|
Rhodium Carbonyl Chloride
Rhodium carbonyl chloride is an organorhodium compound with the formula Rh2Cl2(CO)4. It is a red-brown volatile solid that is soluble in nonpolar organic solvents. It is a precursor to other rhodium carbonyl complexes, some of which are useful in homogeneous catalysis. Structure The molecule consists of two planar Rh(I) centers linked by two bridging chloride ligands and four CO ligands. X-ray crystallography shows that the two Rh(I) centers are square planar with the dihedral angle of 53° between the two RhCl2 planes. The metals are nonbonding. Synthesis and reactions First prepared by Walter Hieber, it is typically prepared by treating hydrated rhodium trichloride with flowing carbon monoxide, according to this idealized redox equation: :2 RhCl3(H2O)3 + 6 CO → Rh2Cl2(CO)4 + 2 COCl2 + 6 H2O.McCleverty, J. A.; Wilkinson, G. "Dichlorotetracarbonyldirhodium (rhodium carbonyl chloride)" Inorganic Syntheses 1966, volume 8, pp. 211-14. The complex reacts with triph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organorhodium Compound
Organorhodium chemistry is the chemistry of organometallic compounds containing a rhodium-carbon chemical bond, and the study of rhodium and rhodium compounds as catalysts in organic reactions. Stable organorhodium compounds and transient organorhodium intermediates are used as catalyst such as in olefin hydroformylation, olefin hydrogenation, olefin isomerization and the Monsanto process Classification based on principal oxidation states Organometallic rhodium compounds share many characteristics with those of iridium, but less so with cobalt. Rhodium can exist in oxidation states of -III to +V, but rhodium(I) and rhodium(III) are the more common. Rhodium(I) compounds (d8 configuration) usually occur with square planar or trigonal bipyramidal geometries, while rhodium (III) compounds (d6 configuration) typically have an octahedral geometry. Rhodium(0) Rhodium(0) complexes are binary carbonyls, the principal examples being tetrarhodium dodecacarbonyl, Rh4(CO)12, and hexadecacarb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylacetone
Acetylacetone is an organic compound with the chemical formula . It is a colorless liquid, classified as a 1,3-diketone. It exists in equilibrium with a tautomer . These tautomers interconvert so rapidly under most conditions that they are treated as a single compound in most applications. It is a colorless liquid that is a precursor to acetylacetonate anion (commonly abbreviated acac−), a bidentate ligand. It is also a building block for the synthesis of heterocyclic compounds. Properties Tautomerism The keto and enol tautomers of acetylacetone coexist in solution. The enol form has C2v symmetry, meaning the hydrogen atom is shared equally between the two oxygen atoms. In the gas phase, the equilibrium constant, ''K''keto→enol, is 11.7, favoring the enol form. The two tautomeric forms can be distinguished by NMR spectroscopy, IR spectroscopy and other methods. The equilibrium constant tends to be high in nonpolar solvents; when k = >1, the enol form is favour ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl Complexes
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound. The term carbonyl can also refer to carbon monoxide as a ligand in an inorganic or organometallic complex (a metal carbonyl, e.g. nickel carbonyl). The remainder of this article concerns itself with the organic chemistry definition of carbonyl, where carbon and oxygen share a double bond. Carbonyl compounds In organic chemistry, a carbonyl group characterizes the following types of compounds: Other organic carbonyls are urea and the carbamates, the derivatives of acyl chlorides chloroformates and phosgene, carbonate esters, thioesters, lactones, lactams, hydroxamates, and isocyanates. Examples of inorganic carbonyl compounds are carbon dioxide and carbonyl sulfide. A sp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimers (chemistry)
Dimer may refer to: * Dimer (chemistry), a chemical structure formed from two similar sub-units ** Protein dimer, a protein quaternary structure ** d-dimer * Dimer model, an item in statistical mechanics, based on ''domino tiling'' * Julius Dimer Julius Dimer (1 August 1871 – 20 October 1945) was a German chess master. At the beginning of his career, he played in several mini tournaments (''Quadrangular'') in Germany; at Altona 1897, Elmshorn 1898, Munich 1900, Kiel 1901, Hamburg 1903, ... (1871–1945), German chess master See also * Dimery (botany), having two parts in a distinct whorl of a plant structure * Di (other), a prefix * Dymer (other) * -mer, a suffix * Oligomer * Peierls transition, sometimes called dimerization {{Disambiguation, surname ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homogeneous Catalysis
In chemistry, homogeneous catalysis is catalysis by a soluble catalyst in a solution. Homogeneous catalysis refers to reactions where the catalyst is in the same phase as the reactants, principally in solution. In contrast, heterogeneous catalysis describes processes where the catalysts and substrate are in distinct phases, typically solid-gas, respectively. The term is used almost exclusively to describe solutions and implies catalysis by organometallic compounds. Homogeneous catalysis is an established technology that continues to evolve. An illustrative major application is the production of acetic acid. Enzymes are examples of homogeneous catalysts. Examples Acid catalysis The proton is a pervasive homogeneous catalyst because water is the most common solvent. Water forms protons by the process of self-ionization of water. In an illustrative case, acids accelerate (catalyze) the hydrolysis of esters: :CH3CO2CH3 + H2O CH3CO2H + CH3OH At neutral pH, aqueous solutions of most e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
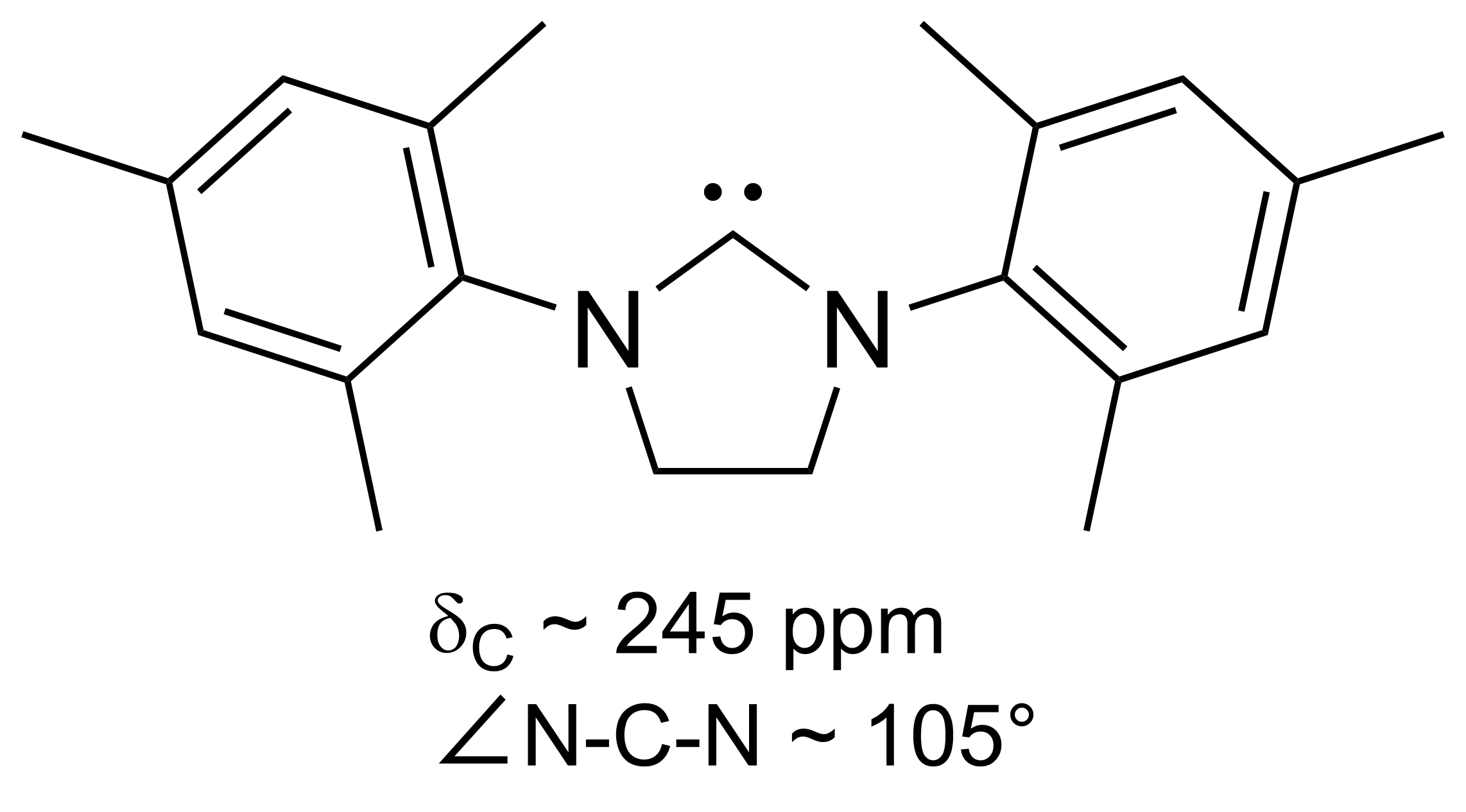
N-heterocyclic Carbene
A persistent carbene (also known as stable carbene) is a type of carbene demonstrating particular stability. The best-known examples and by far largest subgroup are the ''N''-heterocyclic carbenes (NHC) (sometimes called Arduengo carbenes), for example diaminocarbenes with the general formula (R2N)2C:, where the four R moieties are typically alkyl and aryl groups. The groups can be linked to give heterocyclic carbenes, such as those derived from imidazole, imidazoline, thiazole or triazole. Traditionally carbenes are viewed as so reactive that were only studied indirectly, such as by trapping reactions. This situation has changed dramatically with the emergence of persistent carbenes. Although they are fairly reactive substances, undergoing dimerization, many can be isolated as pure substances. Persistent carbenes tend to exist in the singlet. Their stability is only partly due to steric hindrance by bulky groups. Some singlet carbenes are thermodynamically stable and can be iso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrahydrothiophene
Tetrahydrothiophene is an organosulfur compound with the formula (CH2)4S. The molecule consists of a five-membered saturated ring with four methylene groups and a sulfur atom. It is the saturated analog of thiophene. It is a volatile, colorless liquid with an intensely unpleasant odor. It is also known as thiophane, thiolane, or THT. While THT is not particularly common, the vitamin biotin is essential for life in aerobic organisms. Synthesis and reactions Tetrahydrothiophene is prepared by the reaction of tetrahydrofuran with hydrogen sulfide. This vapor-phase reaction is catalyzed by alumina and other heterogenous acid catalysts. This compound is a ligand in coordination chemistry, an example being the complex chloro(tetrahydrothiophene)gold(I). Oxidation of THT gives the sulfone sulfolane, which is of interest as a polar, odorless solvent: : Sulfolane is, however, more conventionally prepared from butadiene. Natural occurrence Both unsubstituted and substituted tetra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dicarbonyl(acetylacetonato)rhodium(I)
Dicarbonyl(acetylacetonato)rhodium(I) is an organorhodium compound with the formula Rh(O2C5H7)(CO)2. The compound consists of two CO ligands and an acetylacetonate. It is a dark green solid that dissolves in acetone and benzene, giving yellow solutions. The compound is used as a precursor to homogeneous catalysts. It is prepared by treating rhodium carbonyl chloride with sodium acetylacetonate in the presence of base: : CO)2RhClsub>2 + 2 NaO2C5H7 → 2 Rh(O2C5H7)(CO)2 + 2 NaCl The complex adopts square planar molecular geometry The square planar molecular geometry in chemistry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the corne .... The molecules stack with Rh---Rh distances of about 326 pm. As such, it is representative of a linear chain compound. References {{Acetylacetonate complexes Organorhodium compounds Homogeneo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bis(triphenylphosphine)rhodium Carbonyl Chloride
Bis(triphenylphosphine)rhodium carbonyl chloride is the organorhodium complex with the formula [RhCl(CO)(PPh3)2]. This complex of rhodium(I) is a bright yellow, air-stable solid. It is the Rh analogue of Vaska's complex, the corresponding iridium complex. With regards to its structure, the complex is square planar with mutually ''trans'' triphenylphosphine (PPh3) ligands. The complex is a versatile homogeneous catalyst. Synthesis and reactions Bis(triphenylphosphine)rhodium carbonyl chloride was originally prepared by treating rhodium carbonyl chloride, Rh2Cl2(CO)4 with triphenylphosphine. However, it is typically produced by the carbonylation of Wilkinson's catalyst:{{cite book , author=D. Evans , author2=J. A. Osborn , author3=G. Wilkinson, doi=10.1002/9780470132593.ch18, title=''trans''‐Carbonylchlorobis(Triphenylphosphine)Rhodium and Related Complexes, chapter=Trans -Carbonylchlorobis(Triphenyl-Phosphine)Rhodium and Related Complexes, volume=28, series=Inorganic Synthese ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl Complex
Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. Metal carbonyls are useful in organic synthesis and as catalysts or catalyst precursors in homogeneous catalysis, such as hydroformylation and Reppe chemistry. In the Mond process, nickel tetracarbonyl is used to produce pure nickel. In organometallic chemistry, metal carbonyls serve as precursors for the preparation of other organometallic complexes. Metal carbonyls are toxic by skin contact, inhalation or ingestion, in part because of their ability to carbonylate hemoglobin to give carboxyhemoglobin, which prevents the binding of oxygen. Nomenclature and terminology The nomenclature of the metal carbonyls depends on the charge of the complex, the number and type of central atoms, and the number and type of ligands and their binding modes. They occur as neutral complexes, as positively-charged metal carbonyl cations or as negatively charged metal carbonylates. The carbon monoxide ligan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |