|
Pauling's Rules
Pauling's rules are five rules published by Linus Pauling in 1929 for predicting and rationalizing the crystal structures of ionic compounds. First rule: the radius ratio rule For typical ionic solids, the cations are smaller than the anions, and each cation is surrounded by coordinated anions which form a polyhedron. The sum of the ionic radii determines the cation-anion distance, while the cation-anion radius ratio r_+ / r_- (or r_c / r_a) determines the coordination number (C.N.) of the cation, as well as the shape of the coordinated polyhedron of anions. For the coordination numbers and corresponding polyhedra in the table below, Pauling mathematically derived the ''minimum'' radius ratio for which the cation is in contact with the given number of anions (considering the ions as rigid spheres). If the cation is smaller, it will not be in contact with the anions which results in instability leading to a lower coordination number. The three diagrams at right corr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linus Pauling
Linus Carl Pauling (; February 28, 1901August 19, 1994) was an American chemist, biochemist, chemical engineer, peace activist, author, and educator. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. ''New Scientist'' called him one of the 20 greatest scientists of all time, and as of 2000, he was rated the 16th most important scientist in history. For his scientific work, Pauling was awarded the Nobel Prize in Chemistry in 1954. For his peace activism, he was awarded the Nobel Peace Prize in 1962. He is one of five people to have won more than one Nobel Prize (the others being Marie Curie, John Bardeen, Frederick Sanger and Karl Barry Sharpless). Of these, he is the only person to have been awarded two unshared Nobel Prizes, and one of two people to be awarded Nobel Prizes in different fields, the other being Marie Curie. Pauling was one of the founders of the fields of quantum chemistry and molecular biology. His contributions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cube
In geometry, a cube is a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex. Viewed from a corner it is a hexagon and its net is usually depicted as a cross. The cube is the only regular hexahedron and is one of the five Platonic solids. It has 6 faces, 12 edges, and 8 vertices. The cube is also a square parallelepiped, an equilateral cuboid and a right rhombohedron a 3- zonohedron. It is a regular square prism in three orientations, and a trigonal trapezohedron in four orientations. The cube is dual to the octahedron. It has cubical or octahedral symmetry. The cube is the only convex polyhedron whose faces are all squares. Orthogonal projections The ''cube'' has four special orthogonal projections, centered, on a vertex, edges, face and normal to its vertex figure. The first and third correspond to the A2 and B2 Coxeter planes. Spherical tiling The cube can also be represented as a spheric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
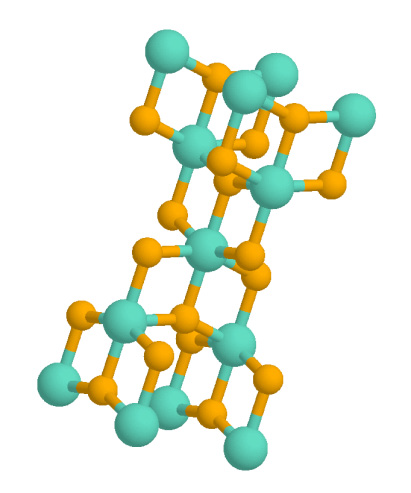
Rutile
Rutile is an oxide mineral composed of titanium dioxide (TiO2), the most common natural form of TiO2. Rarer polymorphs of TiO2 are known, including anatase, akaogiite, and brookite. Rutile has one of the highest refractive indices at visible wavelengths of any known crystal and also exhibits a particularly large birefringence and high dispersion. Owing to these properties, it is useful for the manufacture of certain optical elements, especially polarization optics, for longer visible and infrared wavelengths up to about 4.5 micrometres. Natural rutile may contain up to 10% iron and significant amounts of niobium and tantalum. Rutile derives its name from the Latin ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner. Occurrence Rutile is a common accessory mineral in high-temperature and high-pressure metamorphic rocks and in igneous rocks. Thermodynamically, ru ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanium Dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insoluble to water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint, sunscreen, and food coloring. When used as a food coloring, it has E number E171. World production in 2014 exceeded 9 million tonnes. It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion. Structure In all three of its main dioxides, titanium exhibits octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structure of rutile is tetragonal in symmetry whereas anatase and brookite are orthorhombic. The oxygen substructures are all slight dist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orthoclase
Orthoclase, or orthoclase feldspar ( endmember formula K Al Si3 O8), is an important tectosilicate mineral which forms igneous rock. The name is from the Ancient Greek for "straight fracture," because its two cleavage planes are at right angles to each other. It is a type of potassium feldspar, also known as K-feldspar. The gem known as moonstone (see below) is largely composed of orthoclase. Formation and subtypes Orthoclase is a common constituent of most granites and other felsic igneous rocks and often forms huge crystals and masses in pegmatite. Typically, the pure potassium endmember of orthoclase forms a solid solution with albite, the sodium endmember (NaAlSi3O8), of plagioclase. While slowly cooling within the earth, sodium-rich albite lamellae form by exsolution, enriching the remaining orthoclase with potassium. The resulting intergrowth of the two feldspars is called perthite. The higher-temperature polymorph of KAlSi3O8 is sanidine. Sanidine is commo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminosilicate
Aluminosilicate minerals ( IMA symbol: Als) are minerals composed of aluminium, silicon, and oxygen, plus countercations. They are a major component of kaolin and other clay minerals. Andalusite, kyanite, and sillimanite are naturally occurring aluminosilicate minerals that have the composition Al2 Si O5. The triple point of the three polymorphs is located at a temperature of and a pressure of . These three minerals are commonly used as index minerals in metamorphic rocks. Naturally occurring microporous, hydrous aluminosilicate minerals are referred to as zeolites. Feldspar is a common tectosilicate aluminosilicate mineral made of potassium, sodium, and calcium cations surrounded by a negatively charged network of silicon, aluminium and oxygen atoms. The catalyst silica-alumina is an amorphous substance which is not an aluminosilicate compound. Aluminosilicate glasses There exist a wide variety of glass types. The characteristics of these different typ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electric Charge
Electric charge is the physical property of matter that causes charged matter to experience a force when placed in an electromagnetic field. Electric charge can be ''positive'' or ''negative'' (commonly carried by protons and electrons respectively). Like charges repel each other and unlike charges attract each other. An object with an absence of net charge is referred to as neutral. Early knowledge of how charged substances interact is now called classical electrodynamics, and is still accurate for problems that do not require consideration of quantum effects. Electric charge is a conserved property; the net charge of an isolated system, the amount of positive charge minus the amount of negative charge, cannot change. Electric charge is carried by subatomic particles. In ordinary matter, negative charge is carried by electrons, and positive charge is carried by the protons in the nuclei of atoms. If there are more electrons than protons in a piece of matter, it will have ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxide
An oxide () is a chemical compound that contains at least one oxygen atom and one other element in its chemical formula. "Oxide" itself is the dianion of oxygen, an O2– (molecular) ion. with oxygen in the oxidation state of −2. Most of the Earth's crust consists of oxides. Even materials considered pure elements often develop an oxide coating. For example, aluminium foil develops a thin skin of Al2O3 (called a passivation layer) that protects the foil from further corrosion.Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the Elements (2nd Edn.), Oxford:Butterworth-Heinemann. . Stoichiometry (the measurable relationship between reactants and chemical equations of a equation or reaction) Oxides are extraordinarily diverse in terms of stoichiometries and in terms of the structures of each stoichiometry. Most elements form oxides of more than one stoichiometry. A well known example is carbon monoxide and carbon dioxide.Greenwood, N. N.; & Earnshaw, A. (1997). Chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Chloride
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contains 39.34 g Na and 60.66 g Cl. Sodium chloride is the salt most responsible for the salinity of seawater and of the extracellular fluid of many multicellular organisms. In its edible form, salt (also known as '' table salt'') is commonly used as a condiment and food preservative. Large quantities of sodium chloride are used in many industrial processes, and it is a major source of sodium and chlorine compounds used as feedstocks for further chemical syntheses. Another major application of sodium chloride is de-icing of roadways in sub-freezing weather. Uses In addition to the familiar domestic uses of salt, more dominant applications of the approximately 250 million tonnes per year productio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CsCl Crystal China.
{{disambig ...
CSCL can refer to: * Caesium chloride (CsCl), a chemical compound. * Computer Supported Collaborative Learning, a research topic on supporting collaborative learning with the assistance of computer artifacts. * China Shipping Container Lines, a containerized marine shipping company, based in Shanghai Shanghai (; , , Standard Mandarin pronunciation: ) is one of the four direct-administered municipalities of the People's Republic of China (PRC). The city is located on the southern estuary of the Yangtze River, with the Huangpu River flowin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NaCl Polyhedra
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/mol respectively, 100 g of NaCl contains 39.34 g Na and 60.66 g Cl. Sodium chloride is the salt most responsible for the salinity of seawater and of the extracellular fluid of many multicellular organisms. In its edible form, salt (also known as ''table salt'') is commonly used as a condiment and food preservative. Large quantities of sodium chloride are used in many industrial processes, and it is a major source of sodium and chlorine compounds used as feedstocks for further chemical syntheses. Another major application of sodium chloride is de-icing of roadways in sub-freezing weather. Uses In addition to the familiar domestic uses of salt, more dominant applications of the approximately 250 million tonnes per year production (2008 dat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of Chemical Education
The ''Journal of Chemical Education'' is a monthly peer-reviewed academic journal available in both print and electronic versions. It is published by the Division of Chemical Education of the American Chemical Society and was established in 1924 by Neil Gordon Neil Elbridge Gordon (October 7, 1886 – May 30, 1949) was an American chemist and educator. He is known for founding the Journal of Chemical Education (c. 1924) and establishing the Gordon Research Conferences (c. 1931). He held several Chair .... The journal covers research on chemical education, and its target audience includes instructors of chemistry from middle school through graduate school and some scientists in commerce, industry, and government. References External links * Chemical education journals American Chemical Society academic journals Monthly journals Publications established in 1924 English-language journals {{chemistry-journal-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |