|
Scrutinyite
Scrutinyite is a rare oxide mineral and is the alpha crystalline form of lead dioxide (α-PbO2), plattnerite being the other, beta form. The mineral was first reported in 1988 and its name reflects the scrutiny and efforts required to identify it from a very limited amount of available sample material. Identification The synthetic orthorhombic form of lead dioxide, α-PbO2, was known from 1941. Although natural lead dioxide has been known, as the mineral plattnerite (β-PbO2), since 1845,Haidinger W (1845Zweite Klasse: Geogenide. II. Ordnung. Baryte VII. Bleibaryt. Plattnerit. p. 500 in Handbuch der Bestimmenden Mineralogie Bei Braumüller and Seidel Wien pp. 499-506 (in German) its alpha form could only be recognized in 1981 and reliably identified in 1988. The new mineral was spotted in several samples collected at Bingham, New Mexico and Mapimí, Durango, Mexico. It was first thought to be minium ( lead tetroxide mineral) because of its high lead content, brown color and a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead Dioxide
Lead(IV) oxide is the inorganic compound with the formula PbO2. It is an oxide where lead is in an oxidation state of +4. It is a dark-brown solid which is insoluble in water. It exists in two crystalline forms. It has several important applications in electrochemistry, in particular as the positive plate of lead acid batteries. Properties Physical Lead dioxide has two major polymorphs, alpha and beta, which occur naturally as rare minerals scrutinyite and plattnerite, respectively. Whereas the beta form had been identified in 1845, α-PbO2 was first identified in 1946 and found as a naturally occurring mineral 1988. The alpha form has orthorhombic symmetry, space group Pbcn (No. 60), Pearson symbol ''oP''12, lattice constants ''a'' = 0.497 nm, ''b'' = 0.596 nm, ''c'' = 0.544 nm, ''Z'' = 4 (four formula units per unit cell). The lead atoms are six-coordinate. The symmetry of the beta form is tetragonal, space group P42/mnm (No. 136), Pearson symbol ''tP''6, la ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide Mineral
The oxide mineral class includes those minerals in which the oxide anion (O2−) is bonded to one or more metal alloys. The hydroxide-bearing minerals are typically included in the oxide class. The minerals with complex anion groups such as the silicates, sulfates, carbonates and phosphates are classed separately. Simple oxides: *XO **Periclase group ***Periclase ***Manganosite **Zincite group ***Zincite *** Bromellite ***Tenorite ***Litharge * **Cuprite **Ice * **Hematite group ***Corundum ***Hematite ***Ilmenite * **Rutile group ***Rutile ***Pyrolusite ***Cassiterite **Baddeleyite **Uraninite **Thorianite * **Spinel group ***Spinel ***Gahnite ***Magnetite ***Franklinite ***Chromite **Chrysoberyl **Columbite *Hydroxide subgroup: **Brucite **Manganite ** Romanèchite **Goethite group: ***Diaspore ***Goethite Nickel–Strunz Classification -04- Oxides IMA-CNMNC proposes a new hierarchical scheme (Mills et al., 2009). This list uses it to modify ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide Minerals
The oxide mineral class includes those minerals in which the oxide anion (O2−) is bonded to one or more metal alloys. The hydroxide-bearing minerals are typically included in the oxide class. The minerals with complex anion groups such as the silicates, sulfates, carbonates and phosphates are classed separately. Simple oxides: *XO **Periclase group ***Periclase ***Manganosite **Zincite group ***Zincite *** Bromellite ***Tenorite ***Litharge * **Cuprite **Ice * **Hematite group ***Corundum ***Hematite ***Ilmenite * **Rutile group ***Rutile ***Pyrolusite *** Cassiterite ** Baddeleyite **Uraninite **Thorianite * **Spinel group ***Spinel ***Gahnite ***Magnetite ***Franklinite *** Chromite **Chrysoberyl **Columbite *Hydroxide subgroup: **Brucite **Manganite ** Romanèchite **Goethite group: ***Diaspore ***Goethite Nickel–Strunz Classification -04- Oxides IMA-CNMNC proposes a new hierarchical scheme (Mills et al., 2009). This list uses it to mod ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide Mineral
The oxide mineral class includes those minerals in which the oxide anion (O2−) is bonded to one or more metal alloys. The hydroxide-bearing minerals are typically included in the oxide class. The minerals with complex anion groups such as the silicates, sulfates, carbonates and phosphates are classed separately. Simple oxides: *XO **Periclase group ***Periclase ***Manganosite **Zincite group ***Zincite *** Bromellite ***Tenorite ***Litharge * **Cuprite **Ice * **Hematite group ***Corundum ***Hematite ***Ilmenite * **Rutile group ***Rutile ***Pyrolusite ***Cassiterite **Baddeleyite **Uraninite **Thorianite * **Spinel group ***Spinel ***Gahnite ***Magnetite ***Franklinite ***Chromite **Chrysoberyl **Columbite *Hydroxide subgroup: **Brucite **Manganite ** Romanèchite **Goethite group: ***Diaspore ***Goethite Nickel–Strunz Classification -04- Oxides IMA-CNMNC proposes a new hierarchical scheme (Mills et al., 2009). This list uses it to modify ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
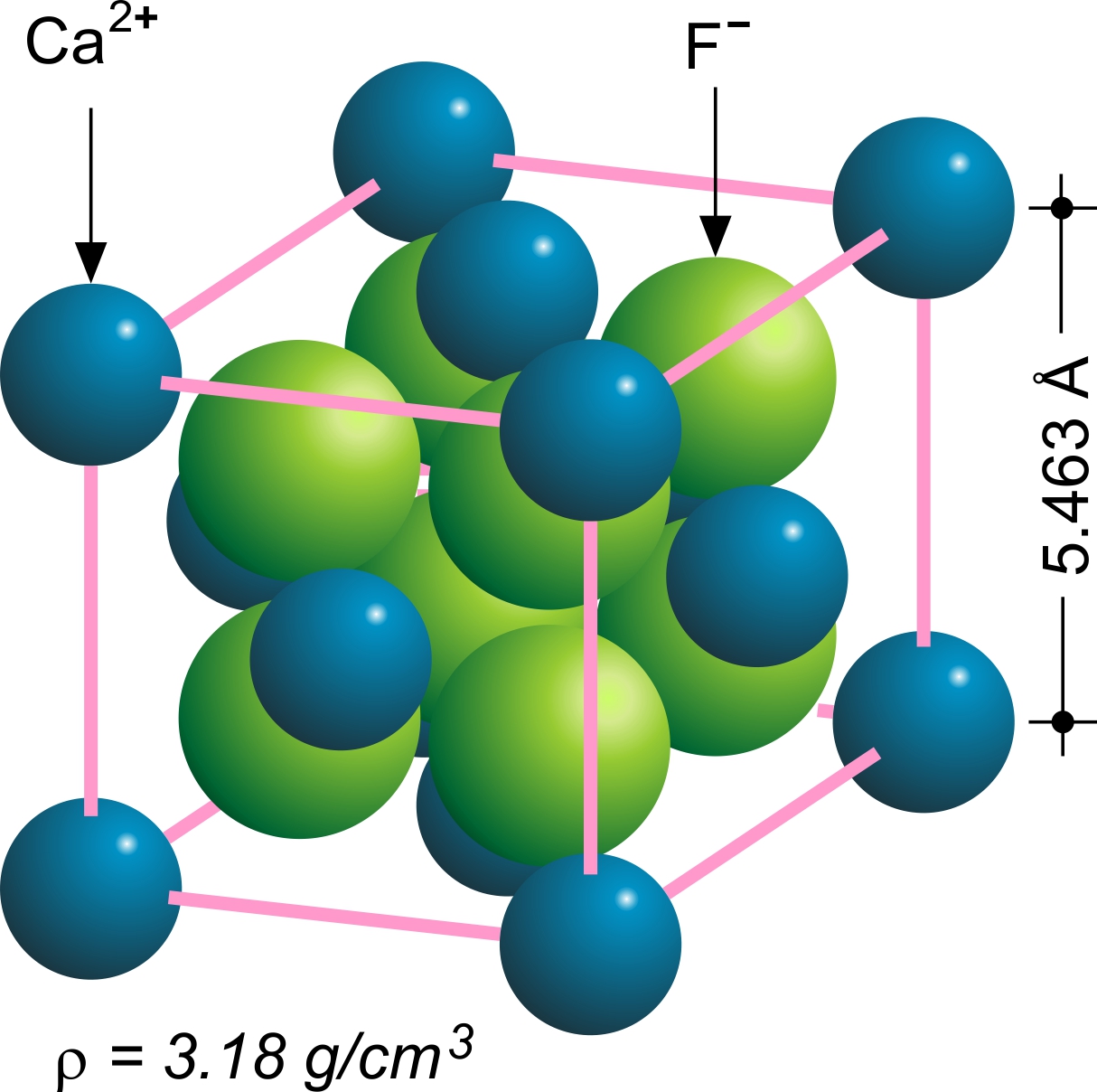
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite lenses have low dispersion, so lenses made from it exhibit less chromatic aberration, making them valuable in microscopes and telescopes. Fluorite optics are also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pearson Symbol
The Pearson symbol, or Pearson notation, is used in crystallography as a means of describing a crystal structure, and was originated by W. B. Pearson. The symbol is made up of two letters followed by a number. For example: * Diamond structure, ''cF''8 * Rutile structure, ''tP''6 The two (italicised) letters specify the Bravais lattice. The lower-case letter specifies the crystal family, and the upper-case letter the centering type. The number at the end of the Pearson symbol gives the number of the atoms in the conventional unit cell.Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 IR-3.4.4, pp. 49–51; IR-11.5, pp. 241–242. |
Space Group
In mathematics, physics and chemistry, a space group is the symmetry group of an object in space, usually in three dimensions. The elements of a space group (its symmetry operations) are the rigid transformations of an object that leave it unchanged. In three dimensions, space groups are classified into 219 distinct types, or 230 types if chiral copies are considered distinct. Space groups are discrete cocompact groups of isometries of an oriented Euclidean space in any number of dimensions. In dimensions other than 3, they are sometimes called Bieberbach groups. In crystallography, space groups are also called the crystallographic or Fedorov groups, and represent a description of the symmetry of the crystal. A definitive source regarding 3-dimensional space groups is the ''International Tables for Crystallography'' . History Space groups in 2 dimensions are the 17 wallpaper groups which have been known for several centuries, though the proof that the list was complete was only ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion , called hydroxide, and the neutral radical , known as the hydroxyl radical, consist of an unbonded hydroxy group. According to IUPAC definitions, the term ''hydroxyl'' refers to the hydroxyl radical () only, while the functional group is called a ''hydroxy group''. Properties Water, alcohols, carboxylic acids, and many other hydroxy-containing compounds can be readily deprotonated due to a large difference between the electronegativity of oxygen (3.5) and that of hydrogen (2.1). Hydroxy-containing compounds engage in intermolecular hydrogen bonding increasing the electrostatic attraction between molecules and thus to higher boiling and melting points than found for compounds that lack this f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy-dispersive X-ray Spectroscopy
Energy-dispersive X-ray spectroscopy (EDS, EDX, EDXS or XEDS), sometimes called energy dispersive X-ray analysis (EDXA or EDAX) or energy dispersive X-ray microanalysis (EDXMA), is an analytical technique used for the elemental analysis or chemical characterization of a sample. It relies on an interaction of some source of X-ray excitation and a sample. Its characterization capabilities are due in large part to the fundamental principle that each element has a unique atomic structure allowing a unique set of peaks on its electromagnetic emission spectrum (which is the main principle of spectroscopy). The peak positions are predicted by the Moseley's law with accuracy much better than experimental resolution of a typical EDX instrument. To stimulate the emission of characteristic X-rays from a specimen a beam of electrons is focused into the sample being studied. At rest, an atom within the sample contains ground state (or unexcited) electrons in discrete energy levels or electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
X-ray Crystallography
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles and intensities of these diffracted beams, a crystallographer can produce a three-dimensional picture of the density of electrons within the crystal. From this electron density, the mean positions of the atoms in the crystal can be determined, as well as their chemical bonds, their crystallographic disorder, and various other information. Since many materials can form crystals—such as salts, metals, minerals, semiconductors, as well as various inorganic, organic, and biological molecules—X-ray crystallography has been fundamental in the development of many scientific fields. In its first decades of use, this method determined the size of atoms, the lengths and types of chemical bonds, and the atomic-scale differences among various mat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rosasite
Rosasite is a carbonate mineral with minor potential for use as a zinc and copper ore. Chemically, it is a copper zinc carbonate hydroxide with a copper to zinc ratio of 3:2, occurring in the secondary oxidation zone of copper-zinc deposits. It was originally discovered in 1908 in the Rosas mine in Sardinia, Italy, and is named after the location. Fibrous blue-green rosasite crystals are usually found in globular aggregates, often associated with red limonite and other colorful minerals. It is very similar to aurichalcite Aurichalcite is a carbonate mineral, usually found as a secondary mineral in copper and zinc deposits. Its chemical formula is . The zinc to copper ratio is about 5:4. Occurrence Aurichalcite typically occurs in the oxidized zone of copper and z ..., but can be distinguished by its superior hardness. References Mineral galleries Copper(II) minerals Zinc minerals Carbonate minerals Monoclinic minerals Minerals in space group 14 Minerals described in 1908< ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |