|
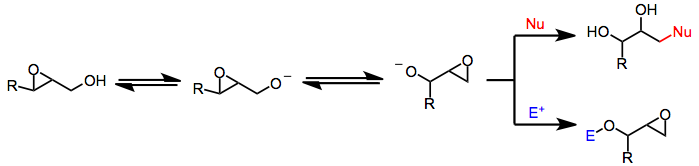
Payne Rearrangement
The Payne rearrangement is the Base-promoted epoxide isomerization, isomerization, under basic conditions, of 2,3-epoxy alcohols to isomeric 1,2-epoxy alcohols with inversion of configuration. Aza- and thia-Payne rearrangements of aziridines and thiiraniums, respectively, are also known. Introduction Under basic, protic conditions, 2,3-epoxy alcohols undergo a rearrangement in which the alcohol oxygen opens the epoxide with inversion of configuration, forming an isomeric 1,2-epoxy alcohol. Overall, the Payne rearrangement represents a migration of the epoxide. Although the migration itself is fully reversible, nucleophilic opening under Curtin–Hammett conditions provides good yields of functionalized diols derived from a single epoxy alcohol isomer. Intramolecular electrophilic trapping of the new alkoxide generated upon rearrangement may also be used to drive the reaction to completion. In some cases, the thermodynamic difference between epoxide isomers is large enough to obtain ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Base-promoted Epoxide Isomerization
Base-promoted epoxide isomerization is the conversion of Alkane, alkyl epoxides to Cyclic compound#Ring-closing & opening reactions, ring-opened products through the action of strong Base (chemistry), base. Isomerizations of this type are most often used to synthesize allylic alcohols, although other products are possible. Introduction In the presence of lithium or aluminum amide bases, epoxides may open to give the corresponding allylic alcohols. Removal of a proton adjacent to the epoxide, elimination, and neutralization of the resulting alkoxide lead to synthetically useful allylic alcohol products. In reactions of chiral, non-racemic epoxides, the configuration of the allylic alcohol product matches that of the epoxide substrate at the carbon whose C–O bond does not break (the starred carbon below). Besides Dehydrohalogenation, β-elimination some other reactions are possible, as metalation of the epoxide ring can take place competitively. Vinylogy, Vinylogous eliminations ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzyl Bromide
Benzyl bromide is an organic compound with the formula . The molecule consists of a benzene ring substituted with a bromomethyl group. It is a colorless liquid with lachrymatory properties. The compound is a reagent for introducing benzyl groups. Synthesis and structure Benzyl bromide can be synthesized by the bromination of toluene under conditions suitable for a free radical halogenation: : The structure has been examined by electron diffraction. Applications Benzyl bromide is used in organic synthesis for the introduction of the benzyl groups when the less expensive benzyl chloride is insufficiently reactive. Benzylations are often achieved in the presence of catalytic amounts of sodium iodide, which generates the more reactive benzyl iodide in situ. In some cases, benzyl serves as protecting group for alcohols and carboxylic acids. Safety Benzyl bromide is a strong lachrymator and is also intensely irritating to skin and mucous membranes. Because of these properties, it ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlorides, such as extraordinary solubility in polar solvents (83.05 g/100 mL of water at 20 °C) and its hygroscopic properties. Chemical properties The salt forms crystalline hydrates, unlike the other alkali metal chlorides. Mono-, tri-, and pentahydrates are known. The anhydrous salt can be regenerated by heating the hydrates. LiCl also absorbs up to four equivalents of ammonia/mol. As with any other ionic chloride, solutions of lithium chloride can serve as a source of chloride ion, e.g., forming a precipitate upon treatment with silver nitrate: : LiCl + AgNO3 → AgCl + LiNO3 Preparation Lithium chloride is produced by treatment of lithium carbonate with hydrochloric acid. Anhydrous LiCl is prepared from the hydrate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |