|
Q Value (nuclear Science)
In nuclear physics and chemistry, the value for a reaction is the amount of energy absorbed or released during the nuclear reaction. The value relates to the enthalpy of a chemical reaction or the energy of radioactive decay products. It can be determined from the masses of reactants and products. values affect reaction rates. In general, the larger the positive value for the reaction, the faster the reaction proceeds, and the more likely the reaction is to "favor" the products. : Q = (\,m_\text - m_\text\,) \times \text where the masses are in atomic mass units. Also both \;m_\text\; and \;m_\text\; are the sums of the reactant and product masses respectively. Definition The conservation of energy, between the initial and final energy of a nuclear process \text E_\text = E_\text \text enables the general definition of based on the mass–energy equivalence. For any radioactive particle decay, the kinetic energy difference will be given by: : Q = K_\text - K_\text = (\ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Physics
Nuclear physics is the field of physics that studies atomic nuclei and their constituents and interactions, in addition to the study of other forms of nuclear matter. Nuclear physics should not be confused with atomic physics, which studies the atom as a whole, including its electrons. Discoveries in nuclear physics have led to applications in many fields. This includes nuclear power, nuclear weapons, nuclear medicine and magnetic resonance imaging, industrial and agricultural isotopes, ion implantation in materials engineering, and radiocarbon dating in geology and archaeology. Such applications are studied in the field of nuclear engineering. Particle physics evolved out of nuclear physics and the two fields are typically taught in close association. Nuclear astrophysics, the application of nuclear physics to astrophysics, is crucial in explaining the inner workings of stars and the origin of the chemical elements. History The history of nuclear physics as a di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behave similarly within the nucleus, and each has a mass of approximately one atomic mass unit, they are both referred to as nucleons. Their properties and interactions are described by nuclear physics. Protons and neutrons are not elementary particles; each is composed of three quarks. The chemical properties of an atom are mostly determined by the configuration of electrons that orbit the atom's heavy nucleus. The electron configuration is determined by the charge of the nucleus, which is determined by the number of protons, or atomic number. The number of neutrons is the neutron number. Neutrons do not affect the electron configuration, but the sum of atomic and neutron numbers is the mass of the nucleus. Atoms of a chemical element t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
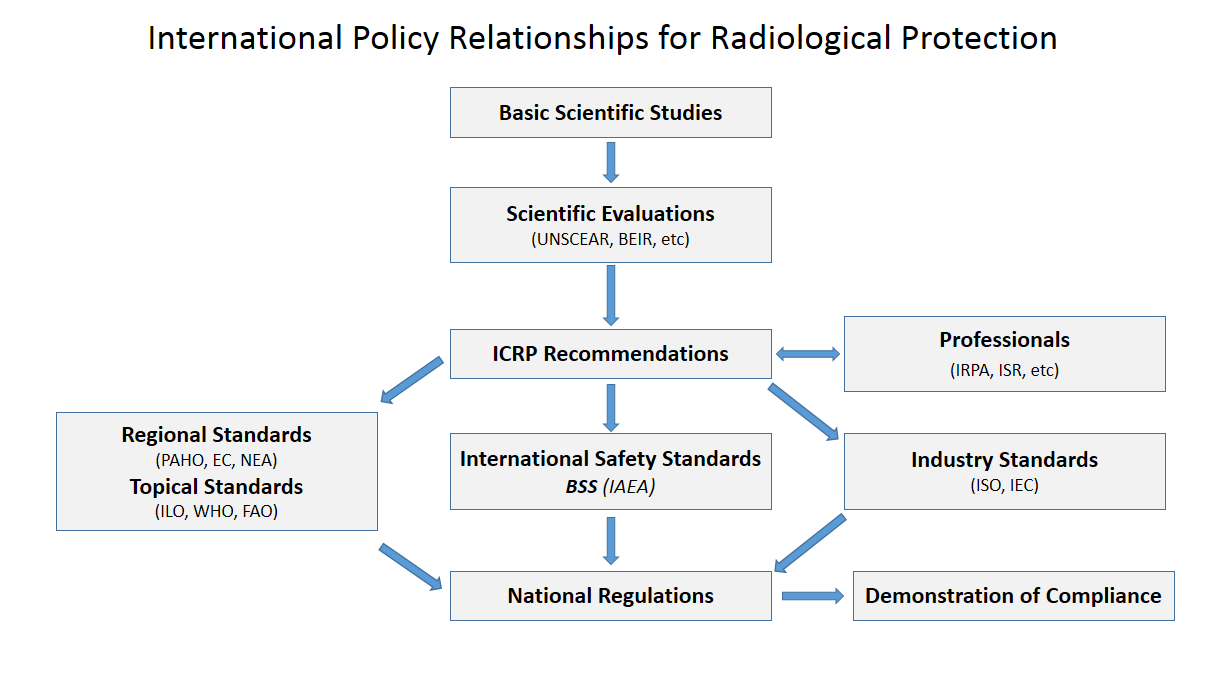
IAEA
The International Atomic Energy Agency (IAEA) is an intergovernmental organization that seeks to promote the peaceful use of nuclear energy and to inhibit its use for any military purpose, including nuclear weapons. It was established in 1957 as an autonomous organization within the United Nations system; though governed by its own founding treaty, the organization reports to both the General Assembly and the Security Council of the United Nations, and is headquartered at the UN Office at Vienna, Austria. The IAEA was created in response to growing international concern toward nuclear weapons, especially amid rising tensions between the foremost nuclear powers, the United States and the Soviet Union. U.S. President Dwight D. Eisenhower's " Atoms for Peace" speech, which called for the creation of an international organization to monitor the global proliferation of nuclear resources and technology, is credited with catalyzing the formation of the IAEA, whose treaty came int ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pandemonium Effect
The pandemonium effect is a problem that may appear when high resolution detectors (usually germanium detectors) are used in beta decay studies. It can affect the correct determination of the feeding to the different levels of the daughter nucleus. It was first introduced in 1977. Context Typically, when a parent nucleus beta-decays into its daughter, there is some final energy available which is shared between the final products of the decay. This is called the ''Q'' value of the beta decay (''Qβ''). The daughter nucleus doesn't necessarily end up in the ground state after the decay, this only happens when the other products have taken all the available energy with them (usually as kinetic energy). So, in general, the daughter nucleus keeps an amount of the available energy as excitation energy and ends up in an excited state associated to some energy level, as seen in the picture. The daughter nucleus can only stay in that excited state for a small amount of time (the ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fusion Energy Gain Factor
A fusion energy gain factor, usually expressed with the symbol ''Q'', is the ratio of fusion power produced in a nuclear fusion reactor to the power required to maintain the plasma in steady state. The condition of ''Q'' = 1, when the power being released by the fusion reactions is equal to the required heating power, is referred to as breakeven, or in some sources, scientific breakeven. The energy given off by the fusion reactions may be captured within the fuel, leading to ''self-heating''. Most fusion reactions release at least some of their energy in a form that cannot be captured within the plasma, so a system at ''Q'' = 1 will cool without external heating. With typical fuels, self-heating in fusion reactors is not expected to match the external sources until at least ''Q'' ≈ 5. If ''Q'' increases past this point, increasing self-heating eventually removes the need for external heating. At this point the reaction becomes self-sustaining, a condition called combustion, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Decay Energy
The decay energy is the energy change of a nucleus having undergone a radioactive decay. Radioactive decay is the process in which an unstable atomic nucleus loses energy by emitting ionizing particles and radiation. This decay, or loss of energy, results in an atom of one type (called the parent nuclide) transforming to an atom of a different type (called the daughter nuclide). Decay calculation The energy difference of the reactants is often written as ''Q'': :Q = \left( \text \right)_\text - \left( \text \right)_\text, :Q = \left(\text \right)_ c^2 - \left( \text \right )_\text c^2 . Decay energy is usually quoted in terms of the energy units MeV (million electronvolts) or keV (thousand electronvolts): : Q \text = -931.5 \Delta M \text,~~(\text\Delta M = \Sigma M_\text - \Sigma M_\text). Types of radioactive decay include * gamma ray * beta decay (decay energy is divided between the emitted electron and the neutrino which is emitted at the same time) * alpha decay The dec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calorimeter (particle Physics)
In particle physics, a calorimeter is an experimental apparatus that measures the energy of particles. Most particles enter the calorimeter and initiate a particle shower and the particles' energy is deposited in the calorimeter, collected, and measured. The energy may be measured in its entirety, requiring total containment of the particle shower, or it may be sampled. Typically, calorimeters are segmented transversely to provide information about the direction of the particle or particles, as well as the energy deposited, and longitudinal segmentation can provide information about the identity of the particle based on the shape of the shower as it develops. Calorimetry design is an active area of research in particle physics. Types of calorimeters Electromagnetic versus hadronic An electromagnetic calorimeter is one specifically designed to measure the energy of particles that interact primarily via the electromagnetic interaction, while a hadronic calorimeter is one designed t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Binding Energy
In physics and chemistry, binding energy is the smallest amount of energy required to remove a particle from a system of particles or to disassemble a system of particles into individual parts. In the former meaning the term is predominantly used in condensed matter physics, atomic physics, and chemistry, whereas in nuclear physics the term '' separation energy'' is used. A bound system is typically at a lower energy level than its unbound constituents. According to relativity theory, a decrease in the total energy of a system is accompanied by a decrease in the total mass, where . Types of binding energy There are several types of binding energy, each operating over a different distance and energy scale. The smaller the size of a bound system, the higher its associated binding energy. Mass–energy relation A bound system is typically at a lower energy level than its unbound constituents because its mass must be less than the total mass of its unbound constituents. For s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
KATRIN
Katrin is a feminine given name. It is a German and Swedish contracted form of Katherine. Katrin may refer to: Sports * Katrin Apel (born 1973), German biathlete *Katrin Beinroth (born 1981), German judoka *Katrin Borchert (born 1969), German-born Australian sprint canoer * Katrín Davíðsdóttir (born 1993), Icelandic CrossFit athlete *Katrin Dörre-Heinig (born 1961), German long-distance runner *Katrin Engel (born 1984), Austrian handball player *Katrin Green (born 1985), German Paralympian track and field athlete *Katrin Käärt (born 1983), Estonian athletics sprinter *Katrin Kauschke (born 1971), German field hockey player *Katrin Kieseler, German-born, Australian sprint canoer *Katrin Kliehm (born 1981), German football player * Katrin Krabbe (born 1969), German athlete * Katrin Krüger (born 1959), German handball player * Katrin Loo (born 1991), Estonian footballer *Katrin Mattscherodt (born 1981), German long track speed skater *Katrin Meissner (born 1973), German f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron's mass is approximately 1/1836 that of the proton. Quantum mechanical properties of the electron include an intrinsic angular momentum ( spin) of a half-integer value, expressed in units of the reduced Planck constant, . Being fermions, no two electrons can occupy the same quantum state, per the Pauli exclusion principle. Like all elementary particles, electrons exhibit properties of both particles and waves: They can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Antineutrino
The electron neutrino () is an elementary particle which has zero electric charge and a spin of . Together with the electron, it forms the first generation of leptons, hence the name electron neutrino. It was first hypothesized by Wolfgang Pauli in 1930, to account for missing momentum and missing energy in beta decay, and was discovered in 1956 by a team led by Clyde Cowan and Frederick Reines (see Cowan–Reines neutrino experiment). Proposal In the early 1900s, theories predicted that the electrons resulting from beta decay should have been emitted at a specific energy. However, in 1914, James Chadwick showed that electrons were instead emitted in a continuous spectrum. : → + :The early understanding of beta decay In 1930, Wolfgang Pauli theorized that an undetected particle was carrying away the observed difference between the energy, momentum, and angular momentum of the initial and final particles.Niels Bohr was notably opposed to this interpretation of beta de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ratio). Protons and neutrons, each with masses of approximately one atomic mass unit, are jointly referred to as "nucleons" (particles present in atomic nuclei). One or more protons are present in the Atomic nucleus, nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each chemical element, element has a unique number of protons, each element has its own unique atomic number, which determines the number of atomic electrons and consequently the chemical characteristics of the element. The word ''proton'' is Greek language, G ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |