|
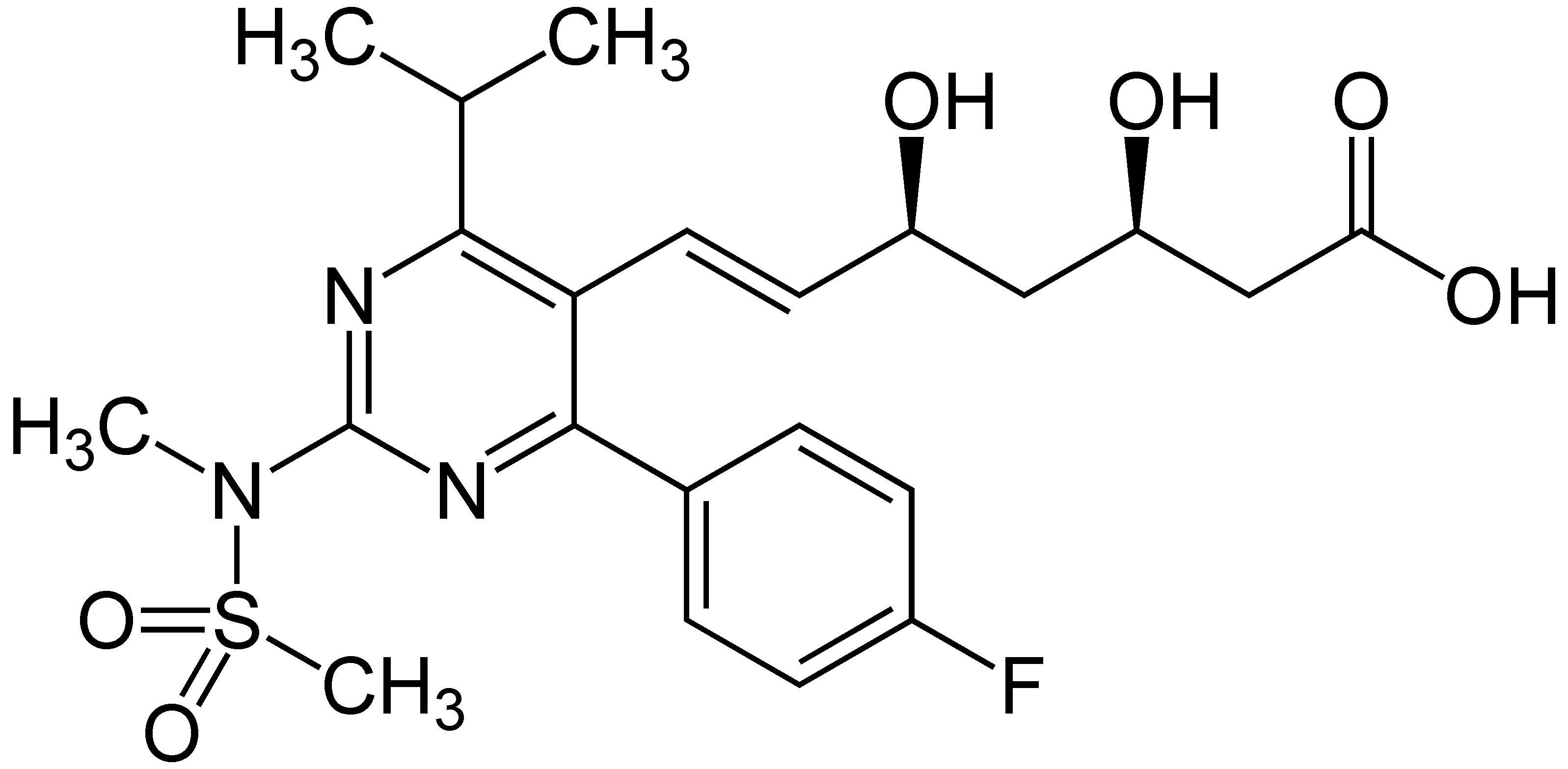
Belzutifan
Belzutifan, sold under the brand name Welireg, is an anti-cancer medication used for the treatment of von Hippel–Lindau disease-associated renal cell carcinoma. It is taken by mouth. Belzutifan is an hypoxia-inducible factor-2 alpha (HIF-2α) inhibitor. The most common side effects include decreased hemoglobin, anemia, fatigue, increased creatinine, headache, dizziness, increased glucose, and nausea. Belzutifan is the first drug to be awarded an "innovation passport" from the UK Medicines and Healthcare products Regulatory Agency (MHRA). Belzutifan was approved for medical use in the United States in August 2021. Belzutifan is the first hypoxia-inducible factor-2 alpha inhibitor therapy approved in the US. The US Food and Drug Administration (FDA) considers it to be a first-in-class medication. Medical uses Belzutifan is indicated for treatment of adults with von Hippel-Lindau (VHL) disease who require therapy for associated renal cell carcinoma (RCC), central nerv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hypoxia-inducible Factor
Hypoxia-inducible factors (HIFs) are transcription factors that respond to decreases in available oxygen in the cellular environment, or hypoxia. They are only present in parahoxozoan animals. Discovery The HIF transcriptional complex was discovered in 1995 by Gregg L. Semenza and postdoctoral fellow Guang Wang. In 2016, William Kaelin Jr., Peter J. Ratcliffe and Gregg L. Semenza were presented the Lasker Award for their work in elucidating the role of HIF-1 in oxygen sensing and its role in surviving low oxygen conditions. In 2019, the same three individuals were jointly awarded the Nobel Prize in Physiology or Medicine for their work in elucidating how HIF senses and adapts cellular response to oxygen availability. Structure Most, if not all, oxygen-breathing species express the highly conserved transcriptional complex HIF-1, which is a heterodimer composed of an alpha and a beta subunit, the latter being a constitutively-expressed aryl hydrocarbon receptor nucle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Von Hippel–Lindau Disease
Von Hippel–Lindau disease (VHL), also known as Von Hippel–Lindau syndrome, is a rare genetic disorder with multisystem involvement. It is characterized by visceral cysts and benign tumors with potential for subsequent malignant transformation. It is a type of phakomatosis that results from a mutation in the Von Hippel–Lindau tumor suppressor gene on chromosome 3p25.3. Signs and symptoms Signs and symptoms associated with VHL disease include headaches, problems with balance and walking, dizziness, weakness of the limbs, vision problems, and high blood pressure. Conditions associated with VHL disease include angiomatosis, hemangioblastomas, pheochromocytoma, renal cell carcinoma, pancreatic cysts (pancreatic serous cystadenoma), endolymphatic sac tumor, and bilateral papillary cystadenomas of the epididymis (men) or broad ligament of the uterus (women). Angiomatosis occurs in 37.2% of patients presenting with VHL disease and usually occurs in the retina. As a result, loss ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pacak–Zhuang Syndrome
The Pacak-Zhuang syndrome is a recently described disease manifestation in females that includes multiple paragangliomas or pheochromocytomas and somatostatinomas (in some), both neuroendocrine tumors, and secondary polycythemia associated with high erythropoietin levels. Paragangliomas in these patients are mainly localized to the abdomen whereas somatostatinomas are found in the second portion of the duodenum, as shown by imaging or biochemistry.Pacak K, Jochmanova I, Prodanov T, et al: New syndrome of paraganglioma and somatostatinoma associated with polycythemia. J Clin Oncol, 31(13):1690-1698, 2013. This syndrome is of special interest as finding more than one type of neuroendocrine tumor in one individual is unusual. Such co-occurrences are usually seen in patients carrying hereditary syndromes like multiple endocrine neoplasia (MEN), neurofibromatosis 1 (NF1), or von Hippel-Lindau (VHL) disease. Genetic pathway Mutations in the genes encoding alpha subunits of hypo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oral Administration
Oral administration is a route of administration where a substance is taken through the mouth. Per os abbreviated to P.O. is sometimes used as a direction for medication to be taken orally. Many medications are taken orally because they are intended to have a systemic effect, reaching different parts of the body via the bloodstream, for example. Oral administration can be easier and less painful than other routes, such as injection. However, the onset of action is relatively low, and the effectiveness is reduced if it is not absorbed properly in the digestive system, or if it is broken down by digestive enzymes before it can reach the bloodstream. Some medications may cause gastrointestinal side effects, such as nausea or vomiting, when taken orally. Oral administration can also only be applied to conscious patients, and patients willing and able to swallow. Terminology ''Per os'' (; ''P.O.'') is an adverbial phrase meaning literally from Latin "through the mouth" or "by mouth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfonates
In organosulfur chemistry, a sulfonate is a salt or ester of a sulfonic acid. It contains the functional group , where R is an organic group. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non-oxidizing, and colorless. Many useful compounds and even some biochemicals feature sulfonates. Sulfonate salts Anions with the general formula are called sulfonates. They are the conjugate bases of sulfonic acids with formula . As sulfonic acids tend to be strong acids, the corresponding sulfonates are weak bases. Due to the stability of sulfonate anions, the cations of sulfonate salts such as scandium triflate have application as Lewis acids. A classic preparation of sulfonates is the Strecker sulfite alkylation, in which an alkali sulfite salt displaces a halide, typically in the presence of an iodine catalyst: :RX + M2SO3 -> RSO3M + MX An alternative is the condensation of a sulfonyl halide with an alcohol in pyridine: :ROH + R' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drugs
An orphan drug is a pharmaceutical agent developed to treat medical conditions which, because they are so rare, would not be profitable to produce without government assistance. The conditions are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy in many countries and has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug research and development. In the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to encourage development of drugs which would otherwise lack sufficient profit motive to attract corporate research budgets and personnel. Definition According to the US Food and Drug Administration (FDA), an orphan drug is defined as one "intended for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Breakthrough Therapy
Breakthrough therapy is a United States Food and Drug Administration designation that expedites drug development that was created by Congress under Section 902 of the 9 July 2012 Food and Drug Administration Safety and Innovation Act. The FDA's "breakthrough therapy" designation is not intended to imply that a drug is actually a "breakthrough" or that there is high-quality evidence of treatment efficacy for a particular condition; rather, it allows the FDA to grant priority review to drug candidates if preliminary clinical trials indicate that the therapy may offer substantial treatment advantages over existing options for patients with serious or life-threatening diseases. The FDA has other mechanisms for expediting the review and approval process for promising drugs, including fast track designation, accelerated approval, and priority review. Requirements A breakthrough therapy designation can be assigned to a drug if "it is a drug which is intended alone or in combination with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indication (medicine)
In medicine, an indication is a valid reason to use a certain test, medication, procedure, or surgery. There can be multiple indications to use a procedure or medication. An indication can commonly be confused with the term diagnosis. A diagnosis is the assessment that a particular edicalcondition is present while an indication is a reason for use. The opposite of an indication is a contraindication, a reason to withhold a certain medical treatment because the risks of treatment clearly outweigh the benefits. In the United States, indications for prescription drugs are approved by the FDA. Indications are included in the Indications and Usage section of the Prescribing Information. The primary role of this section of labeling is to enable health care practitioners to readily identify appropriate therapies for patients by clearly communicating the drug’s approved indication(s). The Indications and Usage section states the disease or condition, or manifestation or symptoms thereof ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drug
An orphan drug is a pharmaceutical agent developed to treat medical conditions which, because they are so rare, would not be profitable to produce without government assistance. The conditions are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy in many countries and has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug research and development. In the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to encourage development of drugs which would otherwise lack sufficient profit motive to attract corporate research budgets and personnel. Definition According to the US Food and Drug Administration (FDA), an orphan drug is defined as one "intended for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |