|
Stannosis
Stannosis is an occupational, non-fibrotic pneumoconiosis caused by chronic exposure and inhalation of tin. Pneumoconiosis is essentially when inorganic dust is found on the lung tissue; in this case, caused by tin oxide minerals. Dust particles and fumes from tin industries, stannous oxide (SnO) and stannic oxide (SnO2), are specific to stannosis diagnoses. Hazardous occupations such as, tinning, tin-working, and smelting are where most cases of stannosis are documented. When melted tin ions are inhaled as a fume, the tin oxides deposit onto the lung nodules and immune response cells. If a worker is exposed to tin oxides over multiple events for an extended time, they are at risk of developing stannosis. Toxicology and physiology Workers with acute exposures, short duration with varying dose concentrations, to tin oxides develop a mild irritation on the eyes, skin, and mucous membranes. When the inorganic metal materials are inhaled, the body activates the immune system and sen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pneumoconiosis
Pneumoconiosis is the general term for a class of interstitial lung disease where inhalation of dust ( for example, ash dust, lead particles, pollen grains etc) has caused interstitial fibrosis. The three most common types are asbestosis, silicosis, and coal miner's lung. Pneumoconiosis often causes restrictive impairment, although diagnosable pneumoconiosis can occur without measurable impairment of lung function. Depending on extent and severity, it may cause death within months or years, or it may never produce symptoms. It is usually an occupational lung disease, typically from years of dust exposure during work in mining; textile milling; shipbuilding, ship repairing, and/or shipbreaking; sandblasting; industrial tasks; rock drilling (subways or building pilings); or agriculture. It is one of the most common occupational diseases in the world. Types Depending upon the type of dust, the disease is given different names: * Coalworker's pneumoconiosis (also known as coal miner' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Insoluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. M. Diepen (1966): "Gas—Gas Equilibria". ''Journal of Chemical Physics'', vo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
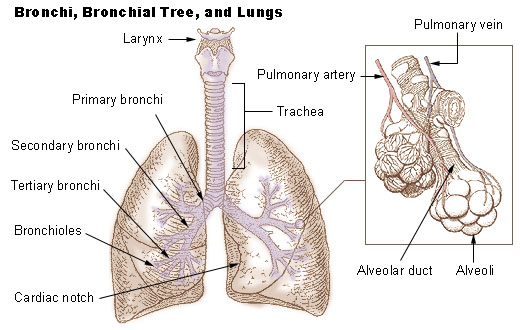
Pulmonary Lesion
The lungs are the primary Organ (anatomy), organs of the respiratory system in humans and most other animals, including some snails and a small number of fish. In mammals and most other vertebrates, two lungs are located near the vertebral column, backbone on either side of the heart. Their function in the respiratory system is to extract oxygen from the air and transfer it into the bloodstream, and to release carbon dioxide from the bloodstream into the Atmosphere of Earth, atmosphere, in a process of gas exchange. Respiration (physiology), Respiration is driven by different muscle, muscular systems in different species. Mammals, reptiles and birds use their different muscles to support and foster breathing. In earlier tetrapods, air was driven into the lungs by the pharyngeal muscles via buccal pumping, a mechanism still seen in amphibians. In humans, the main muscles of respiration, muscle of respiration that drives breathing is the thoracic diaphragm, diaphragm. The lungs a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bronchoalveolar Lavage
Bronchoalveolar lavage (BAL) (also known as bronchoalveolar washing) is a diagnostic method of the lower respiratory system in which a bronchoscope is passed through the mouth or nose into an appropriate airway in the lungs, with a measured amount of fluid introduced and then collected for examination. This method is typically performed to diagnose pathogenic infections of the lower respiratory airways (leading to, for example pneumonia and COVID-19), though it also has been shown to have utility in diagnosing interstitial lung disease. Bronchoalveolar lavage can be a more sensitive method of detection than nasal swabs in respiratory molecular diagnostics, as has been the case with SARS-CoV-2 where bronchoalveolar lavage samples detect copies of viral RNA after negative nasal swab testing. In particular, bronchoalveolar lavage is commonly used to diagnose infections in people with immune system problems, pneumonia in people on ventilators, and acute respiratory distress syndrome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Personal Protective Equipment
Personal protective equipment (PPE) is protective clothing, helmets, goggles, or other garments or equipment designed to protect the wearer's body from injury or infection. The hazards addressed by protective equipment include physical, electrical, heat, chemicals, biological hazard, biohazards, and Atmospheric particulate matter, airborne particulate matter. Protective equipment may be worn for job-related occupational safety and health purposes, as well as for sports and other recreation, recreational activities. ''Protective clothing'' is applied to traditional categories of clothing, and ''protective gear'' applies to items such as pads, guards, shields, or masks, and others. PPE suits can be similar in appearance to a cleanroom suit. The purpose of personal protective equipment is to reduce employee exposure to hazards when engineering controls and administrative controls are not feasible or effective to reduce these risks to acceptable levels. PPE is needed when there a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Engineering Controls
Engineering controls are strategies designed to protect workers from hazardous conditions by placing a barrier between the worker and the hazard or by removing a hazardous substance through air ventilation. Engineering controls involve a physical change to the workplace itself, rather than relying on workers' behavior or requiring workers to wear protective clothing. Engineering controls is the third of five members of the hierarchy of hazard controls, which orders control strategies by their feasibility and effectiveness. Engineering controls are preferred over administrative controls and personal protective equipment (PPE) because they are designed to remove the hazard at the source, before it comes in contact with the worker. Well-designed engineering controls can be highly effective in protecting workers and will typically be independent of worker interactions to provide this high level of protection. The initial cost of engineering controls can be higher than the cost of ad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid Acid strength is the tendency of an acid, symbolised by the chemical formula HA, to dissociate into a proton, H+, and an anion, A-. The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions .... It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. History In the early tenth century, the Persian physician and alchemist Abu Bakr al-Razi ( 865–925, Latin: Rhazes) conducted experiments with sal ammoniac (ammonium chloride) and vitriol (hydrated sulfates of various metals), which he distilled together, thus producing the gas hydrogen chloride. In doing so, al-Razi may have stumbled upon a primitive method ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfuric Acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular formula . It is a colorless, odorless and viscous liquid that is miscible with water. Pure sulfuric acid does not exist naturally on Earth due to its strong affinity to water vapor; it is hygroscopic and readily absorbs water vapor from the air. Concentrated sulfuric acid is highly corrosive towards other materials, from rocks to metals, since it is an oxidant with powerful dehydrating properties. Phosphorus pentoxide is a notable exception in that it is not dehydrated by sulfuric acid, but to the contrary dehydrates sulfuric acid to sulfur trioxide. Upon addition of sulfuric acid to water, a considerable amount of heat is released; thus the reverse procedure of adding water to the acid should not be performed since the heat released may boi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SnO2
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. The mineral form of SnO2 is called cassiterite, and this is the main ore of tin. With many other names, this oxide of tin is an important material in tin chemistry. It is a colourless, diamagnetic, amphoteric solid. Structure Tin(IV) oxide crystallises with the rutile structure. As such the tin atoms are six coordinate and the oxygen atoms three coordinate. SnO2 is usually regarded as an oxygen-deficient n-type semiconductor. Hydrous forms of SnO2 have been described as stannic acid. Such materials appear to be hydrated particles of SnO2 where the composition reflects the particle size. Preparation Tin(IV) oxide occurs naturally. Synthetic tin(IV) oxide is produced by burning tin metal in air. Annual production is in the range of 10 kilotons. SnO2 is reduced industrially to the metal with carbon in a reverberatory furnace at 1200–1300 °C. Amphoterism Although SnO2 is insolu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Safety Data Sheet
A safety data sheet (SDS), material safety data sheet (MSDS), or product safety data sheet (PSDS) is a document that lists information relating to occupational safety and health for the use of various substances and products. SDSs are a widely used system for cataloguing information on chemicals, chemical compounds, and chemical mixtures. SDS information may include instructions for the safe use and potential hazards associated with a particular material or product, along with spill-handling procedures. The older MSDS formats could vary from source to source within a country depending on national requirements; however, the newer SDS format is internationally standardized. An SDS for a substance is not primarily intended for use by the general consumer, focusing instead on the hazards of working with the material in an occupational setting. There is also a duty to properly label substances on the basis of physico-chemical, health, or environmental risk. Labels can include hazard ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allotropy
Allotropy or allotropism () is the property of some chemical elements to exist in two or more different forms, in the same physical state, known as allotropes of the elements. Allotropes are different structural modifications of an element: the atoms of the element are bonded together in a different manner. For example, the allotropes of carbon include diamond (the carbon atoms are bonded together to form a cubic lattice of tetrahedra), graphite (the carbon atoms are bonded together in sheets of a hexagonal lattice), graphene (single sheets of graphite), and fullerenes (the carbon atoms are bonded together in spherical, tubular, or ellipsoidal formations). The term ''allotropy'' is used for elements only, not for compounds. The more general term, used for any compound, is polymorphism, although its use is usually restricted to solid materials such as crystals. Allotropy refers only to different forms of an element within the same physical phase (the state of matter, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stannous Oxide
Tin(II) oxide (stannous oxide) is a compound with the formula SnO. It is composed of tin and oxygen where tin has the oxidation state of +2. There are two forms, a stable blue-black form and a metastable red form. Preparation and reactions Blue-black SnO can be produced by heating the tin(II) oxide hydrate, SnO·xH2O (x<1) precipitated when a tin(II) salt is reacted with an alkali hydroxide such as NaOH.Egon Wiberg, Arnold Frederick Holleman (2001) ''Inorganic Chemistry'', Elsevier Metastable, red SnO can be prepared by gentle heating of the precipitate produced by the action of aqueous ammonia on a tin(II) salt. SnO may be prepared as a pure substance in the laboratory, by controlled heating of tin(II) oxalate ( stannous oxalate) in the absence of air or under a CO2 atmosphere. This method is als ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |