|
Carboxylated
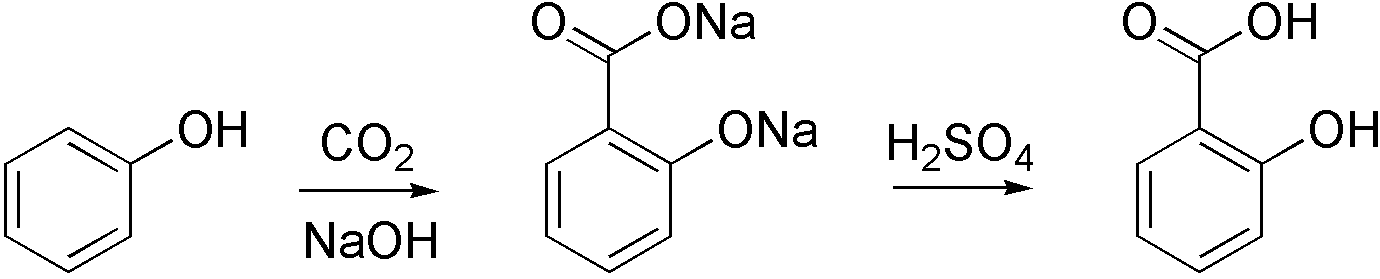
Carboxylation is a chemical reaction in which a carboxylic acid is produced by treating a substrate with carbon dioxide. The opposite reaction is decarboxylation. In chemistry, the term carbonation is sometimes used synonymously with carboxylation, especially when applied to the reaction of carbanionic reagents with CO2. More generally, carbonation usually describes the production of carbonates. Organic chemistry Carboxylation is a standard conversion in organic chemistry. Specifically carbonation (i.e. carboxylation) of Grignard reagents and organolithium compounds is a classic way to convert organic halides into carboxylic acids. Sodium salicylate, precursor to aspirin, is commercially prepared by treating sodium phenolate (the sodium salt of phenol) with carbon dioxide at high pressure (100 atm) and high temperature (390 K) – a method known as the Kolbe-Schmitt reaction. Acidification of the resulting salicylate salt gives salicylic acid. : Many detailed procedures are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the Atomic nucleus, nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive Chemical element, elements where both electronic and nuclear changes can occur. The substance (or substances) initially involved in a chemical reaction are called reagent, reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more Product (chemistry), products, which usually have properties different from the reactants. Reactions often consist of a sequence o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Syntheses
''Organic Syntheses'' is a peer-reviewed scientific journal that was established in 1921. It publishes detailed and checked procedures for the synthesis of organic compounds. A unique feature of the review process is that all of the data and experiments reported in an article must be successfully repeated in the laboratory of a member of the editorial board as a check for reproducibility prior to publication. The journal is published by Organic Syntheses, Inc., a non-profit corporation. An annual print version is published by John Wiley & Sons on behalf of Organic Syntheses, Inc. History Prior to World War I, work on synthetic organic chemistry in the United States had been quite limited, and most of the reagents used in laboratories had to be imported from Europe. When export stoppages and trade embargoes cut off this source, Clarence Derick, a professor of chemistry at University of Illinois at Urbana-Champaign, began an effort to synthesize these needed chemicals in industr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Posttranslational Modification
Post-translational modification (PTM) is the covalent and generally enzymatic modification of proteins following protein biosynthesis. This process occurs in the endoplasmic reticulum and the golgi apparatus. Proteins are synthesized by ribosomes translating mRNA into polypeptide chains, which may then undergo PTM to form the mature protein product. PTMs are important components in cell signal transduction, signaling, as for example when prohormones are converted to hormones. Post-translational modifications can occur on the amino acid side chains or at the protein's C-terminus, C- or N-terminus, N- termini. They can extend the chemical repertoire of the 20 standard amino acids by modifying an existing functional group or introducing a new one such as phosphorylation, phosphate. Phosphorylation is a highly effective mechanism for regulating the activity of enzymes and is the most common post-translational modification. Many eukaryotic and prokaryotic proteins also have carbohydra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
National Institutes Of Health
The National Institutes of Health, commonly referred to as NIH (with each letter pronounced individually), is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the late 1880s and is now part of the United States Department of Health and Human Services. The majority of NIH facilities are located in Bethesda, Maryland, and other nearby suburbs of the Washington metropolitan area, with other primary facilities in the Research Triangle Park in North Carolina and smaller satellite facilities located around the United States. The NIH conducts its own scientific research through the NIH Intramural Research Program (IRP) and provides major biomedical research funding to non-NIH research facilities through its Extramural Research Program. , the IRP had 1,200 principal investigators and more than 4,000 postdoctoral fellows in basic, translational, and clinical research, being the largest biomedical research instit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biotin
Biotin (or vitamin B7) is one of the B vitamins. It is involved in a wide range of metabolic processes, both in humans and in other organisms, primarily related to the utilization of fats, carbohydrates, and amino acids. The name ''biotin'', borrowed from the German , derives from the Ancient Greek word (; 'life') and the suffix "-in" (a suffix used in chemistry usually to indicate 'forming'). Chemical description Biotin is classified as a heterocyclic compound, with a sulfur-containing ring fused ureido and tetrahydrothiophene group. A C5-carboxylic acid side chain is appended to one of the rings. The ureido ring, containing the −N−CO−N− group, serves as the carbon dioxide carrier in carboxylation reactions. Biotin is a coenzyme for five carboxylase enzymes, which are involved in the catabolism of amino acids and fatty acids, synthesis of fatty acids, and gluconeogenesis. Biotinylation of histone proteins in nuclear chromatin plays a role in chromatin stability and g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyruvate Carboxylase
Pyruvate carboxylase (PC) encoded by the gene PC is an enzyme () of the ligase class that catalyzes (depending on the species) the physiologically irreversible carboxylation of pyruvate to form oxaloacetate (OAA). Image:Pyruvic-acid-2D-skeletal.png , Pyruvic acid Image:Oxaloacetic acid.png , Oxaloacetic acid The reaction it catalyzes is: :pyruvate + + ATP → oxaloacetate + ADP + P It is an important anaplerotic reaction that creates oxaloacetate from pyruvate. The enzyme is a mitochondrial protein containing a biotin prosthetic group, requiring magnesium or manganese and acetyl-CoA. Pyruvate carboxylase was first discovered in 1959 at Case Western Reserve University by M. F. Utter and D. B. Keech. Since then it has been found in a wide variety of prokaryotes and eukaryotes including fungi, bacteria, plants, and animals. In mammals, PC plays a crucial role in gluconeogenesis and lipogenesis, in the biosynthesis of neurotransmitters, and in glucose-induced insulin secr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propionyl-CoA Carboxylase
Propionyl-CoA carboxylase (, PCC) catalyses the carboxylation reaction of propionyl-CoA in the mitochondrial matrix. PCC has been classified both as a ligase and a lyase. The enzyme is biotin-dependent. The product of the reaction is (S)-methylmalonyl CoA. : ATP + propionyl-CoA + HCO3− ADP + phosphate + (S)-methylmalonyl-CoA (S)-Methylmalonyl-CoA cannot be directly utilized by animals. It is acted upon by a racemase, yielding (R)-methylmalonyl-CoA, which is then converted into succinyl-CoA by methylmalonyl-CoA mutase (one of the few metabolic enzymes which requires vitamin B12 as a cofactor). Succinyl-CoA, a Krebs cycle intermediate, is further metabolized into fumarate, then malate, and then oxaloacetate. Oxaloacetate may be transported into the cytosol to form phosphoenol pyruvate and other gluconeogenic intermediates. Propionyl-CoA is therefore an important precursor to glucose. Propionyl-CoA is the end product of odd-chain fatty acid metabolism, including most methylated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylcrotonyl-CoA Carboxylase
Methylcrotonyl CoA carboxylase (, MCC) (3-methylcrotonyl CoA carboxylase, methylcrotonoyl-CoA carboxylase) is a biotin-requiring enzyme located in the mitochondria. MCC uses bicarbonate as a carboxyl group source to catalyze the carboxylation of a carbon adjacent to a carbonyl group performing the fourth step in processing leucine, an essential amino acid. Structure Gene Human MCC is a biotin dependent mitochondrial enzyme formed by the two subunits MCCCα and MCCCβ, encoded by ''MCCC1'' and ''MCCC2'' respectively. MCCC1 gene has 21 exons and resides on chromosome 3 at q27. MCCC2 gene has 19 exons and resides on chromosome 5 at q12-q13. Protein The enzyme contains α and β subunits. Human MCCCα is composed of 725 amino acids which harbor a covalently bound biotin essential for the ATP-dependent carboxylation; MCCCβ has 563 amino acids that possess carboxyltransferase activity which presumably is essential for binding to 3-methylcrotonyl CoA. The MCC holoenzyme is thou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
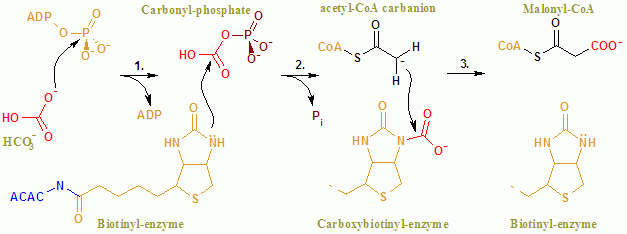
Acetyl-CoA Carboxylase
Acetyl-CoA carboxylase (ACC) is a biotin-dependent enzyme () that catalyzes the irreversible carboxylation of acetyl-CoA to produce malonyl-CoA through its two catalytic activities, biotin carboxylase (BC) and carboxyltransferase (CT). ACC is a multi-subunit enzyme in most prokaryotes and in the chloroplasts of most plants and algae, whereas it is a large, multi-domain enzyme in the cytoplasm of most eukaryotes. The most important function of ACC is to provide the malonyl-CoA substrate for the biosynthesis of fatty acids. The activity of ACC can be controlled at the transcriptional level as well as by small molecule modulators and covalent modification. The human genome contains the genes for two different ACCs—'' ACACA'' and ''ACACB''. Structure Prokaryotes and plants have multi-subunit ACCs composed of several polypeptides. Biotin carboxylase (BC) activity, biotin carboxyl carrier protein (BCCP), and carboxyl transferase (CT) activity are each contained on a different s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxyglutamic Acid
Carboxyglutamic acid (or the conjugate base, carboxyglutamate), is an uncommon amino acid introduced into proteins by a post-translational carboxylation of glutamic acid residues. This modification is found, for example, in clotting factors and other proteins of the coagulation cascade. This modification introduces an affinity for calcium ions. In the blood coagulation cascade, vitamin K is required to introduce γ-carboxylation of clotting factors II, VII, IX, X and protein Z. Synthesis In the biosynthesis of γ-carboxyglutamic acid, the γ-proton on glutamic acid is abstracted, and CO2 is subsequently added. The reaction intermediate is a γ-glutamyl carbanion. This reaction is catalyzed by a carboxylase that requires vitamin K as its cofactor. It is not exactly known how vitamin K participates, but it is hypothesized that a free cysteine residue in the carboxylase converts vitamin K into an active strong base that in turn abstracts a hydrogen from glutamic acid's γ-carbon. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid residue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |