar plosive sound in Ancient and Modern Greek. In the system of Greek numerals, has a value of 20. It was derived from the Phoenician letter kaph . Letters that arose from kappa include the Roman K and Cyrillic К. The uppercase form is identical to the Latin K. Greek proper names and placenames containing kappa are often written in English with "c" due to the Romans' transliterations into the Latin alphabet: Constantinople, Corinth, Crete. All formal modern romanizations of Greek now use the letter "k", however. The cursive form is generally a simple font variant of lower-case kappa, but it is encoded separately in Unicode for occasions where it is used as a separate symbol in math and science. In mathematics, the kappa curve is named after this letter; the tangents of this curve were first calculated by Isaac Barrow in th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetate
DOTA (also known as tetraxetan) is an organic compound with the formula (CH2CH2NCH2CO2H)4. The molecule consists of a central 12-membered tetraaza (i.e., containing four nitrogen atoms) ring. DOTA is used as a complexing agent, especially for lanthanide ions. Its complexes have medical applications as contrast agents and cancer treatments. Terminology The acronym DOTA (for dodecane tetraacetic acid) is shorthand for both the tetracarboxylic acid and its various conjugate bases. In the area of coordination chemistry, the tetraacid is called H4DOTA and its fully deprotonated derivative is DOTA4−. Many related ligands are referred to using the DOTA acronym, although these derivatives are generally not ''tetra''carboxylic acids or the conjugate bases. Structure DOTA is derived from the macrocycle known as cyclen. The four secondary amine groups are modified by replacement of the N-H centers with N-CH2CO2H groups. The resulting aminopolycarboxylic acid, upon ionization of the carbox ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylene Triamine Pentaacetic Acid
Pentetic acid or diethylenetriaminepentaacetic acid (DTPA) is an aminopolycarboxylic acid consisting of a diethylenetriamine backbone with five carboxymethyl groups. The molecule can be viewed as an expanded version of EDTA and is used similarly. It is a white solid with limited solubility in water. Coordination properties The conjugate base of DTPA has a high affinity for metal cations. Thus, the penta-anion DTPA5− is potentially an octadentate ligand assuming that each nitrogen centre and each –COO− group counts as a centre for coordination. The formation constants for its complexes are about 100 greater than those for EDTA.J. Roger Hart "Ethylenediaminetetraacetic Acid and Related Chelating Agents" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. As a chelating agent, DTPA wraps around a metal ion by forming up to eight bonds. Its complexes can also have an extra water molecule that coordinates the metal ion. Transition metals, however, u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,4,7-Trithiacyclononane
1,4,7-Trithiacyclononane, also called 9-ane-S3, is the thia-crown ether with the formula (CH2CH2S)3. This cyclic thioether is most often encountered as a tridentate ligand in coordination chemistry, where it forms transition metal thioether complexes. 9-ane-S3 forms complexes with many metal ions, including those considered hard Hard may refer to: * Hardness, resistance of physical materials to deformation or fracture * Hard water, water with high mineral content Arts and entertainment * ''Hard'' (TV series), a French TV series * Hard (band), a Hungarian hard rock super ..., such as copper(II) and iron(II). Most of its complexes have the formula (9-ane-S3)2sup>2+ and are octahedral. The point group of (9-ane-S3)2sup>2+ is S6. Synthesis This compound was first reported in 1977, and the current synthesis entails the assembly within the coordination sphere of a metal ion followed by decomplexation: : References {{DEFAULTSORT:Trithiacyclononane, 1,4,7- Chelating age ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

4-3D-balls.png)