|
Approved Drugs
An approved drug is a medicinal preparation that has been validated for a therapeutic use by a ruling authority of a government. This process is usually specific by country, unless specified otherwise. Process by country United States In the United States, the FDA approves drugs. Before a drug can be prescribed, it must undergo the FDA's approval process. While a drug can feasibly be used off-label (for non-approved indications), it still is required to be approved for a specific disease or medical condition. Drug companies seeking to sell a drug in the United States must first test it. The company then sends the Food and Drug Administration's Center for Drug Evaluation and Research (CDER) evidence from these tests to prove the drug is safe and effective for its intended use. A fee is required to make such FDA submission. For financial year 2020, this fee was: for an application requiring clinical data ($2,942,965) and for an application not requiring clinical data ($1,471,483 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Medicine
Medicine is the science and practice of caring for a patient, managing the diagnosis, prognosis, prevention, treatment, palliation of their injury or disease, and promoting their health. Medicine encompasses a variety of health care practices evolved to maintain and restore health by the prevention and treatment of illness. Contemporary medicine applies biomedical sciences, biomedical research, genetics, and medical technology to diagnose, treat, and prevent injury and disease, typically through pharmaceuticals or surgery, but also through therapies as diverse as psychotherapy, external splints and traction, medical devices, biologics, and ionizing radiation, amongst others. Medicine has been practiced since prehistoric times, and for most of this time it was an art (an area of skill and knowledge), frequently having connections to the religious and philosophical beliefs of local culture. For example, a medicine man would apply herbs and say prayers for healing, o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Preclinical Development
In drug development, preclinical development, also termed preclinical studies or nonclinical studies, is a stage of research that begins before clinical trials (testing in humans) and during which important feasibility, iterative testing and drug safety data are collected, typically in laboratory animals. The main goals of preclinical studies are to determine a starting, safe dose for first-in-human study and assess potential toxicity of the product, which typically include new medical devices, prescription drugs, and diagnostics. Companies use stylized statistics to illustrate the risks in preclinical research, such as that on average, only one in every 5,000 compounds that enters drug discovery to the stage of preclinical development becomes an approved drug. Types of preclinical research Each class of product may undergo different types of preclinical research. For instance, drugs may undergo pharmacodynamics (what the drug does to the body) (PD), pharmacokinetics (wha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clinical Research
Clinical research is a branch of healthcare science that determines the safety and effectiveness ( efficacy) of medications, devices, diagnostic products and treatment regimens intended for human use. These may be used for prevention, treatment, diagnosis or for relieving symptoms of a disease. Clinical research is different from clinical practice. In clinical practice established treatments are used, while in clinical research evidence is collected to establish a treatment. Overview The term "clinical research" refers to the entire bibliography of a drug/device/biologic, in fact any test article from its inception in the lab to its introduction to the consumer market and beyond. Once the promising candidate or the molecule is identified in the lab, it is subjected to pre-clinical studies or animal studies where different aspects of the test article (including its safety toxicity if applicable and efficacy, if possible at this early stage) are studied. In the United States ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Patent Medicine
A patent medicine, sometimes called a proprietary medicine, is an over-the-counter (nonprescription) medicine or medicinal preparation that is typically protected and advertised by a trademark and trade name (and sometimes a patent) and claimed to be effective against minor disorders and symptoms. Its contents are typically incompletely disclosed. Antiseptics, analgesics, some sedatives, laxatives, and antacids, cold and cough medicines, and various skin preparations are included in the group. The safety and effectiveness of patent medicines and their sale is controlled and regulated by the Food and Drug Administration in the United States and corresponding authorities in other countries.https://www.merriam-webster.com/dictionary/patent%20medicine The term is sometimes still used to describe quack remedies of unproven effectiveness and questionable safety sold especially by peddlers in past centuries, who often also called them elixirs, tonics, or liniments. Current examples o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Abbreviated New Drug Application
An Abbreviated New Drug Application (ANDA) is an application for a U.S. generic drug approval for an existing licensed medication or approved drug. The ANDA is submitted to Food and Drug Administration, FDA's Center for Drug Evaluation and Research, Office of Generic Drugs, which provides for the review and ultimate approval of a generic drug product. Once approved, an applicant may manufacture and market the generic drug product to provide a safe, effective, low cost alternative to the American public. Electronic submissions of ANDAs have grown by 70% since November 2008. The Section IV challenge has been credited with suppressing new drug innovation. A generic drug product is one that is comparable to a patented drug product in dosage form, strength, route of administration, quality, performance characteristics and intended use. All approved products, both innovator and generic, are listed in FDA's Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
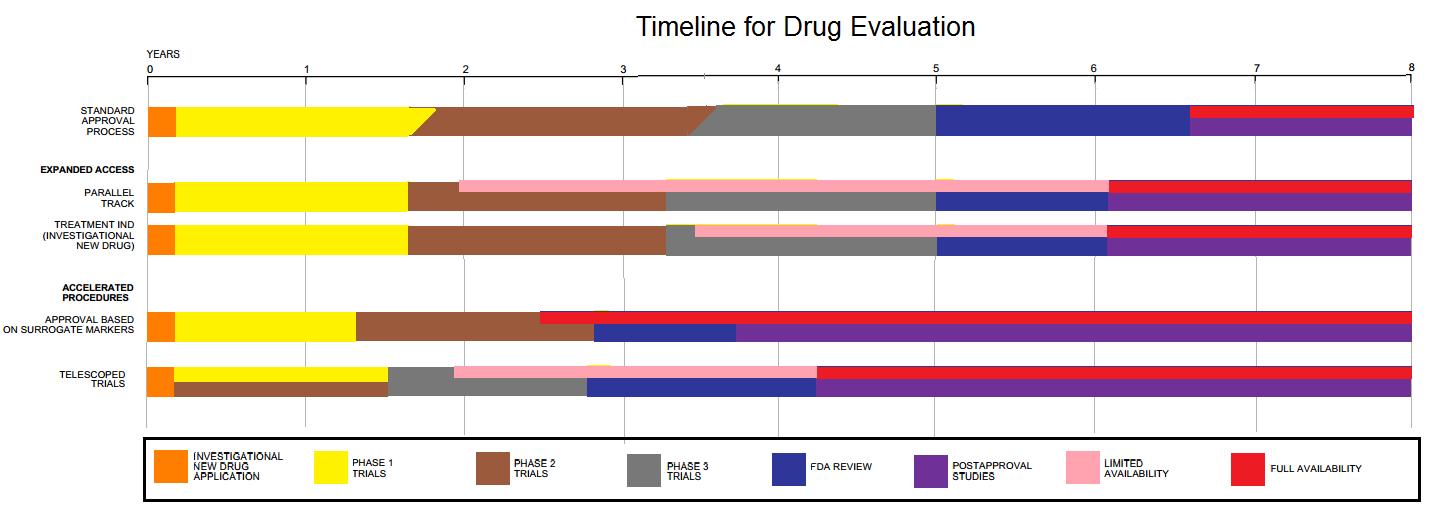
Drug Development
Drug development is the process of bringing a new pharmaceutical drug to the market once a lead compound has been identified through the process of drug discovery. It includes preclinical research on microorganisms and animals, filing for regulatory status, such as via the United States Food and Drug Administration for an investigational new drug to initiate clinical trials on humans, and may include the step of obtaining regulatory approval with a new drug application to market the drug. The entire process – from concept through preclinical testing in the laboratory to clinical trial development, including Phase I–III trials – to approved vaccine or drug typically takes more than a decade. New chemical entity development Broadly, the process of drug development can be divided into preclinical and clinical work. Pre-clinical New chemical entities (NCEs, also known as new molecular entities or NMEs) are compounds that emerge from the process of drug discovery. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Drug Design
Drug design, often referred to as rational drug design or simply rational design, is the inventive process of finding new medications based on the knowledge of a biological target. The drug is most commonly an organic small molecule that activates or inhibits the function of a biomolecule such as a protein, which in turn results in a therapeutic benefit to the patient. In the most basic sense, drug design involves the design of molecules that are complementary in shape and charge to the biomolecular target with which they interact and therefore will bind to it. Drug design frequently but not necessarily relies on computer modeling techniques. This type of modeling is sometimes referred to as computer-aided drug design. Finally, drug design that relies on the knowledge of the three-dimensional structure of the biomolecular target is known as structure-based drug design. In addition to small molecules, biopharmaceuticals including peptides and especially therapeutic antibodies a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Drug Discovery
In the fields of medicine, biotechnology and pharmacology, drug discovery is the process by which new candidate medications are discovered. Historically, drugs were discovered by identifying the active ingredient from traditional remedies or by serendipitous discovery, as with penicillin. More recently, chemical libraries of synthetic small molecules, natural products or extracts were screened in intact cells or whole organisms to identify substances that had a desirable therapeutic effect in a process known as classical pharmacology. After sequencing of the human genome allowed rapid cloning and synthesis of large quantities of purified proteins, it has become common practice to use high throughput screening of large compounds libraries against isolated biological targets which are hypothesized to be disease-modifying in a process known as reverse pharmacology. Hits from these screens are then tested in cells and then in animals for efficacy. Modern drug discovery involves the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
The New York Times
''The New York Times'' (''the Times'', ''NYT'', or the Gray Lady) is a daily newspaper based in New York City with a worldwide readership reported in 2020 to comprise a declining 840,000 paid print subscribers, and a growing 6 million paid digital subscribers. It also is a producer of popular podcasts such as '' The Daily''. Founded in 1851 by Henry Jarvis Raymond and George Jones, it was initially published by Raymond, Jones & Company. The ''Times'' has won 132 Pulitzer Prizes, the most of any newspaper, and has long been regarded as a national " newspaper of record". For print it is ranked 18th in the world by circulation and 3rd in the U.S. The paper is owned by the New York Times Company, which is publicly traded. It has been governed by the Sulzberger family since 1896, through a dual-class share structure after its shares became publicly traded. A. G. Sulzberger, the paper's publisher and the company's chairman, is the fifth generation of the family to head the pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clinical Trials
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, dietary choices, dietary supplements, and medical devices) and known interventions that warrant further study and comparison. Clinical trials generate data on dosage, safety and efficacy. They are conducted only after they have received health authority/ethics committee approval in the country where approval of the therapy is sought. These authorities are responsible for vetting the risk/benefit ratio of the trial—their approval does not mean the therapy is 'safe' or effective, only that the trial may be conducted. Depending on product type and development stage, investigators initially enroll volunteers or patients into small pilot studies, and subsequently conduct progressively larger scale comparative studies. Clinical trials can vary i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead Compound
A lead compound (, i.e. a "leading" compound, not to be confused with various compounds of the metallic element lead) in drug discovery is a chemical compound that has pharmacological or biological activity likely to be therapeutically useful, but may nevertheless have suboptimal structure that requires modification to fit better to the target; lead drugs offer the prospect of being followed by back-up compounds. Its chemical structure serves as a starting point for chemical modifications in order to improve potency, selectivity, or pharmacokinetic parameters. Furthermore, newly invented pharmacologically active moieties may have poor druglikeness and may require chemical modification to become drug-like enough to be tested biologically or clinically. Terminology Lead compounds are sometimes called developmental candidates. This is because the discovery and selection of lead compounds occurs prior to preclinical and clinical development of the candidate. Discovering lead compo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Regulation Of Therapeutic Goods
The regulation of therapeutic goods, defined as drugs and therapeutic devices, varies by jurisdiction. In some countries, such as the United States, they are regulated at the national level by a single agency. In other jurisdictions they are regulated at the state level, or at both state and national levels by various bodies, as in Australia. The role of therapeutic goods regulation is designed mainly to protect the health and safety of the population. Regulation is aimed at ensuring the safety, quality, and efficacy of the therapeutic goods which are covered under the scope of the regulation. In most jurisdictions, therapeutic goods must be registered before they are allowed to be sold. There is usually some degree of restriction on the availability of certain therapeutic goods, depending on their risk to consumers. History Modern drug regulation has historical roots in the response to the proliferation of universal antidotes which appeared in the wake of Mithridates' death. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



.png)
.jpg)