|
Cyclopropylacetylene
Cyclopropylacetylene is an hydrocarbon with the chemical formula . Under normal conditions, the substance is a colorless liquid. Cyclopropylacetylene is a precursor pharmaceuticals and other organic compounds. Synthesis Several methods have been published on the synthesis of cyclopropylacetylene. The earliest preparation starts with the chlorination reaction, chlorination of cyclopropylmethylketone with phosphorus pentachloride. Thereafter, the reaction product, 1-cyclopropyl-1,1-dichloroethane, is converted into cyclopropylacetylene via double dehydrochlorination. This occurs in presence of a strongly basic solution, such as potassium tert-butoxide, potassium ''tert''-butoxide in dimethyl sulfoxide: However, the yield of this method is not substantial (20-25%). A one-pot synthesis of cyclopropylacetylene has been reported in which 5-chloro-1-pentyne reacts with n-butyl lithium, ''n''-butyl lithium or Hexyllithium, ''n''-hexyl lithium. Cyclohexane is used as a solvent. The react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or exemplified by the odors of gasoline and lighter fluid. They occur in a diverse range of molecular structures and phases: they can be gases (such as methane and propane), liquids (such as hexane and benzene), low melting solids (such as paraffin wax and naphthalene) or polymers (such as polyethylene and polystyrene). In the fossil fuel industries, ''hydrocarbon'' refers to the naturally occurring petroleum, natural gas and coal, and to their hydrocarbon derivatives and purified forms. Combustion of hydrocarbons is the main source of the world's energy. Petroleum is the dominant raw-material source for organic commodity chemicals such as solvents and polymers. Most anthropogenic (human-generated) emissions of greenhouse gases are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexane is mainly used for the industrial production of adipic acid and caprolactam, which are precursors to nylon. Cyclohexyl () is the alkyl substituent of cyclohexane and is abbreviated Cy. Production Modern On an industrial scale, cyclohexane is produced by hydrogenation of benzene in the presence of a Raney nickel catalyst. Producers of cyclohexane account for approximately 11.4% of global demand for benzene. The reaction is highly exothermic, with ΔH(500 K) = -216.37 kJ/mol. Dehydrogenation commenced noticeably above 300 °C, reflecting the favorable entropy for dehydrogenation. : Early Unlike benzene, cyclohexane is not found in natural resources such as coal. For this reason, early investigators synthesized their cyclohex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
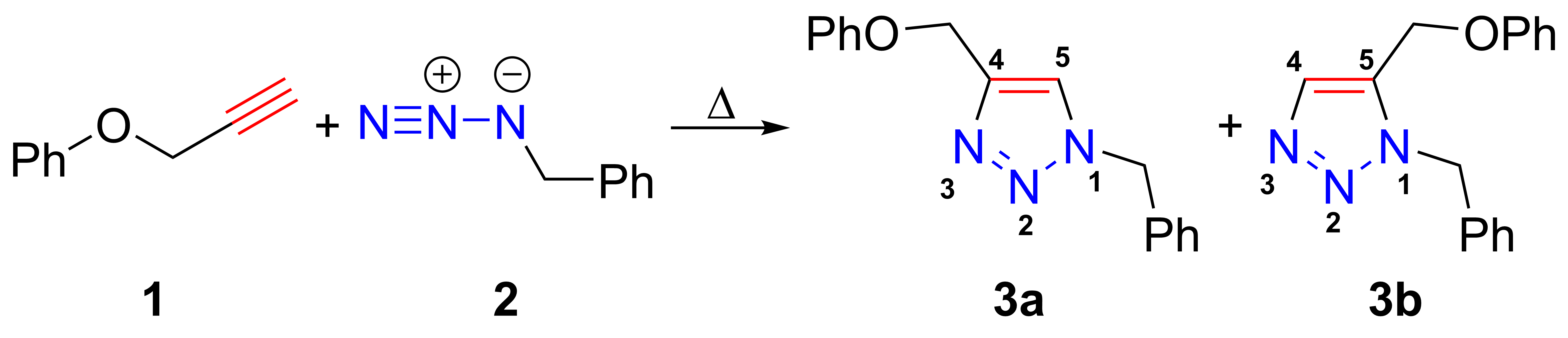
Azide-alkyne Huisgen Cycloaddition
The azide-alkyne Huisgen cycloaddition is a 1,3-dipolar cycloaddition between an azide and a terminal or internal alkyne to give a 1,2,3-triazole. Rolf Huisgen was the first to understand the scope of this organic reaction. American chemist Karl Barry Sharpless has referred to this cycloaddition as "the cream of the crop" of click chemistry and "the premier example of a click reaction". In the reaction above azide 2 reacts neatly with alkyne 1 to afford the product triazole as a mixture of 1,4-adduct (3a) and 1,5-adduct (3b) at 98 °C in 18 hours. The standard 1,3-cycloaddition between an azide 1,3-dipole and an alkene as dipolarophile has largely been ignored due to lack of reactivity as a result of electron-poor olefins and elimination side reactions. Some success has been found with non-metal-catalyzed cycloadditions, such as the reactions using dipolarophiles that are electron-poor olefins or alkynes. Although azides are not the most reactive 1,3-dipole available for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Efavirenz
Efavirenz (EFV), sold under the brand names Sustiva among others, is an antiretroviral medication used to treat and prevent HIV/AIDS. It is generally recommended for use with other antiretrovirals. It may be used for prevention after a needlestick injury or other potential exposure. It is sold both by itself and in combination as efavirenz/emtricitabine/tenofovir. It is taken by mouth. Common side effects include rash, nausea, headache, feeling tired, and trouble sleeping. Some of the rashes may be serious such as Stevens–Johnson syndrome. Other serious side effects include depression, thoughts of suicide, liver problems, and seizures. It is not safe for use during pregnancy. It is a non-nucleoside reverse transcriptase inhibitor (NNRTI) and works by blocking the function of reverse transcriptase. Efavirenz was approved for medical use in the United States in 1998, and in the European Union in 1999. It is on the World Health Organization's List of Essential Medicines. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antiretroviral
The management of HIV/AIDS normally includes the use of multiple antiretroviral drugs as a strategy to control HIV infection. There are several classes of antiretroviral agents that act on different stages of the HIV life-cycle. The use of multiple drugs that act on different viral targets is known as highly active antiretroviral therapy (HAART). HAART decreases the patient's total burden of HIV, maintains function of the immune system, and prevents opportunistic infections that often lead to death. HAART also prevents the transmission of HIV between serodiscordant same sex and opposite sex partners so long as the HIV-positive partner maintains an undetectable viral load. Treatment has been so successful that in many parts of the world, HIV has become a chronic condition in which progression to AIDS is increasingly rare. Anthony Fauci, head of the United States National Institute of Allergy and Infectious Diseases, has written, "With collective and resolute action now and a st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclization
A cyclic compound (or ring compound) is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where all the atoms are carbon (i.e., are carbocycles), none of the atoms are carbon (inorganic cyclic compounds), or where both carbon and non-carbon atoms are present ( heterocyclic compounds). Depending on the ring size, the bond order of the individual links between ring atoms, and their arrangements within the rings, carbocyclic and heterocyclic compounds may be aromatic or non-aromatic; in the latter case, they may vary from being fully saturated to having varying numbers of multiple bonds between the ring atoms. Because of the tremendous diversity allowed, in combination, by the valences of common atoms and their ability to form rings, the number of possible cyclic structures, even of small size (e.g., < 17 total atoms) numbers in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metalation
Metalation (Alt. spelling: Metallation) is a chemical reaction that forms a bond to a metal. This reaction usually refers to the replacement of a halogen atom in an organic molecule with a metal atom, resulting in an organometallic compound. In the laboratory, metalation is commonly used to activate organic molecules during the formation of C—X bonds (where X is typically carbon, oxygen, or nitrogen), which are necessary for the synthesis of many organic molecules. In synthesis, metallated reagents are typically involved in nucleophilic substitution, single-electron-transfer (SET), and redox chemistry with functional groups on other molecules (including but not limited to ketones, aldehydes and alkyl halides). Metallated molecules may also participate in acid-base chemistry, with one organometallic reagent deprotonating an organic molecule to create a new organometallic reagent. The most common classes of metallated compounds are organolithium reagents and Grignard reagen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexyllithium
''n''-Hexyllithium, C6H13Li, sometimes abbreviated to HxLi or NHL, is an organolithium compound used in organic synthesis as a strong base or as a lithiation reagent. It is usually encountered as a colorless or pale yellow solution in hexanes. Such solutions are highly sensitive to air and can ignite when treated with water. In terms of chemical properties, hexyllithium and ''n''-butyllithium (BuLi) are very similar. As a base, hexyllithium generates ''n''-hexane as a byproduct rather than gaseous butane Butane () or ''n''-butane is an alkane with the formula C4H10. Butane is a gas at room temperature and atmospheric pressure. Butane is a highly flammable, colorless, easily liquefied gas that quickly vaporizes at room temperature. The name bu ..., which results from the use of BuLi. Another advantage for HxLi is that it is slightly less reactive. Both of these aspects encourage industrial applications. It is commercially available as a solution in mixed hexanes, usua ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorination Reaction
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (F2, Cl2, Br2, I2). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for and introducing halogens into diverse substrates, e.g. thionyl chloride. Organic chemistry Several pathways exist for the halogenation of organic compounds, including free radical halogenation, ketone halogenation, electrophilic halogenation, and halogen addition reaction. The nature of the substrate determines the pathway. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-butyl Lithium
''n''-Butyllithium C4H9Li (abbreviated ''n''-BuLi) is an organolithium reagent. It is widely used as a polymerization initiator in the production of elastomers such as polybutadiene or styrene-butadiene-styrene (SBS). Also, it is broadly employed as a strong base (superbase) in the synthesis of organic compounds as in the pharmaceutical industry. Butyllithium is commercially available as solutions (15%, 25%, 1.5 M, 2 M, 2.5 M, 10 M, etc.) in alkanes such as pentane, hexanes, and heptanes. Solutions in diethyl ether and THF can be prepared, but are not stable enough for storage. Annual worldwide production and consumption of butyllithium and other organolithium compounds is estimated at 2000 to 3000 tonnes. Although butyllithium is colorless, ''n''-butyllithium is usually encountered as a pale yellow solution in alkanes. Such solutions are stable indefinitely if properly stored,. but in practice, they degrade upon aging. Fine white precipitate (lit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |