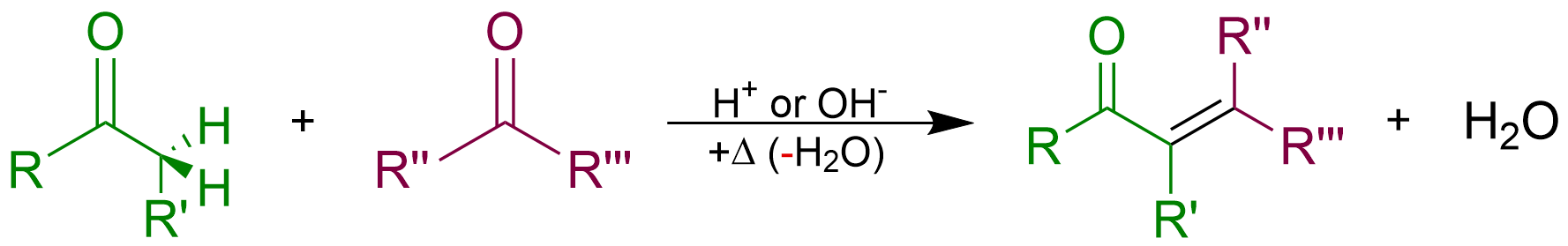|
Butanal
Butyraldehyde, also known as butanal, is an organic compound with the formula CH3(CH2)2CHO. This compound is the aldehyde derivative of butane. It is a colorless flammable liquid with an unpleasant smell. It is miscible with most organic solvents. Production Butyraldehyde is produced almost exclusively by the hydroformylation of propylene: : CH3CH=CH2 + H2 + CO → CH3CH2CH2CHO Traditionally, hydroformylation was catalyzed by cobalt carbonyl and later rhodium complexes of triphenylphosphine. The dominant technology involves the use of rhodium catalysts derived from the water-soluble ligand tppts. An aqueous solution of the rhodium catalyst converts the propylene to the aldehyde, which forms a lighter immiscible phase. About 6 billion kilograms are produced annually by hydroformylation. Butyraldehyde can be produced by the catalytic dehydrogenation of ''n''-butanol. At one time, it was produced industrially by the catalytic hydrogenation of crotonaldehyde, which is derived from a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tppts
3,3′,3′′-Phosphanetriyltris(benzenesulfonic acid) trisodium salt (abbreviated TPPTS), is an organic compound that is also known as sodium triphenylphosphine trisulfonate. The compound has the formula P(C6H4SO3Na)3. This white solid is an unusual example of a water-soluble phosphine. Its complexes are also water-soluble. Its complex with rhodium is used in the industrial production of butyraldehyde. Synthesis TPPTS is synthesized by sulfonation of triphenylphosphine. The sulfonation occurs at one meta-position of each of the three phenyl rings. The sulfonation agent is oleum, a solution of sulfur trioxide in sulfuric acid. Immediately upon dissolving in the oleum, the phosphine is protonated. It is the phosphonium salt that undergoes the sulfonation which explains its meta selectivity: :HP(C6H5)3+ + 3 SO3 → P(C6H4SO3H)3sup>+ As a Lewis base, tppts is stronger than triphenylphosphine. TPPTS at the origin of two-phase homogeneous catalysis TPPTS was first syn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isobutyraldehyde
Isobutyraldehyde is the chemical compound with the formula (CH3)2CHCHO. It is an aldehyde, isomeric with n-butyraldehyde (butanal). Isobutyraldehyde is made, often as a side-product, by the hydroformylation of propene. Its odour is described as that of wet cereal or straw. It undergoes the Cannizaro reaction even though it has alpha hydrogen atom. It is a colorless volatile liquid. Synthesis Isobutyraldehyde is produced industrially by the hydroformylation of propene. Several million tons are produced annually.Boy Cornils, Richard W. Fischer, Christian Kohlpaintner "Butanals" in Ullmann's Encyclopedia of Industrial Chemistry, 2000, Wiley-VCH, Weinheim. Biological routes In the context of butanol fuel, isobutyraldehyde is of interest as a precursor to isobutanol. ''E. coli'' as well as several other organisms has been genetically modified to produce isobutanol. α-Ketoisovalerate, derived from oxidative deamination of valine, is prone to decarboxylation to give isobutyraldehyd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Solvents
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for polar molecules and the most common solvent used by living things; all the ions and proteins in a cell are dissolved in water within the cell. The quantity of solute that can dissolve in a specific volume of solvent varies with temperature. Major uses of solvents are in paints, paint removers, inks, and dry cleaning. Specific uses for organic solvents are in dry cleaning (e.g. tetrachloroethylene); as paint thinners (toluene, turpentine); as nail polish removers and solvents of glue (acetone, methyl acetate, ethyl acetate); in spot removers (hexane, petrol ether); in detergents ( citrus terpenes); and in perfumes (ethanol). Solvents find various applications in chemical, pharmaceutical, oil, and gas industries, including in chemical synt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavors
Flavor or flavour is either the sensory perception of taste or smell, or a flavoring in food that produces such perception. Flavor or flavour may also refer to: Science *Flavors (programming language), an early object-oriented extension to Lisp * Flavour (particle physics), a quantum number of elementary particles related to their weak interactions *Flavor of Linux, another term for any particular Linux distribution; by extension, "flavor" can be applied to any program or other computer code that exists in more than one current variant at the same time Film and TV * ''Flavors'' (film), romantic comedy concerning Asian-Indian immigrants in America Music Artists and bands *Flavor Flav (born 1959), former rap/hip-hop promoter and current reality television actor *Flavour N'abania (born 1983), Nigerian singer-songwriter * Flavor (band), minor hit with "Sally Had A Party" in 1968 Albums * ''Flavours'' (album), 1975 album by The Guess Who * ''Flavors'' (album), by American R&B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bis(2-ethylhexyl) Phthalate
Bis(2-ethylhexyl) phthalate (di-2-ethylhexyl phthalate, diethylhexyl phthalate, diisooctyl phthalate, DEHP; incorrectly — dioctyl phthalate, DIOP) is an organic compound with the formula C6H4(CO2C8H17)2. DEHP is the most common member of the class of phthalates, which are used as plasticizers. It is the diester of phthalic acid and the branched-chain 2-ethylhexanol. This colorless viscous liquid is soluble in oil, but not in water. Production Di(2-ethylhexyl) phthalate is produced commercially by the reaction of excess 2-ethylhexanol with phthalic anhydride in the presence of an acid catalyst such as sulfuric acid or ''para''-toluenesulfonic acid. It was first produced in commercial quantities in Japan around 1933 and in the United States in 1939. : As 2-ethylhexanol is produced as a racemic mixture, DEHP consists of the (''R'',''R'')- and (''S'',''S'')-diasteromers, and the '' meso''-isomer. : Use Due to its suitable properties and the low cost, DEHP is widely used as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyd
An alkyd is a polyester resin modified by the addition of fatty acids and other components. Alkyds are derived from polyols and organic acids including dicarboxylic acids or carboxylic acid anhydride and triglyceride oils. The term ''alkyd'' is a modification of the original name "alcid", reflecting the fact that they are derived from ''alc''ohol and organic ac''id''s. The inclusion of a fatty acid confers a tendency to form flexible coatings. Alkyds are used in paints, varnishes and in moulds for casting. They are the dominant resin or binder in most commercial oil-based coatings. Approximately 200,000 tons of alkyd resins are produced each year.. Published online: 15 January 2003. The original alkyds were compounds of glycerol and phthalic acid sold under the name Glyptal. These were sold as substitutes for the darker-colored copal resins, thus creating alkyd varnishes that were much paler in colour. From these, the alkyds that are known today were developed. Manufacture Alky ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trimethylolpropane
300 C--> Trimethylolpropane (TMP) is the organic compound with the formula CH3CH2C(CH2OH)3. This colourless to white solid with a faint odor is a triol. Containing three hydroxy functional groups, TMP is a widely used building block in the polymer industry. Production TMP is produced via a two step process, starting with the condensation of butanal with formaldehyde: :CH3CH2CH2CHO + 2 CH2O → CH3CH2C(CH2OH)2CHO The second step entails a Cannizaro reaction: :CH3CH2C(CH2OH)2CHO + CH2O + NaOH → CH3CH2C(CH2OH)3 + NaO2CH Approximately 200,000,000 kg are produced annually in this way.Peter Werle, Marcus Morawietz, Stefan Lundmark, Kent Sörensen, Esko Karvinen, Juha Lehtonen “Alcohols, Polyhydric” in Ullmann’s Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2008. Applications TMP is mainly consumed as a precursor to alkyd resins. Otherwise, acrylated and alkoxylated TMP's are used as multifunctional monomers to produce various coatings, Ethoxylated a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bis(2-ethylhexyl) Phthalate
Bis(2-ethylhexyl) phthalate (di-2-ethylhexyl phthalate, diethylhexyl phthalate, diisooctyl phthalate, DEHP; incorrectly — dioctyl phthalate, DIOP) is an organic compound with the formula C6H4(CO2C8H17)2. DEHP is the most common member of the class of phthalates, which are used as plasticizers. It is the diester of phthalic acid and the branched-chain 2-ethylhexanol. This colorless viscous liquid is soluble in oil, but not in water. Production Di(2-ethylhexyl) phthalate is produced commercially by the reaction of excess 2-ethylhexanol with phthalic anhydride in the presence of an acid catalyst such as sulfuric acid or ''para''-toluenesulfonic acid. It was first produced in commercial quantities in Japan around 1933 and in the United States in 1939. : As 2-ethylhexanol is produced as a racemic mixture, DEHP consists of the (''R'',''R'')- and (''S'',''S'')-diasteromers, and the '' meso''-isomer. : Use Due to its suitable properties and the low cost, DEHP is widely used as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2-ethylhexanol
2-Ethylhexanol (abbreviated 2-EH) is an organic compound with formula CHO. It is a branched, eight-carbon chiral alcohol (chemistry), alcohol. It is a colorless liquid that is poorly soluble in water but soluble in most organic solvents. It is produced on a large scale (>2,000,000,000 kg/y) for use in numerous applications such as solvents, flavors, and fragrances and especially as a precursor for production of other chemicals such as emollients and plasticizers. It is encountered in plants, fruits, and wines. The odor has been reported as "heavy, earthy, and slightly floral" for the R enantiomer and "a light, sweet floral fragrance" for the S enantiomer. Properties and applications The branching in 2-ethylhexanol inhibits crystallization. Esters of 2-ethylhexanol are similarly affected, which together with low volatility, is the basis of applications in the production of plasticizers and lubricants, where its presence helps reduce viscosity and lower freezing points. Because 2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction is as follows (where the Rs can be H): Aldol condensations are important in organic synthesis and biochemistry as ways to form carbon–carbon bonds. In its usual form, it involves the nucleophilic addition of a ketone enolate to an aldehyde to form a β-hydroxy ketone, or "aldol" (aldehyde + alcohol), a structural unit found in many naturally occurring molecules and pharmaceuticals. The term ''aldol condensation'' is also commonly used, especially in biochemistry, to refer to just the first (addition) stage of the process—the aldol reaction itself—as catalyzed by aldolases. However, this is formally an addition reaction rather than a condensation reaction because it does not invo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogenation, C=C (and other) bonds ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetaldehyde
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3 CHO, sometimes abbreviated by chemists as MeCHO (Me = methyl). It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body. The International Agency for Research on Cancer (IARC) has listed acetaldehyde as a Group 1 carcinogen. Acetaldehyde is "one of the mos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |