|
Peroxide Value
Detection of peroxide gives the initial evidence of rancidity in unsaturated fats and oils. Other methods are available, but peroxide value is the most widely used. It gives a measure of the extent to which an oil sample has undergone primary oxidation; extent of secondary oxidation may be determined from p-anisidine test.Chemistry And Technology Of Oils And FatChemistry And Technology Of Oils And Fats By Dr. M.M. Chakrabarty The double bonds found in fats and oils play a role in autoxidation. Oils with a high degree of Unsaturated fat, unsaturation are most susceptible to autoxidation. The best test for autoxidation (oxidative rancidity) is determination of the peroxide value. Peroxides are intermediates in the autoxidation reaction. Autoxidation is a Free radical addition, free radical reaction involving oxygen that leads to deterioration of fats and oils which form off-flavours and off-odours. Peroxide value, concentration of peroxide in an oil or fat, is useful for assessing t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined to each other and to adjacent elements through Single bond, single covalent bonds, denoted by dashes or lines. The group in a peroxide is often called the peroxide group, though some nomenclature discrepancies exist. This linkage is recognized as a common polyatomic ion, and exists in many molecules. General structure The characteristic structure of any regular peroxide is the oxygen–oxygen covalent single bond, which connects the two main atoms together. In the event that the molecule has no chemical Substituent, substituents, the peroxide group will have a [−2] Formal charge, net charge. Each oxygen atom has a charge of negative one, as 5 of its Valence electron, valence electrons remain in the outermost Atomic orbital, orbital ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox Titration
A redox titration is a type of titration based on a redox reaction between the analyte and titrant. It may involve the use of a redox indicator and/or a potentiometer. A common example of a redox titration is the treatment of a solution of iodine with a reducing agent to produce iodide using a starch indicator to help detect the endpoint. Iodine (I2) can be reduced to iodide (I−) by, say, thiosulfate (), and when all the iodine is consumed, the blue colour disappears. This is called an iodometry, iodometric titration. Most often, the reduction of iodine to iodide is the last step in a series of reactions where the initial reactions convert an unknown amount of the solute (the substance being analyzed) to an equivalent amount of iodine, which may then be titrated. Sometimes other halogens (or haloalkanes) besides iodine are used in the intermediate reactions because they are available in better measurable standard solutions and/or react more readily with the solute. The extr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saponification Value
Saponification value or saponification number (SV or SN) represents the number of milligrams of potassium hydroxide (KOH) or sodium hydroxide (NaOH) required to saponify one gram of fat under the conditions specified. It is a measure of the average molecular weight (or chain length) of all the fatty acids present in the sample in form of triglycerides. The higher the saponification value, the lower the fatty acids average length, the lighter the mean molecular weight of triglycerides and vice versa. Practically, fats or oils with high saponification value (such as coconut and palm oil) are more suitable for soap making. Determination To determine saponification value, the sample is treated with an excess of alkali (usually an ethanolic solution of potassium hydroxide) for half an hour under reflux. The KOH is consumed by reaction with triglycerides, which consume three equivalents of base. Diglycerides consume two equivalents of KOH. Monoglycerides and free fatty acids, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodine Value
In chemistry, the iodine value (IV; also iodine absorption value, iodine number or iodine index) is the mass of iodine in grams that is consumed by 100 grams of a chemical substance. Iodine numbers are often used to determine the degree of unsaturation in fats, oils and waxes. In fatty acids, unsaturation occurs mainly as double bonds which are very reactive towards halogens, the iodine in this case. Thus, the higher the iodine value, the more unsaturations are present in the fat. It can be seen from the table that coconut oil is very saturated, which means it is good for making soap. On the other hand, linseed oil is highly unsaturated, which makes it a drying oil, well suited for making oil paints. Principle The determination of iodine value is a particular example of iodometry. A solution of iodine is yellow/brown in color. When this is added to a solution to be tested, however, any chemical group (usually in this test double bonds) that react with iodine effectiv ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyl Value
In analytical chemistry, the hydroxyl value is defined as the number of milligrams of potassium hydroxide (KOH) required to neutralize the acetic acid taken up on acetylation of one gram of a chemical substance that contains free hydroxyl groups. The analytical method used to determine hydroxyl value traditionally involves acetylation of the free hydroxyl groups of the substance with acetic anhydride in pyridine solvent. After completion of the reaction, water is added, and the remaining unreacted acetic anhydride is converted to acetic acid and measured by titration with potassium hydroxide. The hydroxyl value can be calculated using the following equation. Note that a chemical substance may also have a measurable acid value affecting the measured endpoint of the titration. The acid value () of the substance, determined in a separate experiment, enters into this equation as a correction factor in the calculation of the hydroxyl value (): ::\mathrm = \frac + \mathrm Where i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxy Value
Epoxy value derives from the Epoxy equivalent weight (EEW) or Weight Per Epoxide (WPE) and is a measure of the epoxy content of an epoxy resin or epoxy reactive diluent, or glycidyl ether. This is an important parameter as it allows determination of the correct mix ratio of an epoxy system with a curing agent. The epoxide equivalent weight is usually measured first and done by titration. The standard test method is ASTM D1652 though this has been modified by certain states of the USA. The epoxy equivalent weight (EEW) maybe defined as: the number of grams of epoxy resin required to give 1 mole of epoxy groups. The epoxy value is defined as the number of moles of epoxy group per 100g resin. Example calculations Polyoxypropylene diglycidyl ether, a reactive diluent with the trade name of Diluent F, has an average molecular weight of 1000 and a functionality of 2 so the EEW is 1000/2 = 500. The epoxy value is defined as the number of moles of epoxy group per 100g resin. So as an e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine Number
In chemistry, the bromine number is the amount of bromine () in grams absorbed by 100 g of a sample. The bromine number was once used as a measure of aliphatic unsaturation in gasoline and related petroleum samples, but this assay has fallen into disuse with the introduction of spectroscopic and chromatographic analyses. Concept It is based on the following reaction: : Bromine is deeply colored but is not. Thus, the consumption of bromine can often be gauged visually. Alternatively and more quantitatively, the bromine consumed by a sample can be determined by iodometry. The bromine number indicates the degree of unsaturation of a sample. The technique can be subject to a variety of problems, whereby the sample consumes more or less bromine than predicted by the equation above. For example, some alkenes dimerize in the presence of bromine. Allylic bromination is another problem. The bromine number is similar to the iodine number, which is a similar technique used in evaluatin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amine Value
In organic chemistry, amine value is a measure of the nitrogen content of an organic molecule. Specifically, it is usually used to measure the amine content of amine functional compounds. It may be defined as the number of milligrams of potassium hydroxide (KOH) equivalent to one gram of epoxy hardener resin. The units are thus mg KOH/g. List of ASTM methods There are a number of ASTM analytical test methods to determine amine value. A number of states in the United States have adopted their own test methods but they are based on ASTM methods. Although there are similarities with the method it is not the same as an acid value. * ASTM D2073 - This is a potentiometric method. * ASTM D2074-07 * ASTM D2896 - potentiometric method with perchloric acid. * ASTM D6979-03 First principles The amine value is useful in helping determine the correct stoichiometry of a two component amine cure epoxy resin system.Howarth, GA. Master of Science thesis "The synthesis of a legislation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Value
