|
Blebbishield Emergency Program
Blebbishield emergency program is a process which acts as a last line of defense for cancer stem cells after induction of apoptosis where the apoptotic blebs fuse to shield the cells/nucleus from the destructive force of apoptosis by forming blebbishields. Blebbishields in turn fuse to each other and generate cancer stem cell spheres/cellular transformation, essentially shifting the balance of dying cells back towards survival. Discovery Blebbishields were first identified in human bladder cancer cell linRT4(HTB-2: ATCC), referred to as RT4P (RT4 parent) in the initial report. Jinesh GG, Choi W, Shah JB, Lee EK, Willis DL, Kamat AM Cell Death Differ. 2013 Mar;20(3):382-95. Blebbishield formation Every cell type, especially cancer cells, are capable of undergoing apoptosis, a process in which the plasma membrane undergoes blebbing followed by orderly deconstruction of cells into apoptotic bodies. Cancer stem cells have the extraordinary ability to construct blebbishields from t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cancer Stem Cells
Cancer stem cells (CSCs) are cancer cells (found within tumors or hematological cancers) that possess characteristics associated with normal stem cells, specifically the ability to give rise to all cell types found in a particular cancer sample. CSCs are therefore tumorigenic (tumor-forming), perhaps in contrast to other non-tumorigenic cancer cells. CSCs may generate tumors through the stem cell processes of self-renewal and differentiation into multiple cell types. Such cells are hypothesized to persist in tumors as a distinct population and cause relapse and metastasis by giving rise to new tumors. Therefore, development of specific therapies targeted at CSCs holds hope for improvement of survival and quality of life of cancer patients, especially for patients with metastatic disease. Existing cancer treatments have mostly been developed based on animal models, where therapies able to promote tumor shrinkage were deemed effective. However, animals do not provide a complete m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vascular Endothelial Growth Factor
Vascular endothelial growth factor (VEGF, ), originally known as vascular permeability factor (VPF), is a signal protein produced by many cells that stimulates the formation of blood vessels. To be specific, VEGF is a sub-family of growth factors, the platelet-derived growth factor family of cystine-knot growth factors. They are important signaling proteins involved in both vasculogenesis (the '' de novo'' formation of the embryonic circulatory system) and angiogenesis (the growth of blood vessels from pre-existing vasculature). It is part of the system that restores the oxygen supply to tissues when blood circulation is inadequate such as in hypoxic conditions. Serum concentration of VEGF is high in bronchial asthma and diabetes mellitus. VEGF's normal function is to create new blood vessels during embryonic development, new blood vessels after injury, muscle following exercise, and new vessels (collateral circulation) to bypass blocked vessels. It can contribute to disease. So ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
V-ATPase
Vacuolar-type ATPase (V-ATPase) is a highly conserved evolutionarily ancient enzyme with remarkably diverse functions in eukaryotic organisms. V-ATPases acidify a wide array of intracellular organelles and pumps protons across the plasma membranes of numerous cell types. V-ATPases couple the energy of ATP hydrolysis to proton transport across intracellular and plasma membranes of eukaryotic cells. It is generally seen as the polar opposite of ATP synthase because ATP synthase is a proton channel that uses the energy from a proton gradient to produce ATP. V-ATPase however, is a proton pump that uses the energy from ATP hydrolysis to produce a proton gradient. The Archaea-type ATPase (A-ATPase) is a related group of ATPases found in archaea that often work as an ATP synthase. It forms a clade V/A-ATPase with V-ATPase. Most members of either group shuttle protons (), but a few members have evolved to use sodium ions () instead. Roles played by V-ATPases V-ATPases are foun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-linked Glycosylation
''N''-linked glycosylation, is the attachment of an oligosaccharide, a carbohydrate consisting of several sugar molecules, sometimes also referred to as glycan, to a nitrogen atom (the amide nitrogen of an asparagine (Asn) residue of a protein), in a process called ''N''-glycosylation, studied in biochemistry. This type of linkage is important for both the structure and function of many eukaryotic proteins. The ''N''-linked glycosylation process occurs in eukaryotes and widely in archaea, but very rarely in bacteria. The nature of ''N''-linked glycans attached to a glycoprotein is determined by the protein and the cell in which it is expressed. It also varies across species. Different species synthesize different types of ''N''-linked glycan. Energetics of bond formation There are two types of bonds involved in a glycoprotein: bonds between the saccharides residues in the glycan and the linkage between the glycan chain and the protein molecule. The sugar moieties are linked t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycolysis
Glycolysis is the metabolic pathway that converts glucose () into pyruvate (). The free energy released in this process is used to form the high-energy molecules adenosine triphosphate (ATP) and reduced nicotinamide adenine dinucleotide (NADH). Glycolysis is a sequence of ten reactions catalyzed by enzymes. Glycolysis is a metabolic pathway that does not require oxygen (In anaerobic conditions pyruvate is converted to lactic acid). The wide occurrence of glycolysis in other species indicates that it is an ancient metabolic pathway. Indeed, the reactions that make up glycolysis and its parallel pathway, the pentose phosphate pathway, occur in the oxygen-free conditions of the Archean oceans, also in the absence of enzymes, catalyzed by metal. In most organisms, glycolysis occurs in the liquid part of cells, the cytosol. The most common type of glycolysis is the ''Embden–Meyerhof–Parnas (EMP) pathway'', which was discovered by Gustav Embden, Otto Meyerhof, and Jakub Karol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bcl-2 Homologous Antagonist Killer
Bcl-2 homologous antagonist/killer is a protein that in humans is encoded by the ''BAK1'' gene on chromosome 6. The protein encoded by this gene belongs to the BCL2 protein family. BCL2 family members form oligomers or heterodimers and act as anti- or pro-apoptotic regulators that are involved in a wide variety of cellular activities. This protein localizes to mitochondria, and functions to induce apoptosis. It interacts with and accelerates the opening of the mitochondrial voltage-dependent anion channel, which leads to a loss in membrane potential and the release of cytochrome c. This protein also interacts with the tumor suppressor P53 after exposure to cell stress. Structure BAK1 is a pro-apoptotic Bcl-2 protein containing four Bcl-2 homology (BH) domains: BH1, BH2, BH3, and BH4. These domains are composed of nine α-helices, with a hydrophobic α-helix core surrounded by amphipathic helices and a transmembrane C-terminal α-helix anchored to the mitochondrial outer membran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bcl-2-associated X Protein
Apoptosis regulator BAX, also known as bcl-2-like protein 4, is a protein that in humans is encoded by the ''BAX'' gene. ''BAX'' is a member of the Bcl-2 gene family. BCL2 family members form hetero- or homodimers and act as anti- or pro-apoptotic regulators that are involved in a wide variety of cellular activities. This protein forms a heterodimer with BCL2, and functions as an apoptotic activator. This protein is reported to interact with, and increase the opening of, the mitochondrial voltage-dependent anion channel (VDAC), which leads to the loss in membrane potential and the release of cytochrome c. The expression of this gene is regulated by the tumor suppressor P53 and has been shown to be involved in P53-mediated apoptosis. Structure The ''BAX'' gene was the first identified pro-apoptotic member of the Bcl-2 protein family. Bcl-2 family members share one or more of the four characteristic domains of homology entitled the Bcl-2 homology (BH) domains (named BH1, BH2, B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P27 (gene)
Cyclin-dependent kinase inhibitor 1B (p27Kip1) is an enzyme inhibitor that in humans is encoded by the CDKN1B gene. It encodes a protein which belongs to the ''Cip/Kip'' family of cyclin dependent kinase (Cdk) inhibitor proteins. The encoded protein binds to and prevents the activation of cyclin E-CDK2 or cyclin D-CDK4 complexes, and thus controls the cell cycle progression at G1. It is often referred to as a cell cycle inhibitor protein because its major function is to stop or slow down the cell division cycle. Function The p27Kip1 gene has a DNA sequence similar to other members of the "Cip/Kip" family which include the p21Cip1/Waf1 and p57Kip2 genes. In addition to this structural similarity the "Cip/Kip" proteins share the functional characteristic of being able to bind several different classes of Cyclin and Cdk molecules. For example, p27Kip1 binds to cyclin D either alone, or when complexed to its catalytic subunit CDK4. In doing so p27Kip1 inhibits the catalytic activ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bcl-2-associated Death Promoter
The BCL2 associated agonist of cell death (BAD) protein is a pro-apoptotic member of the Bcl-2 gene family which is involved in initiating apoptosis. BAD is a member of the BH3-only family, a subfamily of the Bcl-2 family. It does not contain a C-terminal transmembrane domain for outer mitochondrial membrane and nuclear envelope targeting, unlike most other members of the Bcl-2 family. After activation, it is able to form a heterodimer with anti-apoptotic proteins and prevent them from stopping apoptosis. Mechanism of action Bax/ Bak are believed to initiate apoptosis by forming a pore in the mitochondrial outer membrane that allows cytochrome c to escape into the cytoplasm and activate the pro-apoptotic caspase cascade. The anti-apoptotic Bcl-2 and Bcl-xL proteins inhibit cytochrome c release through the mitochondrial pore and also inhibit activation of the cytoplasmic caspase cascade by cytochrome c. Dephosphorylated BAD forms a heterodimer with Bcl-2 and Bcl-xL, inactivati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
K-Ras
''KRAS'' (Kirsten rat sarcoma virus) is a gene that provides instructions for making a protein called K-Ras, a part of the RAS/MAPK pathway. The protein relays signals from outside the cell to the cell's nucleus. These signals instruct the cell to grow and divide ( proliferate) or to mature and take on specialized functions ( differentiate). It is called ''KRAS'' because it was first identified as a viral oncogene in the Kirsten RAt Sarcoma virus. The oncogene identified was derived from a cellular genome, so , when found in a cellular genome, is called a proto-oncogene. The K-Ras protein is a GTPase, a class of enzymes which convert the nucleotide guanosine triphosphate (GTP) into guanosine diphosphate (GDP). In this way the K-Ras protein acts like a switch that is turned on and off by the GTP and GDP molecules. To transmit signals, it must be turned on by attaching (binding) to a molecule of GTP. The K-Ras protein is turned off (inactivated) when it converts the GTP to GDP. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
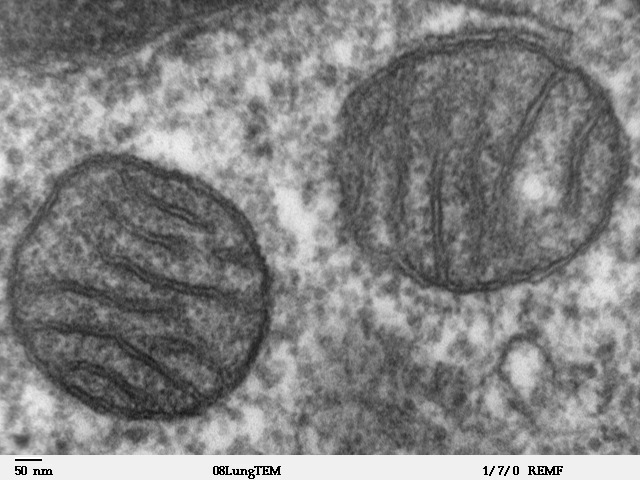
Mitochondria
A mitochondrion (; ) is an organelle found in the Cell (biology), cells of most Eukaryotes, such as animals, plants and Fungus, fungi. Mitochondria have a double lipid bilayer, membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into mitosome, other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitocho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reactive Oxygen Species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen. The reduction of molecular oxygen () produces superoxide (), which is the precursor to most other reactive oxygen species: :O2 + e^- -> \ ^\bullet O2- Dismutation of superoxide produces hydrogen peroxide (): :2 H+ + \ ^\bullet O2^- + \ ^\bullet O2^- -> H2O2 + O2 Hydrogen peroxide in turn may be partially reduced, thus forming hydroxide ions and hydroxyl radicals (), or fully reduced to water: :H2O2 + e^- -> HO^- + \ ^\bullet OH :2 H+ + 2 e- + H2O2 -> 2 H2O In a biological context, ROS are byproducts of the normal metabolism of oxygen. ROS have roles in cell signaling and homeostasis. ROS are intrinsic to cellular functioning, and are present at low and stationary levels in normal cells. In plants, ROS are involved in metabolic processes related to photoprotection and toleran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |