|
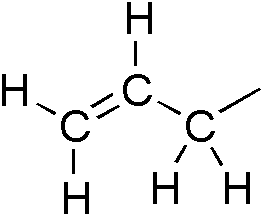
Allylbenzene
Phenylpropene is the organic compound with the formula C6H5CH2CH=CH2. It is a colorless liquid. The compound consists of a phenyl group attached to allyl. Phenylpropene isomerizes to trans-propenylbenzene. In plant biochemistry, the phenylpropene skeleton is the parent (simplest representation) of the phenylpropanoids. Prominent derivatives include eugenol, safrole Safrole is an organic compound with the formula CH2O2C6H3CH2CH=CH2. It is a colorless oily liquid, although impure samples can appear yellow. A member of the phenylpropanoid family of natural products, it is found in sassafras plants, among oth ..., and many others. References External links * {{Phenylpropene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Safrole
Safrole is an organic compound with the formula CH2O2C6H3CH2CH=CH2. It is a colorless oily liquid, although impure samples can appear yellow. A member of the phenylpropanoid family of natural products, it is found in sassafras plants, among others. Small amounts are found in a wide variety of plants, where it functions as a natural antifeedant. ''Ocotea pretiosa'', which grows in Brazil, and ''Sassafras albidum'', which grows in eastern North America, are the main natural sources of safrole. It has a characteristic "sweet-shop" aroma. It is a precursor in the synthesis of the insecticide synergist piperonyl butoxide, the fragrance piperonal via isosafrole, and the empathogenic/entactogenic substance MDMA. History Safrole was obtained from a number of plants, but especially from the sassafras tree (''Sassafras albidum''), which is native to North America, and from Japanese star anise (''Illicium anisatum'', called ''shikimi'' in Japan). In 1844, the French chemist Édouard ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trans-Propenylbenzene
''trans''-Propenylbenzene is an organic compound with the formula C6H5CH=CHCH3. It is one of two isomers of 1-propenylbenzene. Both isomers are colorless flammable liquids. It is formed by the isomerization of allylbenzene Phenylpropene is the organic compound with the formula C6H5CH2CH=CH2. It is a colorless liquid. The compound consists of a phenyl group attached to allyl. Phenylpropene isomerizes to trans-propenylbenzene. In plant biochemistry, the phenylpro .... References {{Hydrocarbons Alkene derivatives Aromatic hydrocarbons Hydrocarbons C3-Benzenes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Eugenol
Eugenol is an allyl chain-substituted guaiacol, a member of the allylbenzene class of chemical compounds. It is a colorless to pale yellow, aromatic oily liquid extracted from certain essential oils especially from clove, nutmeg, cinnamon, basil and bay leaf. It is present in concentrations of 80–90% in clove bud oil and at 82–88% in clove leaf oil. Eugenol has a pleasant, spicy, clove-like scent. The name is derived from ''Eugenia caryophyllata'', the former Linnean nomenclature term for cloves. The currently accepted name is ''Syzygium aromaticum''. Biosynthesis The biosynthesis of eugenol begins with the amino acid tyrosine. L-tyrosine is converted to ''p''-coumaric acid by the enzyme tyrosine ammonia lyase (TAL). From here, ''p''-coumaric acid is converted to caffeic acid by ''p''-coumarate 3-hydroxylase using oxygen and NADPH. ''S''-Adenosyl methionine (SAM) is then used to methylate caffeic acid, forming ferulic acid, which is in turn converted to feruloyl- CoA by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic C−H bonds are about 15% weaker than the C−H bonds in ordinary sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength, and reactivity. Other re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylpropanoid
The phenylpropanoids are a diverse family of organic compounds that are synthesized by plants from the amino acids phenylalanine and tyrosine. Their name is derived from the six-carbon, aromatic phenyl group and the three-carbon propene tail of coumaric acid, which is the central intermediate in phenylpropanoid biosynthesis. From 4-coumaroyl-CoA emanates the biosynthesis of myriad natural products including lignols (precursors to lignin and lignocellulose), flavonoids, isoflavonoids, coumarins, aurones, stilbenes, catechin, and phenylpropanoids. The coumaroyl component is produced from cinnamic acid. Phenylpropanoids are found throughout the plant kingdom, where they serve as essential components of a number of structural polymers, provide protection from ultraviolet light, defend against herbivores and pathogens, and also mediate plant-pollinator interactions as floral pigments and scent compounds. Hydroxycinnamic acids Phenylalanine is first converted to cinnamic aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |