|
Supplementary Protection Certificate
In the European Economic Area (European Union member countries, Iceland, Liechtenstein and Norway), a supplementary protection certificate (SPC) is a '' sui generis'' intellectual property (IP) right that extends the duration of certain rights associated with a patent. It enters into force after expiry of a patent upon which it is based. This type of right is available for various regulated, biologically active agents, namely human or veterinary medicaments and plant protection products (e.g. insecticides, and herbicides). Supplementary protection certificates were introduced to encourage innovation by compensating for the long time needed to obtain regulatory approval of these products (i.e. authorisation to put these products on the market). A supplementary protection certificate comes into force only after the corresponding general patent expires. It normally has a maximum lifetime of 5 years. The duration of the SPC can, however, be extended to 5.5 years when the SPC relat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
European Economic Area
The European Economic Area (EEA) was established via the ''Agreement on the European Economic Area'', an international agreement which enables the extension of the European Union's single market to member states of the European Free Trade Association. The EEA links the EU member states and three EFTA states (Iceland, Liechtenstein, and Norway) into an internal market governed by the same basic rules. These rules aim to enable free movement of persons, goods, services, and capital within the European single market, including the freedom to choose residence in any country within this area. The EEA was established on 1 January 1994 upon entry into force of the EEA Agreement. The contracting parties are the EU, its member states, and Iceland, Liechtenstein, and Norway. The EEA Treaty is a commercial treaty and differs from the EU Treaties in certain key respects. According to Article 1 its purpose is to "promote a continuous and balanced strengthening of trade and economic relati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
European Medicines Agency
The European Medicines Agency (EMA) is an agency of the European Union (EU) in charge of the evaluation and supervision of medicinal products. Prior to 2004, it was known as the European Agency for the Evaluation of Medicinal Products or European Medicines Evaluation Agency (EMEA).Set up by EC Regulation No. 2309/93 as the European Agency for the Evaluation of Medicinal Products, and renamed by EC Regulation No. 726/2004 to the European Medicines Agency, it had the acronym EMEA until December 2009. The European Medicines Agency does not call itself EMA either – it has no official acronym but may reconsider if EMA becomes commonly accepted (secommunication on new visual identity an). The EMA was set up in 1995, with funding from the European Union and the pharmaceutical industry, as well as indirect subsidy from member states, its stated intention to harmonise (but not replace) the work of existing national medicine regulatory bodies. The hope was that this plan would not onl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
United Kingdom Patent Office
The Intellectual Property Office of the United Kingdom (often referred to as the UK IPO) is, since 2 April 2007, the operating name of The Patent Office. It is the official government body responsible for intellectual property rights in the UK and is an executive agency of the Department for Business, Energy and Industrial Strategy (BEIS). Responsibilities The IPO has direct administrative responsibility for examining and issuing or rejecting patents, and maintaining registers of intellectual property including patents, designs and trade marks in the UK. As in most countries, there is no statutory register of copyright and the IPO does not conduct any direct administration in copyright matters. The IPO is led by the Comptroller General of Patents, Designs and Trade Marks, who is also Registrar of Trade Marks, Registrar of Designs and Chief Executive of the IPO. Since 1 May 2017, the Comptroller has been Tim Moss, following the resignation of John Alty who had been Comptroller ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
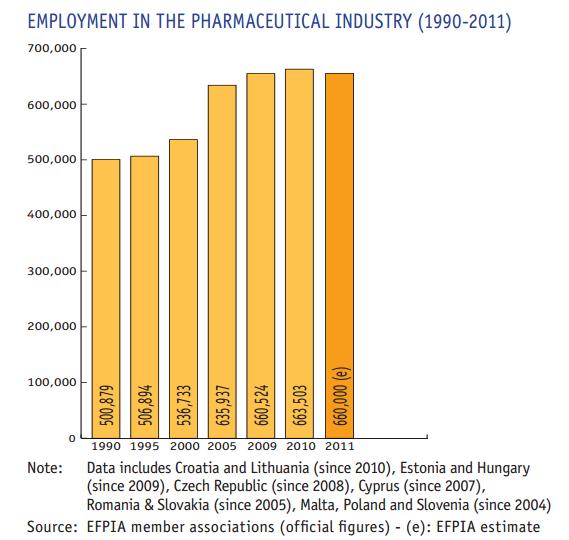
European Federation Of Pharmaceutical Industries And Associations
The European Federation of Pharmaceutical Industries and Associations (EFPIA) is a Brussels-based trade association and lobbying organisation, founded in 1978 and representing the research-based pharmaceutical industry operating in Europe. Through its membership of 37 national associations and 38 leading pharmaceutical companies, EFPIA represents 1,900 european companies in the areas of researching, developing and manufacturing new medical treatments. Figures in 2021 by the European Commission showed that based on the companies included in its scoreboard Health industries spent 19.9% of all the business research & development in Europe, only surpassed by the Automobiles & Parts industry (33.6%). But the percentage of european spending on R&D in this sector was considerably lower than in the United States. The key contribution of the research-based pharmaceutical industry to medical progress is to turn fundamental research into innovative treatments that are widely available and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drug
An orphan drug is a pharmaceutical agent developed to treat medical conditions which, because they are so rare, would not be profitable to produce without government assistance. The conditions are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy in many countries and has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug research and development. In the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to encourage development of drugs which would otherwise lack sufficient profit motive to attract corporate research budgets and personnel. Definition According to the US Food and Drug Administration (FDA), an orphan drug is defined as one "intended for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Generic Pharmaceutical Price Decay
Generic pharmaceutical price decay is what happens (in the UK) once the originator brand has lost its Generic drug#Generic drug exclusivity in the US, patent exclusivity (patent expiry) and generic versions of the originator brand have been launched. The number of license holders entering the market is controlled by the ease of manufacture and the number of companies making the Active pharmaceutical ingredient, active pharmaceutical ingredient (API). For many easy-to-manufacture solid dose tablets and capsules the manufacturing is done in India and China as the costs of production in these countries is significantly lower than in the US or Europe. On day one of generic launch the first to market the generic product usually gets more market share than late entrants. Both manufacturers (who make their own generic products) and license only holders (who use other companies to do the manufacturing) may be represented. If only one company is able to release a generic product into th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
German Federal Court Of Justice
The Federal Court of Justice (german: Bundesgerichtshof, BGH) is the highest court in the system of ordinary jurisdiction (''ordentliche Gerichtsbarkeit'') in Germany, founded in 1950. It has its seat in Karlsruhe with two panels being situated in Leipzig since 1997 and 2020, respectively. It is the supreme court (court of last resort) in all matters of criminal law and private law. A decision handed down by the BGH can be reversed only by the Federal Constitutional Court of Germany on constitutionality (compatibility with the Basic Law for the Federal Republic of Germany) grounds. History Before the Federal Court of Justice of Germany was created in its present form, Germany had several highest courts: As early as 1495 there was the ''Reichskammergericht'', which existed until 1806. As from 1870, in the time of the North German Confederation, there was the '' Bundesoberhandelsgericht'' in Leipzig. In 1871, it was renamed to ''Reichsoberhandelsgericht'' and its area of respo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
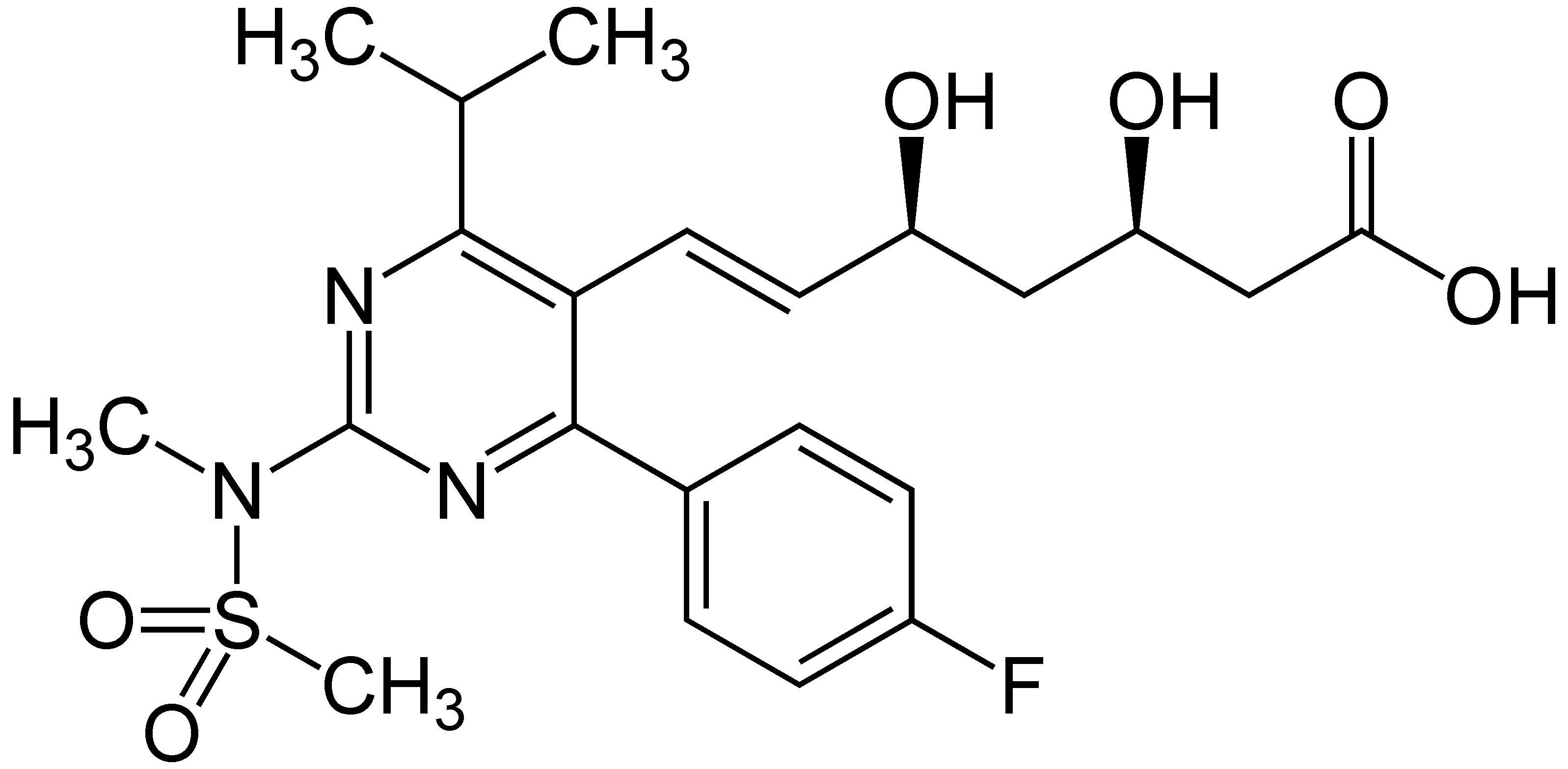
Sitagliptin
Sitagliptin, sold under the brand name Januvia among others, is an anti-diabetic medication used to treat type 2 diabetes. In the United Kingdom it is listed as less preferred than metformin or a sulfonylurea. It is taken by mouth. It is also available in the fixed-dose combination medication sitagliptin/metformin (Janumet, Janumet XR). Common side effects include headaches, swelling of the legs, and upper respiratory tract infections. Serious side effects may include angioedema, low blood sugar, kidney problems, pancreatitis, and joint pain. Whether use in pregnancy or breastfeeding is safe is unclear. It is in the dipeptidyl peptidase-4 (DPP-4) inhibitor class and works by increasing the production of insulin and decreasing the production of glucagon by the pancreas. Sitagliptin was developed by Merck & Co. and approved for medical use in the United States in 2006. In 2020, it was the 74th most commonly prescribed medication in the United States, with more than 9million ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
European Court Of Justice
The European Court of Justice (ECJ, french: Cour de Justice européenne), formally just the Court of Justice, is the supreme court of the European Union in matters of European Union law. As a part of the Court of Justice of the European Union, it is tasked with interpreting EU law and ensuring its uniform application across all EU member states under Article 263 of the Treaty of the Functioning of the European Union (TFEU). The Court was established in 1952, and is based in Luxembourg. It is composed of one judge per member state – currently – although it normally hears cases in panels of three, five or fifteen judges. The Court has been led by president Koen Lenaerts since 2015. The ECJ is the highest court of the European Union in matters of Union law, but not national law. It is not possible to appeal against the decisions of national courts in the ECJ, but rather national courts refer questions of EU law to the ECJ. However, it is ultimately for the national court ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liechtenstein
Liechtenstein (), officially the Principality of Liechtenstein (german: link=no, Fürstentum Liechtenstein), is a German-speaking microstate located in the Alps between Austria and Switzerland. Liechtenstein is a semi-constitutional monarchy headed by the prince of Liechtenstein. Liechtenstein is bordered by Switzerland to the west and south and Austria to the east and north. It is Europe's fourth-smallest country, with an area of just over and a population of 38,749 (). Divided into 11 municipalities, its capital is Vaduz, and its largest municipality is Schaan. It is also the smallest country to border two countries. Liechtenstein is a doubly landlocked country between Switzerland and Austria. Economically, Liechtenstein has one of the highest gross domestic products per person in the world when adjusted for purchasing power parity. The country has a strong financial sector centred in Vaduz. It was once known as a billionaire tax haven, but is no longer on any officia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Switzerland
). Swiss law does not designate a ''capital'' as such, but the federal parliament and government are installed in Bern, while other federal institutions, such as the federal courts, are in other cities (Bellinzona, Lausanne, Luzern, Neuchâtel, St. Gallen a.o.). , coordinates = , largest_city = Zürich , official_languages = , englishmotto = "One for all, all for one" , religion_year = 2020 , religion_ref = , religion = , demonym = , german: Schweizer/Schweizerin, french: Suisse/Suissesse, it, svizzero/svizzera or , rm, Svizzer/Svizra , government_type = Federalism, Federal assembly-independent Directorial system, directorial republic with elements of a direct democracy , leader_title1 = Federal Council (Switzerland), Federal Council , leader_name1 = , leader_title2 = , leader_name2 = Walter Thurnherr , legislature = Fe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Term Of Patent
The term of a patent is the maximum time during which it can be maintained in force. It is usually expressed in a number of years either starting from the filing date of the patent application or from the date of grant of the patent. In most patent laws, annuities or maintenance fees have to be regularly paid in order to keep the patent in force. Thus, a patent may lapse before its term if a renewal fee is not paid in due time. International harmonization Significant international harmonization of patent term across national laws was provided in the 1990s by the implementation of the WTO's Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPs Agreement). Article 33 of the TRIPs Agreement provides that :"The term of protection available or patentsshall not end before the expiration of a period of twenty years counted from the filing date." Consequently, in most patent laws nowadays, the term of patent is 20 years from the filing date of the application. This h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |