|
Uranium–lead Dating
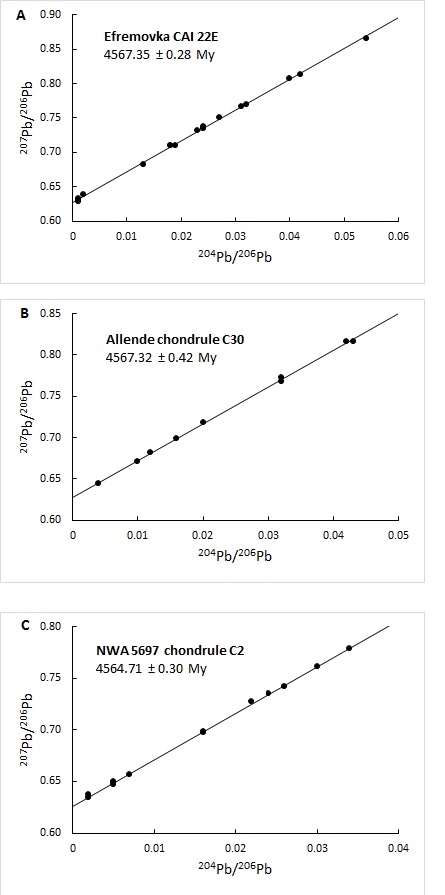
Uranium–lead dating, abbreviated U–Pb dating, is one of the oldest and most refined of the radiometric dating schemes. It can be used to date rocks that formed and crystallised from about 1 million years to over 4.5 billion years ago with routine precisions in the 0.1–1 percent range. The method is usually applied to zircon. This mineral incorporates uranium and thorium atoms into its crystal structure, but strongly rejects lead when forming. As a result, newly-formed zircon deposits will contain no lead, meaning that any lead found in the mineral is radiogenic. Since the exact rate at which uranium decays into lead is known, the current ratio of lead to uranium in a sample of the mineral can be used to reliably determine its age. The method relies on two separate decay chains, the uranium series from 238U to 206Pb, with a half-life of 4.47 billion years and the actinium series from 235U to 207Pb, with a half-life of 710 million years. Decay routes Uranium decays to lead via ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiometric Dating
Radiometric dating, radioactive dating or radioisotope dating is a technique which is used to date materials such as rocks or carbon, in which trace radioactive impurities were selectively incorporated when they were formed. The method compares the abundance of a naturally occurring radioactive isotope within the material to the abundance of its decay products, which form at a known constant rate of decay. The use of radiometric dating was first published in 1907 by Bertram Boltwood and is now the principal source of information about the absolute age of rocks and other geological features, including the age of fossilized life forms or the age of Earth itself, and can also be used to date a wide range of natural and man-made materials. Together with stratigraphic principles, radiometric dating methods are used in geochronology to establish the geologic time scale. Among the best-known techniques are radiocarbon dating, potassium–argon dating and uranium–lead dating. By al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead–lead Dating
Lead–lead dating is a method for dating geological samples, normally based on 'whole-rock' samples of material such as granite. For most dating requirements it has been superseded by uranium–lead dating (U–Pb dating), but in certain specialized situations (such as dating meteorites and the age of the Earth) it is more important than U–Pb dating. Decay equations for common Pb–Pb dating There are three stable "daughter" Pb isotopes that result from the radioactive decay of uranium and thorium in nature; they are 206Pb, 207Pb, and 208Pb. 204Pb is the only non-radiogenic lead isotope, therefore is not one of the daughter isotopes. These daughter isotopes are the final decay products of U and Th radioactive decay chains beginning from 238U, 235U and 232Th respectively. With the progress of time, the final decay product accumulates as the parent isotope decays at a constant rate. This shifts the ratio of radiogenic Pb versus non-radiogenic 204Pb (207Pb/204Pb or 206Pb/204Pb) in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Daughter Isotope
In nuclear physics, a decay product (also known as a daughter product, daughter isotope, radio-daughter, or daughter nuclide) is the remaining nuclide left over from radioactive decay. Radioactive decay often proceeds via a sequence of steps (decay chain). For example, 238U decays to 234Th which decays to 234mPa which decays, and so on, to 206Pb (which is stable): : \ce \overbrace^\ce left, upThe decay chain from lead-212 down to lead-208, showing the intermediate decay products In this example: * 234Th, 234mPa,...,206Pb are the decay products of 238U. * 234Th is the daughter of the parent 238U. * 234mPa (234 metastable) is the granddaughter of 238U. These might also be referred to as the daughter products of 238U. (''Depleted Uranium'' — authors: Naomi H. Harley, Ernest C. Foulkes, Lee H. Hil ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha Decay
Alpha decay or α-decay is a type of radioactive decay in which an atomic nucleus emits an alpha particle (helium nucleus) and thereby transforms or 'decays' into a different atomic nucleus, with a mass number that is reduced by four and an atomic number that is reduced by two. An alpha particle is identical to the nucleus of a helium-4 atom, which consists of two protons and two neutrons. It has a charge of and a mass of . For example, uranium-238 decays to form thorium-234. While alpha particles have a charge , this is not usually shown because a nuclear equation describes a nuclear reaction without considering the electrons – a convention that does not imply that the nuclei necessarily occur in neutral atoms. Alpha decay typically occurs in the heaviest nuclides. Theoretically, it can occur only in nuclei somewhat heavier than nickel (element 28), where the overall binding energy per nucleon is no longer a maximum and the nuclides are therefore unstable toward spontaneou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metamorphic Rock
Metamorphic rocks arise from the transformation of existing rock to new types of rock in a process called metamorphism. The original rock (protolith) is subjected to temperatures greater than and, often, elevated pressure of or more, causing profound physical or chemical changes. During this process, the rock remains mostly in the solid state, but gradually recrystallizes to a new texture or mineral composition. The protolith may be an igneous, sedimentary, or existing metamorphic rock. Metamorphic rocks make up a large part of the Earth's crust and form 12% of the Earth's land surface. They are classified by their protolith, their chemical and mineral makeup, and their texture. They may be formed simply by being deeply buried beneath the Earth's surface, where they are subject to high temperatures and the great pressure of the rock layers above. They can also form from tectonic processes such as continental collisions, which cause horizontal pressure, friction, and distorti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Igneous
Igneous rock (derived from the Latin word ''ignis'' meaning fire), or magmatic rock, is one of the three main rock types, the others being sedimentary and metamorphic. Igneous rock is formed through the cooling and solidification of magma or lava. The magma can be derived from partial melts of existing rocks in either a planet's mantle or crust. Typically, the melting is caused by one or more of three processes: an increase in temperature, a decrease in pressure, or a change in composition. Solidification into rock occurs either below the surface as intrusive rocks or on the surface as extrusive rocks. Igneous rock may form with crystallization to form granular, crystalline rocks, or without crystallization to form natural glasses. Igneous rocks occur in a wide range of geological settings: shields, platforms, orogens, basins, large igneous provinces, extended crust and oceanic crust. Geological significance Igneous and metamorphic rocks make up 90–95% of the top ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonate Mineral
Carbonate minerals are those minerals containing the carbonate ion, . Carbonate divisions Anhydrous carbonates *Calcite group: trigonal **Calcite CaCO3 **Gaspéite (Ni,Mg,Fe2+)CO3 **Magnesite MgCO3 **Otavite CdCO3 **Rhodochrosite MnCO3 **Siderite FeCO3 **Smithsonite ZnCO3 **Spherocobaltite CoCO3 *Aragonite group: orthorhombic **Aragonite CaCO3 **Cerussite PbCO3 **Strontianite SrCO3 **Witherite BaCO3 **Rutherfordine UO2CO3 **Natrite Na2CO3 Anhydrous carbonates with compound formulas *Dolomite group: trigonal **Ankerite CaFe(CO3)2 **Dolomite (mineral), Dolomite CaMg(CO3)2 **Huntite Mg3Ca(CO3)4 **Minrecordite CaZn(CO3)2 **Barytocalcite BaCa(CO3)2 Carbonates with hydroxyl or halogen *Carbonate with hydroxide: monoclinic **Azurite Cu3(CO3)2(OH)2 **Hydrocerussite Pb3(CO3)2(OH)2 **Malachite Cu2CO3(OH)2 **Rosasite (Cu,Zn)2CO3(OH)2 **Phosgenite Pb2(CO3)Cl2 **Hydrozincite Zn5(CO3)2(OH)6 **Aurichalcite (Zn,Cu)5(CO3)2(OH)6 Hydrated carbonates *Hydromagnesite Mg5(CO3)4(OH)2.4H2O *Ikaite ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aragonite
Aragonite is a carbonate mineral, one of the three most common naturally occurring crystal forms of calcium carbonate, (the other forms being the minerals calcite and vaterite). It is formed by biological and physical processes, including precipitation from marine and freshwater environments. The crystal lattice of aragonite differs from that of calcite, resulting in a different crystal shape, an orthorhombic crystal system with acicular crystal. Repeated twinning results in pseudo-hexagonal forms. Aragonite may be columnar or fibrous, occasionally in branching helictitic forms called ''flos-ferri'' ("flowers of iron") from their association with the ores at the Carinthian iron mines. Occurrence The type location for aragonite is Molina de Aragón in the Province of Guadalajara in Castilla-La Mancha, Spain, for which it was named in 1797. Aragonite is found in this locality as cyclic twins inside gypsum and marls of the Keuper facies of the Triassic. This type of arago ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcite
Calcite is a Carbonate minerals, carbonate mineral and the most stable Polymorphism (materials science), polymorph of calcium carbonate (CaCO3). It is a very common mineral, particularly as a component of limestone. Calcite defines hardness 3 on the Mohs scale of mineral hardness, based on Scratch hardness, scratch hardness comparison. Large calcite crystals are used in optical equipment, and limestone composed mostly of calcite has numerous uses. Other polymorphs of calcium carbonate are the minerals aragonite and vaterite. Aragonite will change to calcite over timescales of days or less at temperatures exceeding 300 °C, and vaterite is even less stable. Etymology Calcite is derived from the German ''Calcit'', a term from the 19th century that came from the Latin word for Lime (material), lime, ''calx'' (genitive calcis) with the suffix "-ite" used to name minerals. It is thus etymologically related to chalk. When applied by archaeology, archaeologists and stone trade pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Baddeleyite
Baddeleyite is a rare zirconium oxide mineral (ZrO2 or zirconia), occurring in a variety of monoclinic prismatic crystal forms. It is transparent to translucent, has high indices of refraction, and ranges from colorless to yellow, green, and dark brown. See etymology below. Baddeleyite is a refractory mineral, with a melting point of 2700 °C. Hafnium is a substituting ''impurity'' and may be present in quantities ranging from 0.1 to several percent. It can be found in igneous rocks containing potassium feldspar and plagioclase. Baddeleyite is commonly not found with zircon (ZrSiO4), because it forms in silica-undersaturated rocks, such as mafic rocks. This is because, when silica is free in the system (silica-saturated/oversaturated), zircon is the dominating phase, not baddeleyite. It belongs to the monoclinic-prismatic class, of the P21/c crystal system. It has been used for geochronology. Geologic occurrence Baddeleyite was first found in Sri Lanka in 1892. It can b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanite
Titanite, or sphene (from the Greek ''sphenos'' (σφηνώ), meaning wedge), is a calcium titanium nesosilicate mineral, Ca Ti Si O5. Trace impurities of iron and aluminium are typically present. Also commonly present are rare earth metals including cerium and yttrium; calcium may be partly replaced by thorium. Nomenclature The International Mineralogical Association Commission on New Minerals and Mineral Names (CNMMN) adopted the name titanite and "discredited" the name sphene as of 1982, although commonly papers and books initially identify the mineral using both names. Sphene was the most commonly used name until the IMA decision, although both were well known. Some authorities think it is less confusing as the word is used to describe any chemical or crystal with oxidized titanium such as the rare earth titanate pyrochlores series and many of the minerals with the perovskite structure. The name sphene continues to be publishable in peer-reviewed scientific literature, e.g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monazite Geochronology
Monazite geochronology is a dating technique to study geological history using the mineral monazite. It is a powerful tool in studying the complex history of metamorphic rocks particularly, as well as igneous, sedimentary and hydrothermal rocks.Williams, M. L., Jercinovic, M. J., & Hetherington, C. J. (2007). Microprobe monazite geochronology: understanding geologic processes by integrating composition and chronology. Annual Review of Earth and Planetary Sciences, 35(1), 137.Williams, M. L., & Jercinovic, M. J. (2002). Microprobe monazite geochronology: putting absolute time into microstructural analysis. Journal of Structural Geology, 24(6), 1013–1028. The dating uses the radioactive processes in monazite as a clock. The uniqueness of monazite geochronology comes from the high thermal resistance of monazite, which allows age information to be retained during the geological history.Crowley, J. L., & Ghent, E. D. (1999). An electron microprobe study of the U–Th–Pb systematic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |