|
Protactinium Carbide
Protactinium (formerly protoactinium) is a chemical element with the symbol Pa and atomic number 91. It is a dense, silvery-gray actinide metal which readily reacts with oxygen, water vapor and inorganic acids. It forms various chemical compounds in which protactinium is usually present in the oxidation state +5, but it can also assume +4 and even +3 or +2 states. Concentrations of protactinium in the Earth's crust are typically a few parts per trillion, but may reach up to a few parts per million in some uraninite ore deposits. Because of its scarcity, high radioactivity and high toxicity, there are currently no uses for protactinium outside scientific research, and for this purpose, protactinium is mostly extracted from spent nuclear fuel. The element was first identified in 1913 by Kazimierz Fajans and Oswald Helmuth Göhring and named ''brevium'' because of the short half-life of the specific isotope studied, i.e. protactinium-234. A more stable isotope of protactinium, 231Pa, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler substances by any chemical reaction. The number of protons in the nucleus is the defining property of an element, and is referred to as its atomic number (represented by the symbol ''Z'') – all atoms with the same atomic number are atoms of the same element. Almost all of the baryonic matter of the universe is composed of chemical elements (among rare exceptions are neutron stars). When different elements undergo chemical reactions, atoms are rearranged into new compounds held together by chemical bonds. Only a minority of elements, such as silver and gold, are found uncombined as relatively pure native element minerals. Nearly all other naturally occurring elements occur in the Earth as compounds or mixtures. Air is primarily a m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Precursor (chemistry)
In chemistry, a precursor is a compound that participates in a chemical reaction that produces another compound. In biochemistry, the term "precursor" often refers more specifically to a chemical compound preceding another in a metabolic pathway, such as a protein precursor. Illicit drug precursors In 1988, the United Nations Convention Against Illicit Traffic in Narcotic Drugs and Psychotropic Substances introduced detailed provisions and requirements relating the control of precursors used to produce drugs of abuse. In Europe the Regulation (EC) No. 273/2004 of the European Parliament and of the Council on drug precursors was adopted on 11 February 2004. ( European law on drug precursors) Illicit explosives precursors On January 15, 2013, the Regulation (EU) No. 98/2013 of the European Parliament and of the Council on the marketing and use of explosives precursors was adopted. The Regulation harmonises rules across Europe on the making available, introduction, possession a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiometric Dating
Radiometric dating, radioactive dating or radioisotope dating is a technique which is used to date materials such as rocks or carbon, in which trace radioactive impurities were selectively incorporated when they were formed. The method compares the abundance of a naturally occurring radioactive isotope within the material to the abundance of its decay products, which form at a known constant rate of decay. The use of radiometric dating was first published in 1907 by Bertram Boltwood and is now the principal source of information about the absolute age of rocks and other geological features, including the age of fossilized life forms or the age of Earth itself, and can also be used to date a wide range of natural and man-made materials. Together with stratigraphic principles, radiometric dating methods are used in geochronology to establish the geologic time scale. Among the best-known techniques are radiocarbon dating, potassium–argon dating and uranium–lead dating. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from nuclear fission is passed to a working fluid (water or gas), which in turn runs through steam turbines. These either drive a ship's propellers or turn electrical generators' shafts. Nuclear generated steam in principle can be used for industrial process heat or for district heating. Some reactors are used to produce isotopes for Nuclear medicine, medical and industrial radiography, industrial use, or for production of weapons-grade plutonium. , the International Atomic Energy Agency reports there are 422 nuclear power reactors and 223 nuclear research reactors in operation around the world. In the early era of nuclear reactors (1940s), a reactor was known as a nuclear pile or atomic pile (so-called because the graphite moderator blocks of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-233
Uranium-233 (233U or U-233) is a fissile isotope of uranium that is bred from thorium-232 as part of the thorium fuel cycle. Uranium-233 was investigated for use in nuclear weapons and as a reactor fuel. It has been used successfully in experimental nuclear reactors and has been proposed for much wider use as a nuclear fuel. It has a half-life of 160,000 years. Uranium-233 is produced by the neutron irradiation of thorium-232. When thorium-232 absorbs a neutron, it becomes thorium-233, which has a half-life of only 22 minutes. Thorium-233 decays into protactinium-233 through beta decay. Protactinium-233 has a half-life of 27 days and beta decays into uranium-233; some proposed molten salt reactor designs attempt to physically isolate the protactinium from further neutron capture before beta decay can occur, to maintain the neutron economy (if it misses the 233U window, the next fissile target is 235U, meaning a total of 4 neutrons needed to trigger fission). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thorium
Thorium is a weakly radioactive metallic chemical element with the symbol Th and atomic number 90. Thorium is silvery and tarnishes black when it is exposed to air, forming thorium dioxide; it is moderately soft and malleable and has a high melting point. Thorium is an electropositive actinide whose chemistry is dominated by the +4 oxidation state; it is quite reactive and can ignite in air when finely divided. All known thorium isotopes are unstable. The most stable isotope, 232Th, has a half-life of 14.05 billion years, or about the age of the universe; it decays very slowly via alpha decay, starting a decay chain named the thorium series that ends at stable 208 Pb. On Earth, thorium and uranium are the only significantly radioactive elements that still occur naturally in large quantities as primordial elements. Thorium is estimated to be over three times as abundant as uranium in the Earth's crust, and is chiefly refined from monazite sands as a by-product o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-238
Uranium-238 (238U or U-238) is the most common isotope of uranium found in nature, with a relative abundance of 99%. Unlike uranium-235, it is non-fissile, which means it cannot sustain a chain reaction in a thermal-neutron reactor. However, it is fissionable by fast neutrons, and is ''fertile'', meaning it can be transmuted to fissile plutonium-239. 238U cannot support a chain reaction because inelastic scattering reduces neutron energy below the range where fast fission of one or more next-generation nuclei is probable. Doppler broadening of 238U's neutron absorption resonances, increasing absorption as fuel temperature increases, is also an essential negative feedback mechanism for reactor control. Around 99.284% of natural uranium's mass is uranium-238, which has a half-life of 1.41 seconds (4.468 years, or 4.468 billion years). Due to its natural abundance and half-life relative to other radioactive elements, 238U produces ~40% of the radioactive heat produc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
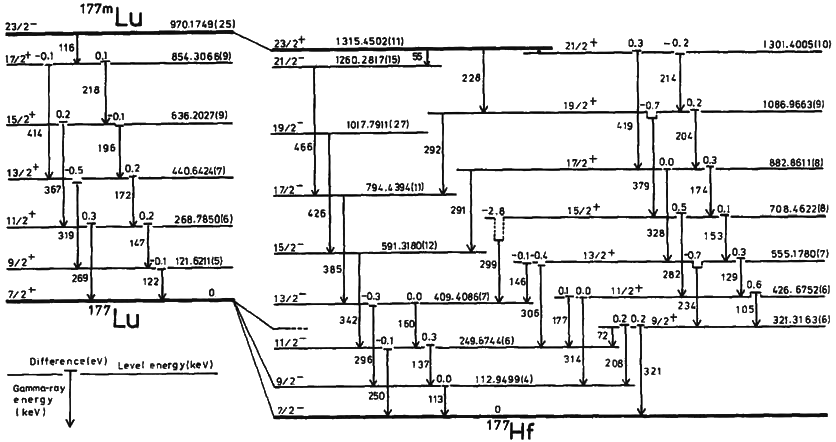
Nuclear Isomer
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have half-lives 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). The term "metastable" is usually restricted to isomers with half-lives of 10−9 seconds or longer. Some references recommend 5 × 10−9 seconds to distinguish the metastable half life from the normal "prompt" gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the nuclear isomer survives so long (at least 1015 years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-235
Uranium-235 (235U or U-235) is an Isotopes of uranium, isotope of uranium making up about 0.72% of natural uranium. Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. It is the only fissile isotope that exists in nature as a primordial nuclide. Uranium-235 has a half-life of 703.8 million years. It was discovered in 1935 by Arthur Jeffrey Dempster. Its fission cross section for slow thermal neutrons is about 584.3±1 Barn (unit), barns. For fast neutrons it is on the order of 1 barn. Most but not all neutron absorptions result in fission; a minority result in neutron capture forming uranium-236. Natural decay chain :\begin \ce \begin \ce \\ \ce \end \ce \\ \ce \begin \ce \\ \ce \end \ce \end Fission properties The fission of one atom of uranium-235 releases () inside the reactor. That corresponds to 19.54 TJ/mole (unit), mol, or 83.14 TJ/kg. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
University Of Glasgow
, image = UofG Coat of Arms.png , image_size = 150px , caption = Coat of arms Flag , latin_name = Universitas Glasguensis , motto = la, Via, Veritas, Vita , mottoeng = The Way, The Truth, The Life , established = , type = Public research university Ancient university , endowment = £225.2 million , budget = £809.4 million , rector = Rita Rae, Lady Rae , chancellor = Dame Katherine Grainger , principal = Sir Anton Muscatelli , academic_staff = 4,680 (2020) , administrative_staff = 4,003 , students = () , undergrad = () , postgrad = () , city = Glasgow , country = Scotland, UK , colours = , website = , logo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
First World War
World War I (28 July 1914 11 November 1918), often abbreviated as WWI, was one of the deadliest global conflicts in history. Belligerents included much of Europe, the Russian Empire, the United States, and the Ottoman Empire, with fighting occurring throughout Europe, the Middle East, Africa, the Pacific, and parts of Asia. An estimated 9 million soldiers were killed in combat, plus another 23 million wounded, while 5 million civilians died as a result of military action, hunger, and disease. Millions more died in genocides within the Ottoman Empire and in the 1918 influenza pandemic, which was exacerbated by the movement of combatants during the war. Prior to 1914, the European great powers were divided between the Triple Entente (comprising France, Russia, and Britain) and the Triple Alliance (containing Germany, Austria-Hungary, and Italy). Tensions in the Balkans came to a head on 28 June 1914, following the assassination of Arch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ada Hitchins
Ada Florence Remfry Hitchins (26 June 1891 – 4 January 1972) was the principal research assistant of British chemist Frederick Soddy, who won the Nobel prize in 1921 for work on radioactive elements and the theory of isotopes. Hitchins isolated samples from uranium ores, taking precise and accurate measurements of atomic mass that provided the first experimental evidence for the existence of different isotopes. She also helped to discover the element protactinium, which Dmitri Mendeleev had predicted should occur in the periodic table between uranium and thorium. Education Ada Hitchins was born on 26 June 1891 in Tavistock, Devon, England, the daughter of William Hedley Hitchins, a supervisor of customs and excise, and his wife Annie Sarah Pearsons. The family lived for a time in Campbeltown, Scotland, where Hitchins attended high school, graduating in 1909. From there, she went to the University of Glasgow, obtaining her bachelor's degree in science, with honors, in 1913. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
%2C_black_and_white.png)