In chemistry, acid value (AV, acid number, neutralization number or acidity) is a number used to quantify the acidity of a given chemical substance. It is the quantity of Base (chemistry), base (usually potassium hydroxide (KOH)), expressed as milligrams of KOH required to Neutralization (chemistry), neutralize the acidic constituents in 1 gram of a sample. The acid value measures the acidity of water-insoluble substances like oils, fats, waxes and resins, which do not have a pH value. The acid number is a measure of the number of carboxylic acid groups () in a chemical compound, such as a fatty acid, or in a mixture of compounds. In other words, it is a measure of free fatty acids (FFAs) present in a substance. In a typical procedure, a known amount of sample dissolved in an organic solvent (often isopropyl alcohol, isopropanol) and titration, titrated with a solution of alcoholic potassium hydroxide (KOH) of known concentration using phenolphthalein as a colour indicator. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox Titration
A redox titration is a type of titration based on a redox reaction between the analyte and titrant. It may involve the use of a redox indicator and/or a potentiometer. A common example of a redox titration is the treatment of a solution of iodine with a reducing agent to produce iodide using a starch indicator to help detect the endpoint. Iodine (I2) can be reduced to iodide (I−) by, say, thiosulfate (), and when all the iodine is consumed, the blue colour disappears. This is called an iodometry, iodometric titration. Most often, the reduction of iodine to iodide is the last step in a series of reactions where the initial reactions convert an unknown amount of the solute (the substance being analyzed) to an equivalent amount of iodine, which may then be titrated. Sometimes other halogens (or haloalkanes) besides iodine are used in the intermediate reactions because they are available in better measurable standard solutions and/or react more readily with the solute. The extr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
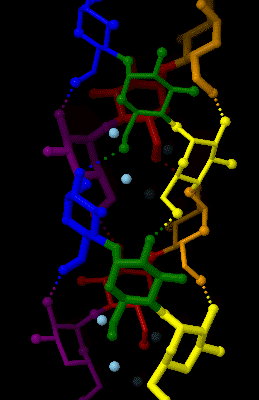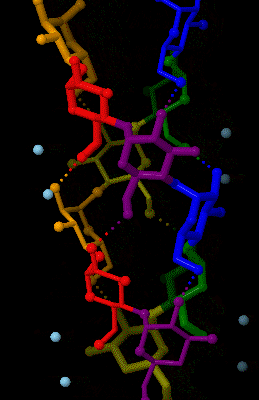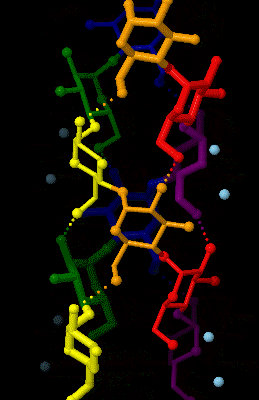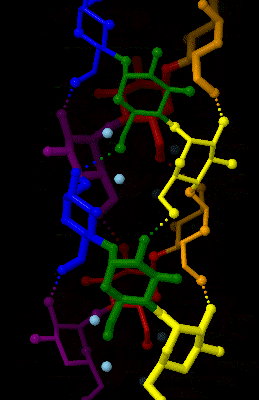
Amylose
Amylose is a polysaccharide made of α-D-glucose units, bonded to each other through α(1→4) glycosidic bonds. It is one of the two components of starch, making up approximately 20–25% of it. Because of its tightly packed Helix, helical structure, amylose is more resistant to digestion than other starch molecules and is therefore an important form of resistant starch. Structure Amylose is made up of α(1→4) bound glucose molecules. The carbon atoms on glucose are numbered, starting at the aldehyde (C=O) carbon, so, in amylose, the 1-carbon on one glucose molecule is linked to the 4-carbon on the next glucose molecule (α(1→4) bonds). The structural formula of amylose is pictured at right. The number of repeated glucose subunits (n) is usually in the range of 300 to 3000, but can be many thousands. There are three main forms of amylose chains can take. It can exist in a disordered amorphous conformation, found both in starch granules and in hydrated amylose (when starch i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Starch Indicator
Starch or amylum is a polymeric carbohydrate consisting of numerous glucose units joined by glycosidic bonds. This polysaccharide is produced by most green plants for energy storage. Worldwide, it is the most common carbohydrate in human diets, and is contained in large amounts in staple foods such as wheat, potatoes, maize (corn), rice, and cassava (manioc). Pure starch is a white, tasteless and odorless powder that is insoluble in cold water or alcohol. It consists of two types of molecules: the linear and helical amylose and the branched amylopectin. Depending on the plant, starch generally contains 20 to 25% amylose and 75 to 80% amylopectin by weight. Glycogen, the energy reserve of animals, is a more highly branched version of amylopectin. In industry, starch is often converted into sugars, for example by malting. These sugars may be fermented to produce ethanol in the manufacture of beer, whisky and biofuel. In addition, sugars produced from processed starch are use ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |