|
Anodized Aluminum
Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of metal parts. The process is called ''anodizing'' because the part to be treated forms the anode electrode of an electrolytic cell. Anodizing increases resistance to corrosion and wear, and provides better adhesion for paint primers and glues than bare metal does. Anodic films can also be used for several cosmetic effects, either with thick porous coatings that can absorb dyes or with thin transparent coatings that add reflected light wave interference effects. Anodizing is also used to prevent galling of threaded components and to make dielectric films for electrolytic capacitors. Anodic films are most commonly applied to protect aluminium alloys, although processes also exist for titanium, zinc, magnesium, niobium, zirconium, hafnium, and tantalum. Iron or carbon steel metal exfoliates when oxidized under neutral or alkaline micro-electrolytic conditions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cheap Carabiners
Cheap may refer to: *Cheapness * ''Cheap'' (album), debut album from Seasick Steve *Cheap (ward), London, UK *Flatwoods, Kentucky, previously known as Cheap See also *Cheapskate A miser is a person who is reluctant to spend, sometimes to the point of forgoing even basic comforts and some necessities, in order to hoard money or other possessions. Although the word is sometimes used loosely to characterise anyone who ... * Cheep (other) {{disambig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tantalum
Tantalum is a chemical element with the symbol Ta and atomic number 73. Previously known as ''tantalium'', it is named after Tantalus, a villain in Greek mythology. Tantalum is a very hard, ductile, lustrous, blue-gray transition metal that is highly corrosion-resistant. It is part of the refractory metals group, which are widely used as components of strong high-melting-point alloys. It is a group 5 element, along with vanadium and niobium, and it always occurs in geologic sources together with the chemically similar niobium, mainly in the mineral groups tantalite, columbite and coltan. The chemical inertness and very high melting point of tantalum make it valuable for laboratory and industrial equipment such as reaction vessels and vacuum furnaces. It is used in tantalum capacitors for electronic equipment such as computers. Tantalum is considered a technology-critical element by the European Commission. History Tantalum was discovered in Sweden in 1802 by Anders Ekeberg, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
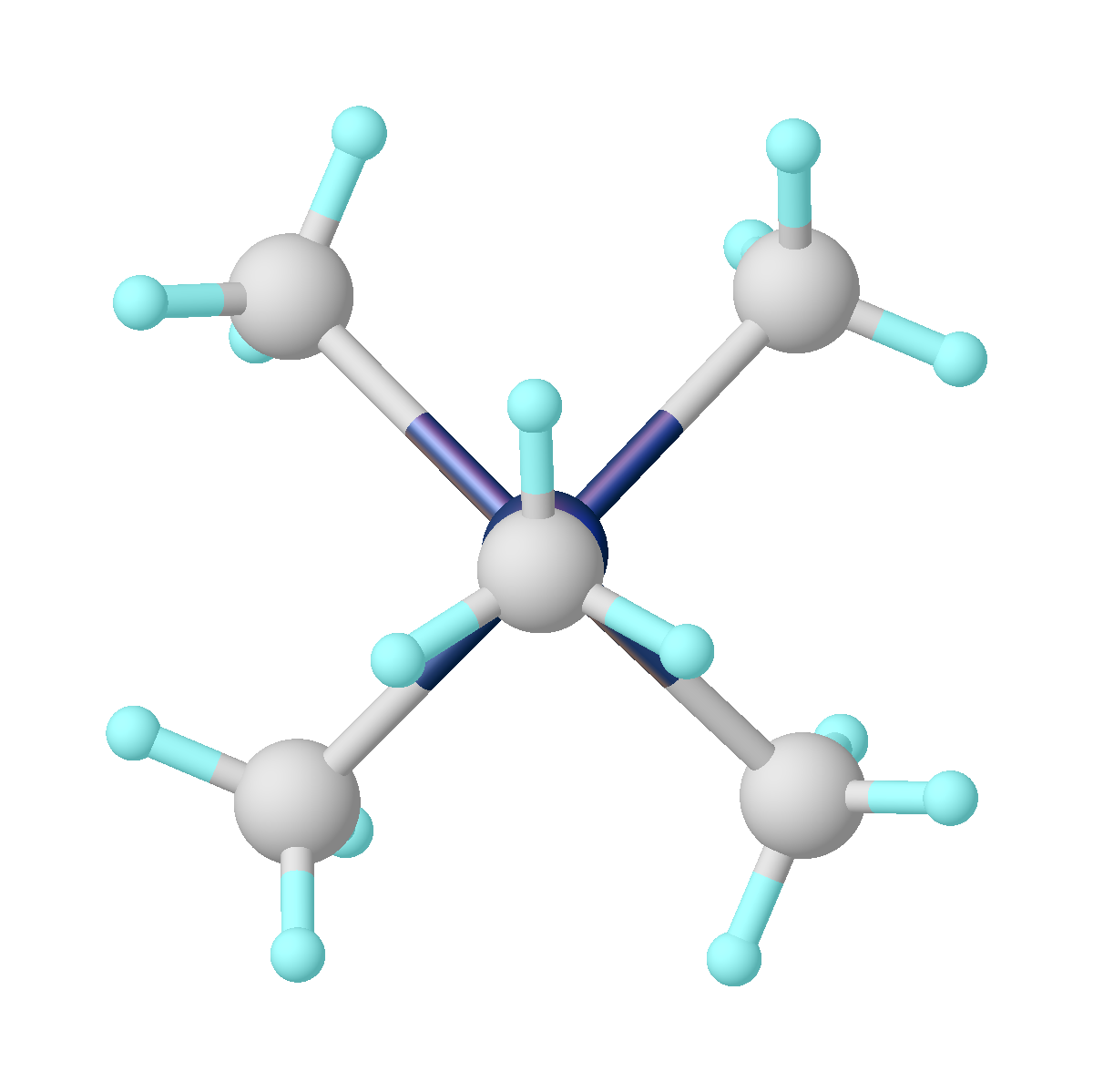
Iron(II,III) Oxide
Iron(II,III) oxide is the chemical compound with formula Fe3O4. It occurs in nature as the mineral magnetite. It is one of a number of iron oxides, the others being iron(II) oxide (FeO), which is rare, and iron(III) oxide (Fe2O3) which also occurs naturally as the mineral hematite. It contains both Fe2+ and Fe3+ ions and is sometimes formulated as FeO ∙ Fe2O3. This iron oxide is encountered in the laboratory as a black powder. It exhibits permanent magnetism and is ferrimagnetic, but is sometimes incorrectly described as ferromagnetic. Its most extensive use is as a black pigment. For this purpose, it is synthesized rather than being extracted from the naturally occurring mineral as the particle size and shape can be varied by the method of production. Preparation Heated iron metal interacts with steam to form iron oxide and hydrogen gas. 3Fe + 4H2O->Fe3O4 + 4H2 Under anaerobic conditions, ferrous hydroxide (Fe(OH)2) can be oxidized by water to form magnetite and m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Red Fuming Nitric Acid
Red fuming nitric acid (RFNA) is a storable oxidizer used as a rocket propellant. It consists of 84% nitric acid (), 13% dinitrogen tetroxide and 1–2% water. The color of red fuming nitric acid is due to the dinitrogen tetroxide, which breaks down partially to form nitrogen dioxide. The nitrogen dioxide dissolves until the liquid is saturated, and produces toxic fumes with a suffocating odor. RFNA increases the flammability of combustible materials and is highly exothermic when reacting with water. It is usually used with an inhibitor (with various, sometimes secret, substances, including hydrogen fluoride; any such combination is called ''inhibited RFNA'', ''IRFNA'') because nitric acid attacks most container materials. Hydrogen fluoride for instance will passivate the container metal with a thin layer of metal fluoride, making it nearly impervious to the nitric acid. It can also be a component of a monopropellant; with substances like amine nitrates dissolved in it, it can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitric Acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitric acid has a concentration of 68% in water. When the solution contains more than 86% , it is referred to as ''fuming nitric acid''. Depending on the amount of nitrogen dioxide present, fuming nitric acid is further characterized as red fuming nitric acid at concentrations above 86%, or white fuming nitric acid at concentrations above 95%. Nitric acid is the primary reagent used for nitration – the addition of a nitro group, typically to an organic molecule. While some resulting nitro compounds are shock- and thermally-sensitive explosives, a few are stable enough to be used in munitions and demolition, while others are still more stable and used as pigments in inks and dyes. Nitric acid is also commonly used as a strong oxidizing agen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferrous Metals
In chemistry, the adjective Ferrous indicates a compound that contains iron(II), meaning iron in its +2 oxidation state, possibly as the divalent cation Fe2+. It is opposed to "ferric" or iron(III), meaning iron in its +3 oxidation state, such as the trivalent cation Fe3+.ferrous entry in the online dictionary. Accessed on 2008-04-19. This usage has been largely replaced by the nomenclature, which calls for the oxidation state being indicated by Roman numerals in parentheses, such as |
Cast Iron
Cast iron is a class of iron–carbon alloys with a carbon content more than 2%. Its usefulness derives from its relatively low melting temperature. The alloy constituents affect its color when fractured: white cast iron has carbide impurities which allow cracks to pass straight through, grey cast iron has graphite flakes which deflect a passing crack and initiate countless new cracks as the material breaks, and ductile cast iron has spherical graphite "nodules" which stop the crack from further progressing. Carbon (C), ranging from 1.8 to 4 wt%, and silicon (Si), 1–3 wt%, are the main alloying elements of cast iron. Iron alloys with lower carbon content are known as steel. Cast iron tends to be brittle, except for malleable cast irons. With its relatively low melting point, good fluidity, castability, excellent machinability, resistance to deformation and wear resistance, cast irons have become an engineering material with a wide range of applications and are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
High-carbon Steel
Carbon steel is a steel with carbon content from about 0.05 up to 2.1 percent by weight. The definition of carbon steel from the American Iron and Steel Institute (AISI) states: * no minimum content is specified or required for chromium, cobalt, molybdenum, nickel, niobium, titanium, tungsten, vanadium, zirconium, or any other element to be added to obtain a desired alloying effect; * the specified minimum for copper does not exceed 0.40%; * or the maximum content specified for any of the following elements does not exceed the percentages noted: manganese 1.65%; silicon 0.60%; copper 0.60%. The term ''carbon steel'' may also be used in reference to steel which is not stainless steel; in this use carbon steel may include alloy steels. High carbon steel has many different uses such as milling machines, cutting tools (such as chisels) and high strength wires. These applications require a much finer microstructure, which improves the toughness. Carbon steel is a popular metal choic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloride
The chloride ion is the anion (negatively charged ion) Cl−. It is formed when the element chlorine (a halogen) gains an electron or when a compound such as hydrogen chloride is dissolved in water or other polar solvents. Chloride salts such as sodium chloride are often very soluble in water.Green, John, and Sadru Damji. "Chapter 3." ''Chemistry''. Camberwell, Vic.: IBID, 2001. Print. It is an essential electrolyte located in all body fluids responsible for maintaining acid/base balance, transmitting nerve impulses and regulating liquid flow in and out of cells. Less frequently, the word ''chloride'' may also form part of the "common" name of chemical compounds in which one or more chlorine atoms are covalently bonded. For example, methyl chloride, with the standard name chloromethane (see IUPAC books) is an organic compound with a covalent C−Cl bond in which the chlorine is not an anion. Electronic properties A chloride ion (diameter 167 pm) is much larger tha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfate
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salts of sulfuric acid and many are prepared from that acid. Spelling "Sulfate" is the spelling recommended by IUPAC, but "sulphate" was traditionally used in British English. Structure The sulfate anion consists of a central sulfur atom surrounded by four equivalent oxygen atoms in a tetrahedral arrangement. The symmetry is the same as that of methane. The sulfur atom is in the +6 oxidation state while the four oxygen atoms are each in the −2 state. The sulfate ion carries an overall charge of −2 and it is the conjugate base of the bisulfate (or hydrogensulfate) ion, , which is in turn the conjugate base of , sulfuric acid. Organic sulfate esters, such as dimethyl sulfate, are covalent compounds and esters of sulfuric acid. The tetrahedral molecular geometry of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rust
Rust is an iron oxide, a usually reddish-brown oxide formed by the reaction of iron and oxygen in the catalytic presence of water or air moisture. Rust consists of hydrous iron(III) oxides (Fe2O3·nH2O) and iron(III) oxide-hydroxide (FeO(OH), Fe(OH)3), and is typically associated with the corrosion of refined iron. Given sufficient time, any iron mass, in the presence of water and oxygen, could eventually convert entirely to rust. Surface rust is commonly flaky and friable, and provides no passivational protection to the underlying iron, unlike the formation of patina on copper surfaces. ''Rusting'' is the common term for corrosion of elemental iron and its alloys such as steel. Many other metals undergo similar corrosion, but the resulting oxides are not commonly called "rust". Several forms of rust are distinguishable both visually and by spectroscopy, and form under different circumstances. Other forms of rust include the result of reactions between iron and chloride ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrated Iron Oxide
Iron(III) oxide-hydroxide or ferric oxyhydroxideA. L. Mackay (1960): "β-Ferric Oxyhydroxide". ''Mineralogical Magazine'' (''Journal of the Mineralogical Society''), volume 32, issue 250, pages 545-557. is the chemical compound of iron, oxygen, and hydrogen with formula . The compound is often encountered as one of its hydrates, ·''n'' ust The monohydrate · is often referred to as iron(III) hydroxide ,CAS , C.I. 77492 hydrated iron oxide, yellow iron oxide, or Pigment Yellow 42. Natural occurrences Minerals Anhydrous ferric hydroxide occurs in the nature as the exceedingly rare mineral bernalite, Fe(OH)3·''n''H2O (n=0.0-0.25). Iron oxyhydroxides, , are much more common and occur naturally as structurally different minerals (polymorphs) denoted by the Greek letters α, β, γ and δ. * Goethite, α-FeO(OH), has been used as an ochre pigment since prehistoric times. * Akaganeite is the β polymorph,A. L. Mackay (1962): "β-Ferric oxyhydroxide—akaganéite", ''Mineral ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_oxide.jpg)