|
Aromatic Amino Acids
An aromatic amino acid is an amino acid that includes an aromaticity, aromatic ring. Among the 20 standard amino acids, the following are classically considered aromatic: phenylalanine, tryptophan and tyrosine. Although histidine contains an aromatic ring, its basic properties cause it to be predominantly classified as a polar amino acid. Chemical structure and properties Aromatic amino acids absorb Ultraviolet, ultraviolet light at a wavelength above 250 nm and produce fluorescence. This characteristic is used in quantitative analysis, notably in determining the concentrations of these amino acids in solution. This achieved through the utilization of a Ultraviolet–visible spectroscopy, UV spectrophotomer and the Beer–Lambert law, Beer-Lambert Law equation. Most proteins will have an Absorption spectroscopy, absorption maximum at 280 nm due to the presence of aromatic amino acids in their primary structure. However, because several aromatic amino acids exist, this method h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrimidine
Pyrimidine (; ) is an aromatic, heterocyclic, organic compound similar to pyridine (). One of the three diazines (six-membered heterocyclics with two nitrogen atoms in the ring), it has nitrogen atoms at positions 1 and 3 in the ring. The other diazines are pyrazine (nitrogen atoms at the 1 and 4 positions) and pyridazine (nitrogen atoms at the 1 and 2 positions). In nucleic acids, three types of nucleobases are pyrimidine derivatives: cytosine (C), thymine (T), and uracil (U). Occurrence and history The pyrimidine ring system has wide occurrence in nature as substituted and ring fused compounds and derivatives, including the nucleotides cytosine, thymine and uracil, thiamine (vitamin B1) and alloxan. It is also found in many synthetic compounds such as barbiturates and the HIV drug, zidovudine. Although pyrimidine derivatives such as alloxan were known in the early 19th century, a laboratory synthesis of a pyrimidine was not carried out until 1879, when Grimaux reported ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
L-DOPA
-DOPA, also known as levodopa and -3,4-dihydroxyphenylalanine, is an amino acid that is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize -DOPA, make it via biosynthesis from the amino acid -tyrosine. -DOPA is the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), which are collectively known as catecholamines. Furthermore, -DOPA itself mediates neurotrophic factor release by the brain and CNS. -DOPA can be manufactured and in its pure form is sold as a psychoactive drug with the INN levodopa; trade names include Sinemet, Pharmacopa, Atamet, and Stalevo. As a drug, it is used in the clinical treatment of Parkinson's disease and dopamine-responsive dystonia. -DOPA has a counterpart with opposite chirality, -DOPA. As is true for many molecules, the human body produces only one of these isomers (the -DOPA form). T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek ''tyrós'', meaning ''cheese'', as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is encoded by the codons UAC and UAU in messenger RNA. Functions Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases. Phosphorylation of the hydroxyl group can change the activity of the targ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
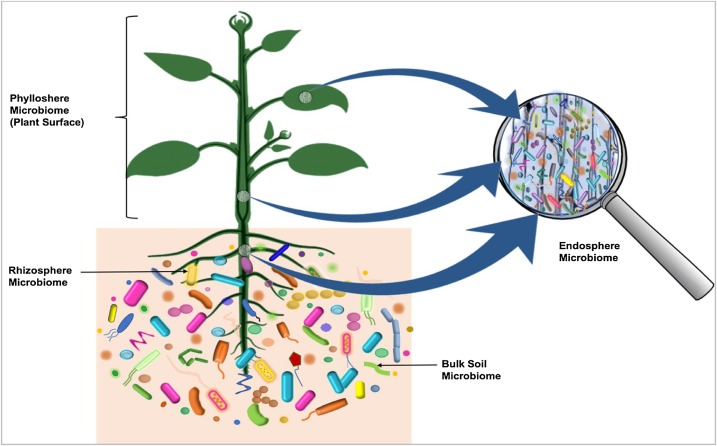
Microbiota
Microbiota are the range of microorganisms that may be commensal, symbiotic, or pathogenic found in and on all multicellular organisms, including plants. Microbiota include bacteria, archaea, protists, fungi, and viruses, and have been found to be crucial for immunologic, hormonal, and metabolic homeostasis of their host. The term ''microbiome'' describes either the collective genomes of the microbes that reside in an ecological niche or within the microbes themselves. The microbiome and host emerged during evolution as a synergistic unit from epigenetics and genetic characteristics, sometimes collectively referred to as a holobiont. The presence of microbiota in human and other metazoan guts has been critical for understanding the co-evolution between metazoans and bacteria. Microbiota play key roles in the intestinal immune and metabolic responses via their fermentation product ( short-chain fatty acid), acetate. Introduction All plants and animals, from simple l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glyphosate
Glyphosate (IUPAC name: ''N''-(phosphonomethyl)glycine) is a broad-spectrum systemic herbicide and crop desiccant. It is an organophosphorus compound, specifically a phosphonate, which acts by inhibiting the plant enzyme 5-enolpyruvylshikimate-3-phosphate synthase. It is used to kill weeds, especially annual broadleaf weeds and grasses that compete with crops. Its herbicidal effectiveness was discovered by Monsanto chemist John E. Franz in 1970. Monsanto brought it to market for agricultural use in 1974 under the trade name Roundup. Monsanto's last commercially relevant United States patent expired in 2000. Farmers quickly adopted glyphosate for agricultural weed control, especially after Monsanto introduced glyphosate-resistant Roundup Ready crops, enabling farmers to kill weeds without killing their crops. In 2007, glyphosate was the most used herbicide in the United States' agricultural sector and the second-most used (after 2,4-D) in home and garden, government and i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
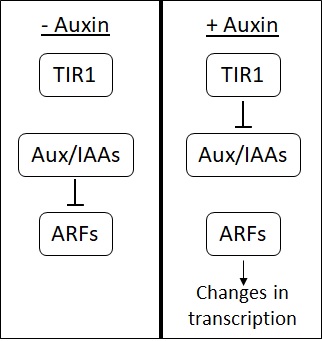
Auxin
Auxins (plural of auxin ) are a class of plant hormones (or plant-growth regulators) with some morphogen-like characteristics. Auxins play a cardinal role in coordination of many growth and behavioral processes in plant life cycles and are essential for plant body development. The Dutch biologist Frits Warmolt Went first described auxins and their role in plant growth in the 1920s. Kenneth V. Thimann became the first to isolate one of these phytohormones and to determine its chemical structure as indole-3-acetic acid (IAA). Went and Thimann co-authored a book on plant hormones, ''Phytohormones'', in 1937. Overview Auxins were the first of the major plant hormones to be discovered. They derive their name from the Greek word αυξειν (''auxein'' – "to grow/increase"). Auxin is present in all parts of a plant, although in very different concentrations. The concentration in each position is crucial developmental information, so it is subject to tight regulation through both ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salicylate
Salicylic acid is an organic compound with the formula HOC6H4CO2H. A colorless, bitter-tasting solid, it is a precursor to and a metabolite of aspirin (acetylsalicylic acid). It is a plant hormone, and has been listed by the EPA Toxic Substances Control Act (TSCA) Chemical Substance Inventory as an experimental teratogen. The name is from Latin ''salix'' for willow tree. It is an ingredient in some anti-acne products. Salts and esters of salicylic acid are known as salicylates. Uses Medicine Salicylic acid as a medication is commonly used to remove the outer layer of the skin. As such, it is used to treat warts, psoriasis, acne vulgaris, ringworm, dandruff, and ichthyosis. Similar to other hydroxy acids, salicylic acid is an ingredient in many skincare products for the treatment of seborrhoeic dermatitis, acne, psoriasis, calluses, corns, keratosis pilaris, acanthosis nigricans, ichthyosis, and warts. Uses in manufacturing Salicylic acid is used as a food preser ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Secondary Metabolite
Secondary metabolites, also called specialised metabolites, toxins, secondary products, or natural products, are organic compounds produced by any lifeform, e.g. bacteria, fungi, animals, or plants, which are not directly involved in the normal growth, development, or reproduction of the organism. Instead, they generally mediate ecological interactions, which may produce a selective advantage for the organism by increasing its survivability or fecundity. Specific secondary metabolites are often restricted to a narrow set of species within a phylogenetic group. Secondary metabolites often play an important role in plant defense against herbivory and other interspecies defenses. Humans use secondary metabolites as medicines, flavourings, pigments, and recreational drugs. The term secondary metabolite was first coined by Albrecht Kossel, a 1910 Nobel Prize laureate for medicine and physiology in 1910. 30 years later a Polish botanist Friedrich Czapek described secondary ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chorismate
Chorismic acid, more commonly known as its anionic form chorismate, is an important biochemical intermediate in plants and microorganisms. It is a precursor for: * The aromatic amino acids phenylalanine, tryptophan, and tyrosine * Indole, indole derivatives and tryptophan * 2,3-Dihydroxybenzoic acid (DHB) used for enterobactin biosynthesis * The plant hormone salicylic acid * Many alkaloids and other aromatic metabolites. *The folate precursor ''para''-aminobenzoate (pABA) * The biosynthesis of Vitamin K and folate in plants and microorganisms. The name chorismic acid derives from a classical Greek word meaning "to separate", because the compound plays a role as a branch-point in aromatic amino acid biosynthesis.{{Cite journal , last1 = Gibson , first1 = F. , title = The elusive branch-point compound of aromatic amino acid biosynthesis , doi = 10.1016/S0968-0004(98)01330-9 , journal = Trends in Biochemical Sciences , volume = 24 , issue = 1 , pages = 36–38 , year = 199 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
L-Histidine
Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the deprotonated –COO− form under biological conditions), and an imidazole side chain (which is partially protonated), classifying it as a positively charged amino acid at physiological pH. Initially thought essential only for infants, it has now been shown in longer-term studies to be essential for adults also. It is encoded by the codons CAU and CAC. Histidine was first isolated by Albrecht Kossel and Sven Gustaf Hedin in 1896. It is also a precursor to histamine, a vital inflammatory agent in immune responses. The acyl radical is histidyl. Properties of the imidazole side chain The conjugate acid (protonated form) of the imidazole side chain in histidine has a p''K''a of approximately 6.0. Thus, below a pH of 6, the imidazo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |