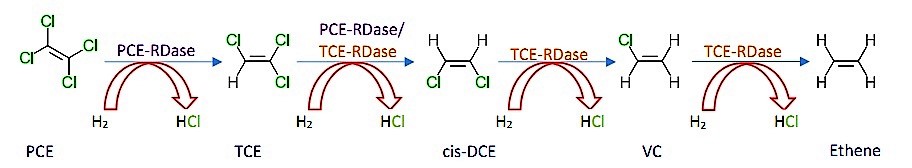
|
Adsorbable Organic Halides
Adsorbable organic halides (AOX) is a measure of the organic halogen load at a sampling site such as soil from a land fill, water, or sewage waste. The procedure measures chlorine, bromine, and iodine as equivalent halogens, but does not measure fluorine levels in the sample. Background Utilization of halogen containing materials in processes such as water treatment, bleaching, or even general synthesis to create the final product, generates a number of organic halides. These organic halides are released in wastewater from the oil, chemical, and paper industries, and find their way to the consumer and eventually to a landfill or oceanic dumps. Within the soil, the halo compounds resist degradation and often react with metal ions, resulting in non-degradable metal complexes, increasing soil toxicity and accumulating in the food chain of aquatic organisms. Up to 2000 ppm of these bio-accumulative organic chlorides were detected in fat of fish from the waters where bleaching effluen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature. It is an extremely reactive element and a strong oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Electronegativity#Pauling electronegativity, Pauling scale, behind only oxygen and fluorine. Chlorine played an important role in the experiments conducted by medieval Alchemy, alchemists, which commonly involved the heating of chloride Salt (chemistry), salts like ammonium chloride (sal ammoniac) and sodium chloride (common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and hydrochloric acid (in the form of ). However ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
High-performance Liquid Chromatography
High-performance liquid chromatography (HPLC), formerly referred to as high-pressure liquid chromatography, is a technique in analytical chemistry used to separate, identify, and quantify each component in a mixture. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out of the column. HPLC has been used for manufacturing (''e.g.'', during the production process of pharmaceutical and biological products), legal (''e.g.'', detecting performance enhancement drugs in urine), research (''e.g.'', separating the components of a complex biological sample, or of similar synthetic chemicals from each other), and medical (''e.g.'', detecting vitamin D levels in blood serum) purposes. Chrom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Desulfuromonas
''Desulfuromonas'' is a Gram negative bacterial genus from the family of Desulfuromonadaceae. ''Desulfuromonas'' can reduce elemental sulfur to H2S. ''Desulfuromonas'' occur in anoxic sediments and saline lake A salt lake or saline lake is a landlocked body of water that has a concentration of salts (typically sodium chloride) and other dissolved minerals significantly higher than most lakes (often defined as at least three grams of salt per litre). ...s. References Further reading * * * * * External links MicrobeWiki Desulfuromonadales Bacteria genera {{Thermodesulfobacteriota-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehalobacter
''Dehalobacter'' is a genus in the phylum Bacillota (Bacteria). Etymology The generic name ''Dehalobacter'' derives from Latin ''de'', from; ''halogenum'' from Swedish, coined by Swedish chemist Baron Jöns Jakob Berzelius (1779–1848) from Greek ''hals'', ''halos'' "salt" + ''gen'' "to produce", so called because a salt is formed in reactions involving these elements; a rod '' bacter'', nominally meaning "a rod", but in effect meaning a bacterium, a rod; giving ''Dehalobacter'', a halogen-removing, rod-shaped bacterium. Species The genus contains a single species, namely '' D. restrictus'' ( Holliger ''et al''. 1998, type species of the genus. The specific name is from Latin ''restrictus'', limited, restricted, confined, referring to the limited substrate range used. Recently, ''Dehalobacter'' sp. UNSWDHB, which dechlorinate chloroform to dichloromethane, and its reductive dehalogenase were identified. See also * Bacterial taxonomy * Microbiology * List of bacterial ord ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Desulfomonile
''Desulfomonile'' is a Gram negative, strict anaerobe An anaerobic organism or anaerobe is any organism that does not require molecular oxygen for growth. It may react negatively or even die if free oxygen is present. In contrast, an aerobic organism (aerobe) is an organism that requires an oxygenate ... and non-motile bacterial genus from the family of Syntrophaceae. ''Desulfomonile'' bacteria can reduce sulfur oxyanions to H2S. References Further reading * * * Bacteria genera Thermodesulfobacteriota {{Bacteria-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds). Ethylene is widely used in the chemical industry, and its worldwide production (over 150 million tonnes in 2016) exceeds that of any other organic compound. Much of this production goes toward polyethylene, a widely used plastic containing polymer chains of ethylene units in various chain lengths. Ethylene is also an important natural plant hormone and is used in agriculture to force the ripening of fruits. The hydrate of ethylene is ethanol. Structure and properties This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a double bond. All six atoms that comprise ethylene are coplanar. The H-C-H angle is 117.4°, close to the 120° for ideal sp² hybridized carbon. The molecule is also relatively weak: rota ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vinyl Chloride
Vinyl chloride is an organochloride with the formula H2C=CHCl. It is also called vinyl chloride monomer (VCM) or chloroethene. This colorless compound is an important industrial chemical chiefly used to produce the polymer polyvinyl chloride (PVC). About 13 million metric tonne are produced annually. VCM is among the top twenty largest petrochemicals (petroleum-derived chemicals) in world production. The United States currently remains the largest VCM manufacturing region because of its low-production-cost position in chlorine and ethylene raw materials. China is also a large manufacturer and one of the largest consumers of VCM. Vinyl chloride is a gas with a sweet odor. It is highly toxic, flammable, and carcinogenic. It can be formed in the environment when soil organisms break down chlorinated solvents. Vinyl chloride that is released by industries or formed by the breakdown of other chlorinated chemicals can enter the air and drinking water supplies. Vinyl chloride is a common ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2-Dichloroethene
1,2-Dichloroethene, commonly called 1,2-dichloroethylene or 1,2-DCE, is the name for a pair of organochlorine compounds with the molecular formula CHCl. They are both colorless liquids with a sweet odor. It can exist as either of two geometric isomers, ''cis''-1,2-dichloroethene or ''trans''-1,2-dichloroethene, but is often used as a mixture of the two. They have modest solubility in water. These compounds have some applications as a degreasing solvent. In contrast to most ''cis-trans'' compounds, the ''Z'' isomer (''cis'') is more stable than the ''E'' isomer (''trans'') by 0.4 kcal/mol. Production and use ''cis''-DCE, the ''Z'' isomer, is obtainable by the controlled chlorination of acetylene: :CH + Cl → CHCl Industrially both isomers arise as byproducts of the production of vinyl chloride, which is produced on a vast scale. Unlike vinyl chloride, the 1,2-dichloroethylene isomers do not polymerize. ''trans''-DCE has applications including electronics cleaning, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanogen
Methanogens are microorganisms that produce methane as a metabolic byproduct in hypoxic conditions. They are prokaryotic and belong to the domain Archaea. All known methanogens are members of the archaeal phylum Euryarchaeota. Methanogens are common in wetlands, where they are responsible for marsh gas, and in the digestive tracts of animals such as ruminants and many humans, where they are responsible for the methane content of belching in ruminants and flatulence in humans. In marine sediments, the biological production of methane, also termed methanogenesis, is generally confined to where sulfates are depleted, below the top layers. Moreover, methanogenic archaea populations play an indispensable role in anaerobic wastewater treatments. Others are extremophiles, found in environments such as hot springs and submarine hydrothermal vents as well as in the "solid" rock of Earth's crust, kilometers below the surface. Physical description Methanogens are coccoid (spherical shap ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and location ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehalococcoides
''Dehalococcoides'' is a genus of bacteria within class Dehalococcoidia that obtain energy via the oxidation of hydrogen and subsequent reductive dehalogenation of halogenated organic compounds in a mode of anaerobic respiration called organohalide respiration. They are well known for their great potential to remediate halogenated ethenes and aromatics. They are the only bacteria known to transform highly chlorinated dioxins, PCBs. In addition, they are the only known bacteria to transform tetrachloroethene ( perchloroethene, PCE) to ethene. Microbiology The first member of the genus ''Dehalococcoides'' was described in 1997 as ''Dehalococcoides ethenogenes'' strain 195 (nom. inval.). Additional ''Dehalococcoides'' members were later described as strains CBDB1, BAV1, FL2, VS, and GT. In 2012 all yet-isolated ''Dehalococcoides'' strains were summarized under the new taxonomic name '' D. mccartyi'', with strain 195 as the type strain. GTDB release 202 clusters the genus into thre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrachloroethylene
Tetrachloroethylene, also known under the systematic name tetrachloroethene, or perchloroethylene, and many other names (and abbreviations such as "perc" or "PERC", and "PCE"), is a chlorocarbon with the formula Cl2C=CCl2 . It is a colorless liquid widely used for dry cleaning of fabrics, hence it is sometimes called "dry-cleaning fluid". It also has its uses as an effective automotive brake cleaner. It has a sweet odor detectable by most people at a concentration of 1 part per million (1 ppm). Worldwide production was about in 1985.M. Rossberg et al. "Chlorinated Hydrocarbons" in ''Ullmann's Encyclopedia of Industrial Chemistry'', 2006, Wiley-VCH, Weinheim. Production British physicist and chemist Michael Faraday first synthesized tetrachloroethylene in 1821 by thermal decomposition of hexachloroethane. :C2Cl6 → C2Cl4 + Cl2 Most tetrachloroethylene is produced by high temperature chlorinolysis of light hydrocarbons. The method is related to Faraday's discovery since hexachl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |