|
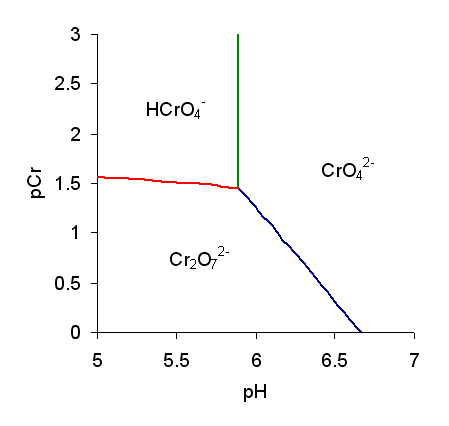
Predominance Diagram
A predominance diagram purports to show the conditions of concentration and pH where a chemical species has the highest concentration in solutions in which there are multiple acid-base equilibria. The lines on a predominance diagram indicate where adjacent species have the same concentration. Either side of such a line one species or the other predominates, that is, has higher concentration relative to the other species. To illustrate a predominance diagram, part of the one for chromate is shown at the right. pCr stands for minus the logarithm of the chromium concentration and pH stands for minus the logarithm of the hydrogen ion concentration. There are two independent equilibria, with equilibrium constants defined as follows. A third equilibrium constant can be derived from ''K''1 and ''K''D. The species and are only formed at very low pH so they do not appear on this diagram. Published values for log ''K''1 and log ''K''D are 5.89 and 2.05, respectively. Using these values ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, using the s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Equilibrium
In a chemical reaction, chemical equilibrium is the state in which both the reactants and products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the system. This state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but they are equal. Thus, there are no net changes in the concentrations of the reactants and products. Such a state is known as dynamic equilibrium. Historical introduction The concept of chemical equilibrium was developed in 1803, after Berthollet found that some chemical reactions are reversible. For any reaction mixture to exist at equilibrium, the rates of the forward and backward (reverse) reactions must be equal. In the following chemical equation, arrows point both ways to indicate equilibrium. A and B are reactant chemical species, S and T are p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Predominance Diagram Cr
''Predominance'' is the debut album by the Norwegian thrash metal band Susperia. Produced at the Abyss Studios, it combines thrash metal with black metal. Track listing #"I Am Pain" – 4:34 #"Vainglory" – 4:35 #"Illusions of Evil" – 5:44 #"Specimen" – 3:57 #"Journey into Black" – 3:50 #"Of Hate We Breed" – 4:57 #"Objects of Desire" – 4:03 #"The Hellchild" – 4:43 #"Blood on My Hands" – 5:14 #"The Coming of a Darker Time" – 3:34 Personnel * Athera – vocals *Cyrus – lead guitar *Elvorn – rhythm guitar *Memnock – bass *Tjodalv Tjodalv (born Ian Kenneth Åkesson in 1976) is a Norwegian drummer. He is a founding member of both Dimmu Borgir and Old Man's Child. He now plays in Black Comedy and Susperia. Tjodalv played guitar on the first Dimmu Borgir albums ''Inn i evig ... – drums References {{Authority control 2001 debut albums Susperia albums ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Ion
A hydrogen ion is created when a hydrogen atom loses or gains an electron. A positively charged hydrogen ion (or proton) can readily combine with other particles and therefore is only seen isolated when it is in a gaseous state or a nearly particle-free space. Due to its extremely high charge density of approximately 2×1010 times that of a sodium ion, the bare hydrogen ion cannot exist freely in solution as it readily hydrates, i.e., bonds quickly. The hydrogen ion is recommended by IUPAC as a general term for all ions of hydrogen and its isotopes. Depending on the charge of the ion, two different classes can be distinguished: positively charged ions and negatively charged ions. Cation (positively charged) A hydrogen atom is made up of a nucleus with charge +1, and a single electron. Therefore, the only positively charged ion possible has charge +1. It is noted H+. Depending on the isotope in question, the hydrogen cation has different names: * Hydron: general name referri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium Constants
The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical equilibrium, a state approached by a dynamic chemical system after sufficient time has elapsed at which its composition has no measurable tendency towards further change. For a given set of reaction conditions, the equilibrium constant is independent of the initial analytical concentrations of the reactant and product species in the mixture. Thus, given the initial composition of a system, known equilibrium constant values can be used to determine the composition of the system at equilibrium. However, reaction parameters like temperature, solvent, and ionic strength may all influence the value of the equilibrium constant. A knowledge of equilibrium constants is essential for the understanding of many chemical systems, as well as biochemical processes such as oxygen transport by hemoglobin in blood and acid–base homeostasis in the human body. Stability constants, formation con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vanadate
In chemistry, a vanadate is an anionic coordination complex of vanadium. Often vanadate refers to oxoanions of vanadium, most of which exist in its highest oxidation state of +5. The complexes and are referred to as hexacyanovanadate(III) and nonachlorodivanadate(III), respectively. A simple vanadate ion is the tetrahedral orthovanadate anion, (which is also called vanadate(V)), which is present in e.g. sodium orthovanadate and in solutions of in strong base ( pH > 13). Conventionally this ion is represented with a single double bond, however this is a resonance form as the ion is a regular tetrahedron with four equivalent oxygen atoms. Additionally a range of polyoxovanadate ions exist which include discrete ions and "infinite" polymeric ions. There are also vanadates, such as rhodium vanadate, , which has a statistical rutile structure where the and ions randomly occupy the positions in the rutile lattice, that do not contain a lattice of cations and balancing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdate
In chemistry a molybdate is a compound containing an oxoanion with molybdenum in its highest oxidation state of 6. Molybdenum can form a very large range of such oxoanions which can be discrete structures or polymeric extended structures, although the latter are only found in the solid state. The larger oxoanions are members of group of compounds termed polyoxometalates, and because they contain only one type of metal atom are often called isopolymetalates. The discrete molybdenum oxoanions range in size from the simplest , found in potassium molybdate up to extremely large structures found in isopoly-molybdenum blues that contain for example 154 Mo atoms. The behaviour of molybdenum is different from the other elements in group 6. Chromium only forms the chromates, , , and ions which are all based on tetrahedral chromium. Tungsten is similar to molybdenum and forms many tungstates containing 6 coordinate tungsten. Examples of molybdate anions Examples of molybdate oxoanion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tungstate
In chemistry, a tungstate is a compound that contains an oxyanion of tungsten or is a mixed oxide containing tungsten. The simplest tungstate ion is , "orthotungstate". Many other tungstates belong to a large group of polyatomic ions that are termed polyoxometalates, ("POMs"), and specifically termed isopolyoxometalates as they contain, along with oxygen and maybe hydrogen, only one other element. Almost all useful tungsten ores are tungstates. Structures Orthotungstates feature tetrahedral W(VI) centres with short W–O distances of 1.79 Å. Structurally, they resemble sulfates. Six-coordinate, octahedral tungsten dominates in the polyoxotungstates. In these compounds, the W–O distances are elongated. Some examples of tungstate ions: * (hydrogentungstate) * polymeric ions of various structures in , and Wells A.F. (1984) ''Structural Inorganic Chemistry'' 5th edition Oxford Science Publications * (paratungstate A) * (tungstate Y)Jon A. McCleverty, N. G. Connelly,Nom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Equilibrium Chemistry
Equilibrium chemistry is concerned with systems in chemical equilibrium. The unifying principle is that the free energy of a system at equilibrium is the minimum possible, so that the slope of the free energy with respect to the reaction coordinate is zero. This principle, applied to mixtures at equilibrium provides a definition of an equilibrium constant. Applications include acid–base, host–guest, metal–complex, solubility, partition, chromatography and redox equilibria. Thermodynamic equilibrium A chemical system is said to be in equilibrium when the quantities of the chemical entities involved do not and ''cannot'' change in time without the application of an external influence. In this sense a system in chemical equilibrium is in a stable state. The system at chemical equilibrium will be at a constant temperature, pressure or volume and a composition. It will be insulated from exchange of heat with the surroundings, that is, it is a closed system. A change of te ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |