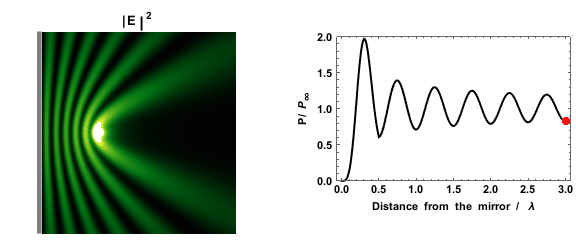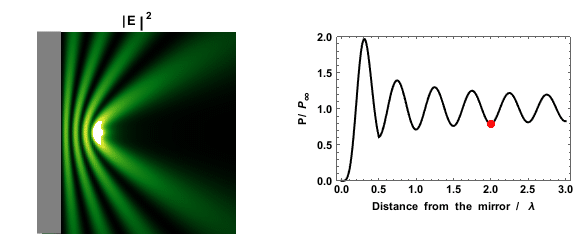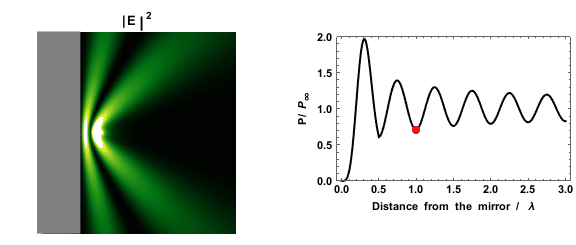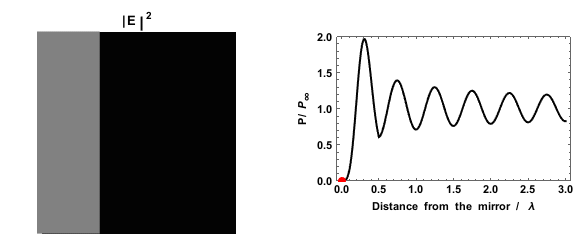
|
SEXAFS
Surface-extended X-ray absorption fine structure (SEXAFS) is the surface-sensitive equivalent of the EXAFS technique. This technique involves the illumination of the sample by high-intensity X-ray beams from a synchrotron and monitoring their photoabsorption by detecting in the intensity of Auger electrons as a function of the incident photon energy. Surface sensitivity is achieved by the interpretation of data depending on the intensity of the Auger electrons (which have an escape depth of ~1–2 nm) instead of looking at the relative absorption of the X-rays as in the parent method, EXAFS. The photon energies are tuned through the characteristic energy for the onset of core level excitation for surface atoms. The core holes thus created can then be filled by nonradiative decay of a higher-lying electron and communication of energy to yet another electron, which can then escape from the surface (Auger emission). The photoabsorption can therefore be monitored by direct dete ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SEXAFS Typical
Surface-extended X-ray absorption fine structure (SEXAFS) is the surface-sensitive equivalent of the EXAFS technique. This technique involves the illumination of the sample by high-intensity X-ray beams from a synchrotron and monitoring their photoabsorption by detecting in the intensity of Auger electrons as a function of the incident photon energy. Surface sensitivity is achieved by the interpretation of data depending on the intensity of the Auger electrons (which have an escape depth of ~1–2 nanometre, nm) instead of looking at the relative absorption of the X-rays as in the parent method, EXAFS. The photon energies are tuned through the characteristic energy for the onset of core electron, core level excitation for surface atoms. The core holes thus created can then be filled by nonradiative decay of a higher-lying electron and communication of energy to yet another electron, which can then escape from the surface (Auger emission). The photoabsorption can therefore be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
EXAFS
Extended X-ray absorption fine structure (EXAFS), along with X-ray absorption near edge structure (XANES), is a subset of X-ray absorption spectroscopy (XAS). Like other absorption spectroscopies, XAS techniques follow Beer's law. The X-ray absorption coefficient of a material as a function of energy is obtained using X-rays of a narrow energy resolution are directed at a sample and the incident and transmitted x-ray intensity is recorded as the incident x-ray energy is incremented. When the incident x-ray energy matches the binding energy of an electron of an atom within the sample, the number of x-rays absorbed by the sample increases dramatically, causing a drop in the transmitted x-ray intensity. This results in an absorption edge. Every element has a set of unique absorption edges corresponding to different binding energies of its electrons, giving XAS element selectivity. XAS spectra are most often collected at synchrotrons because of the high intensity of synchrotron X- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
EXAFS Setup
Extended X-ray absorption fine structure (EXAFS), along with X-ray absorption near edge structure (XANES), is a subset of X-ray absorption spectroscopy (XAS). Like other absorption spectroscopies, XAS techniques follow Beer's law. The X-ray absorption coefficient of a material as a function of energy is obtained using X-rays of a narrow energy resolution are directed at a sample and the incident and transmitted x-ray intensity is recorded as the incident x-ray energy is incremented. When the incident x-ray energy matches the binding energy of an electron of an atom within the sample, the number of x-rays absorbed by the sample increases dramatically, causing a drop in the transmitted x-ray intensity. This results in an absorption edge. Every element has a set of unique absorption edges corresponding to different binding energies of its electrons, giving XAS element selectivity. XAS spectra are most often collected at synchrotrons because of the high intensity of synchrotron X- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Joachim Stöhr
Joachim Stöhr (born September 28, 1947) is a physicist and professor emeritus of the Photon Science Department of Stanford University. His research has focused on the development of X-ray and synchrotron radiation techniques and their applications in different scientific fields with emphasis on surface science and magnetism. During his career he also held several scientific leadership positions, such as the director of the Stanford Synchrotron Radiation Laboratory (SSRL) and he was the founding director of the Linac Coherent Light Source (LCLS), the world's first x-ray free electron laser. Career Stöhr was born in Meinerzhagen, Germany. His university education was at Bonn University and Washington State University, where he spent two years as a Fulbright scholar and received a Master of Science degree. In 1974 he received his Ph.D. in Physics from the Technical University of Munich. After postdoctoral work and a scientific staff position at Lawrence Berkeley National Laboratory ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fermi's Golden Rule
In quantum physics, Fermi's golden rule is a formula that describes the transition rate (the probability of a transition per unit time) from one energy eigenstate of a quantum system to a group of energy eigenstates in a continuum, as a result of a weak perturbation. This transition rate is effectively independent of time (so long as the strength of the perturbation is independent of time) and is proportional to the strength of the coupling between the initial and final states of the system (described by the square of the matrix element of the perturbation) as well as the density of states. It is also applicable when the final state is discrete, i.e. it is not part of a continuum, if there is some decoherence in the process, like relaxation or collision of the atoms, or like noise in the perturbation, in which case the density of states is replaced by the reciprocal of the decoherence bandwidth. General Although the rule is named after Enrico Fermi, most of the work leading to it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Signal-to-noise Ratio
Signal-to-noise ratio (SNR or S/N) is a measure used in science and engineering that compares the level of a desired signal to the level of background noise. SNR is defined as the ratio of signal power to the noise power, often expressed in decibels. A ratio higher than 1:1 (greater than 0 dB) indicates more signal than noise. SNR, bandwidth, and channel capacity of a communication channel are connected by the Shannon–Hartley theorem. Definition Signal-to-noise ratio is defined as the ratio of the power of a signal (meaningful input) to the power of background noise (meaningless or unwanted input): : \mathrm = \frac, where is average power. Both signal and noise power must be measured at the same or equivalent points in a system, and within the same system bandwidth. Depending on whether the signal is a constant () or a random variable (), the signal-to-noise ratio for random noise becomes: : \mathrm = \frac where E refers to the expected value, i.e. in this case ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Auger Electron Spectroscopy
file:HD.6C.037 (11856519893).jpg, A Hanford Site, Hanford scientist uses an Auger electron spectrometer to determine the elemental composition of surfaces. Auger electron spectroscopy (AES; pronounced in French) is a common analytical technique used specifically in the study of surface science, surfaces and, more generally, in the area of materials science. It is a form of electron spectroscopy that relies on the Auger effect, based on the analysis of energetic electrons emitted from an excited atom after a series of internal relaxation events. The Auger effect was discovered independently by both Lise Meitner and Pierre Victor Auger, Pierre Auger in the 1920s. Though the discovery was made by Meitner and initially reported in the journal ''European Physical Journal, Zeitschrift für Physik'' in 1922, Auger is credited with the discovery in most of the scientific community. Until the early 1950s Auger transitions were considered nuisance effects by spectroscopists, not containing mu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Low-energy Electron Diffraction
Low-energy electron diffraction (LEED) is a technique for the determination of the surface structure of single-crystalline materials by bombardment with a collimated beam of low-energy electrons (30–200 eV) and observation of diffracted electrons as spots on a fluorescent screen. LEED may be used in one of two ways: # Qualitatively, where the diffraction pattern is recorded and analysis of the spot positions gives information on the symmetry of the surface structure. In the presence of an adsorbate the qualitative analysis may reveal information about the size and rotational alignment of the adsorbate unit cell with respect to the substrate unit cell. # Quantitatively, where the intensities of diffracted beams are recorded as a function of incident electron beam energy to generate the so-called I–V curves. By comparison with theoretical curves, these may provide accurate information on atomic positions on the surface at hand. Historical perspective An electron-diffractio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adsorbate
Adsorption is the adhesion of atoms, ions or molecules from a gas, liquid or dissolved solid to a surface. This process creates a film of the ''adsorbate'' on the surface of the ''adsorbent''. This process differs from absorption, in which a fluid (the ''absorbate'') is dissolved by or permeates a liquid or solid (the ''absorbent''). Adsorption is a '' surface phenomenon'', while absorption involves the whole volume of the material, although adsorption does often precede absorption. The term ''sorption'' encompasses both processes, while ''desorption'' is the reverse of it. Like surface tension, adsorption is a consequence of surface energy. In a bulk material, all the bonding requirements (be they ionic, covalent or metallic) of the constituent atoms of the material are fulfilled by other atoms in the material. However, atoms on the surface of the adsorbent are not wholly surrounded by other adsorbent atoms and therefore can attract adsorbates. The exact nature of the bond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Auger Emission
The Auger effect or Auger−Meitner effect is a physical phenomenon in which the filling of an inner-shell vacancy of an atom is accompanied by the emission of an electron from the same atom. When a core electron is removed, leaving a vacancy, an electron from a higher energy level may fall into the vacancy, resulting in a release of energy. Although most often this energy is released in the form of an emitted photon, the energy can also be transferred to another electron, which is ejected from the atom; this second ejected electron is called an Auger electron. Effect The effect was first discovered by Lise Meitner in 1922; Pierre Victor Auger independently discovered the effect shortly after and is credited with the discovery in most of the scientific community. Upon ejection, the kinetic energy of the Auger electron corresponds to the difference between the energy of the initial electronic transition into the vacancy and the ionization energy for the electron shell from which t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Core Electron
Core electrons are the electrons in an atom that are not valence electrons and do not participate in chemical bonding. The nucleus and the core electrons of an atom form the atomic core. Core electrons are tightly bound to the nucleus. Therefore, unlike valence electrons, core electrons play a secondary role in chemical bonding and reactions by screening the positive charge of the atomic nucleus from the valence electrons. The number of valence electrons of an element can be determined by the periodic table group of the element (see valence electron): *For main group elements, the number of valence electrons ranges from 1-8 electrons (''n''s and ''n''p orbitals). *For transition metals, the number of valence electrons ranges from 3-12 electrons (''n''s and (''n''−1)d orbitals). *For lanthanides and actinides, the number of valence electrons ranges from 3-16 electrons (''n''s, (''n''−2)f and (''n''−1)d orbitals). All other non-valence electrons for an atom of that element are c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |