|
Layered Materials
In material science, layered materials are solids with highly anisotropic bonding, in which two-dimensional sheets are internally strongly bonded, but only weakly bonded to adjacent layers. Owing to their distinctive structures, layered materials are often suitable for intercalation reactions. One large family of layered materials are metal dichalcogenides. In such materials, the M-chalcogen bonding is strong and covalent. These materials exhibit anisotropic electronic properties such as thermal and electrical conductivity. Exfoliation Because the layers bond to each other by relatively weak van der Waals forces, some layered materials are amenable to exfoliation, the complete separation of the layers of the material. Exfoliation can be done using sonication, mechanical, hydrothermal, electrochemical, laser-assisted, and microwave-assisted methods. Typically aggressive conditions are required involving highly polar solvents and reagents. In the ideal case, exfoliation affords ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Material Science
A material is a substance or mixture of substances that constitutes an object. Materials can be pure or impure, living or non-living matter. Materials can be classified on the basis of their physical and chemical properties, or on their geological origin or biological function. Materials science is the study of materials, their properties and their applications. Raw materials can be processed in different ways to influence their properties, by purification, shaping or the introduction of other materials. New materials can be produced from raw materials by synthesis. In industry, materials are inputs to manufacturing processes to produce products or more complex materials, and the nature and quantity of materials used may form part of the calculation for the cost of a product or delivery under contract, such as where contract costs are calculated on a " time and materials" basis. Historical elements Materials chart the history of humanity. The system of the three prehist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Intercalation Reaction
Intercalation is the reversible inclusion or insertion of a molecule (or ion) into layered materials with layered structures. Examples are found in graphite and transition metal dichalcogenides. : Examples Graphite One famous intercalation host is graphite, which intercalates potassium as a guest. Intercalation expands the van der Waals gap between sheets, which requires energy. Usually this energy is supplied by charge transfer between the guest and the host solid, i.e., redox. Two potassium graphite compounds are KC8 and KC24. Carbon fluorides (e.g., (CF)x and (C4F)) are prepared by reaction of fluorine with graphitic carbon. The color is greyish, white, or yellow. The bond between the carbon and fluorine atoms is covalent, thus fluorine is not intercalated. Such materials have been considered as a cathode in various lithium batteries. Treating graphite with strong acids in the presence of oxidizing agents causes the graphite to oxidise. Graphite bisulfate, 24sup>+ SO4sup>� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
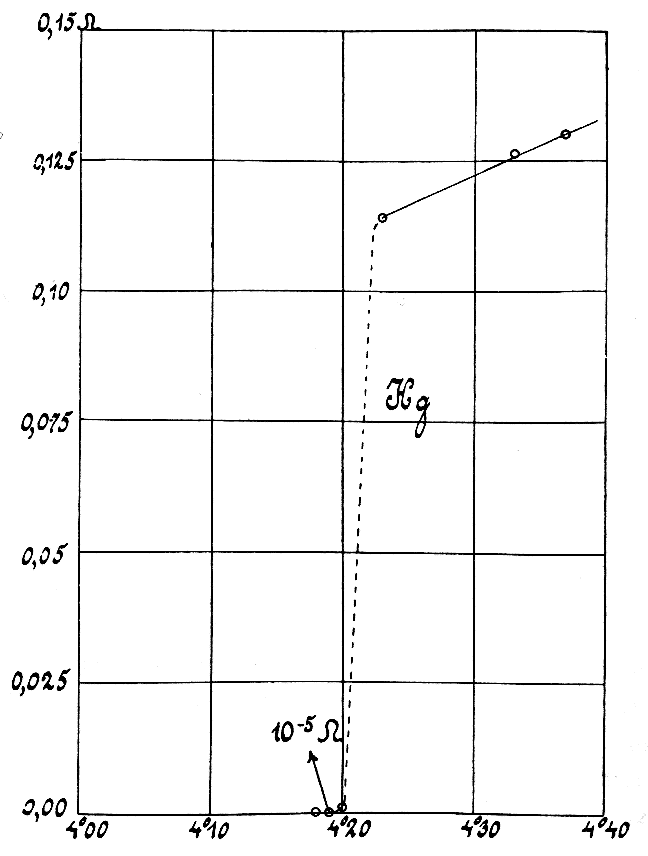
Electrical Conductivity
Electrical resistivity (also called volume resistivity or specific electrical resistance) is a fundamental specific property of a material that measures its electrical resistance or how strongly it resists electric current. A low resistivity indicates a material that readily allows electric current. Resistivity is commonly represented by the Greek alphabet, Greek letter (Rho (letter), rho). The SI unit of electrical resistivity is the ohm-metre (Ω⋅m). For example, if a solid cube of material has sheet contacts on two opposite faces, and the Electrical resistance, resistance between these contacts is , then the resistivity of the material is . Electrical conductivity (or specific conductance) is the reciprocal of electrical resistivity. It represents a material's ability to conduct electric current. It is commonly signified by the Greek letter (Sigma (letter), sigma), but (kappa) (especially in electrical engineering) and (gamma) are sometimes used. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Van Der Waals Force
In molecular physics and chemistry, the van der Waals force (sometimes van der Waals' force) is a distance-dependent interaction between atoms or molecules. Unlike ionic or covalent bonds, these attractions do not result from a chemical electronic bond; they are comparatively weak and therefore more susceptible to disturbance. The van der Waals force quickly vanishes at longer distances between interacting molecules. Named after Dutch physicist Johannes Diderik van der Waals, the van der Waals force plays a fundamental role in fields as diverse as supramolecular chemistry, structural biology, polymer science, nanotechnology, surface science, and condensed matter physics. It also underlies many properties of organic compounds and molecular solids, including their solubility in polar and non-polar media. If no other force is present, the distance between atoms at which the force becomes repulsive rather than attractive as the atoms approach one another is called the van der ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Single-layer Materials
In materials science, the term single-layer materials or 2D materials refers to crystalline solids consisting of a single layer of atoms. These materials are promising for some applications but remain the focus of research. Single-layer materials derived from single elements generally carry the -ene suffix in their names, e.g. graphene. Single-layer materials that are compounds of two or more elements have -ane or -ide suffixes. 2D materials can generally be categorized as either 2D allotropes of various elements or as compounds (consisting of two or more covalently bonding elements). It is predicted that there are hundreds of stable single-layer materials. The atomic structure and calculated basic properties of these and many other potentially synthesisable single-layer materials, can be found in computational databases. 2D materials can be produced using mainly two approaches: top-down exfoliation and bottom-up synthesis. The exfoliation methods include sonication, mechanical, h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Graphene
Graphene () is a carbon allotrope consisting of a Single-layer materials, single layer of atoms arranged in a hexagonal lattice, honeycomb planar nanostructure. The name "graphene" is derived from "graphite" and the suffix -ene, indicating the presence of double bonds within the carbon structure. Graphene is known for its exceptionally high Ultimate tensile strength, tensile strength, Electrical resistivity and conductivity, electrical conductivity, Transparency and translucency, transparency, and being the thinnest two-dimensional material in the world. Despite the nearly transparent nature of a single graphene sheet, graphite (formed from stacked layers of graphene) appears black because it absorbs all visible light wavelengths. On a microscopic scale, graphene is the strongest material ever measured. The existence of graphene was first theorized in 1947 by P. R. Wallace, Philip R. Wallace during his research on graphite's electronic properties, while the term ''graphen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Metal Dichalcogenide
: 220px, Cadmium sulfide, a prototypical metal chalcogenide, is used as a yellow pigment. A chalcogenide is a chemical compound consisting of at least one chalcogen anion and at least one more electropositive element. Although all group 16 elements of the periodic table are defined as chalcogens, the term chalcogenide is more commonly reserved for sulfides, selenides, tellurides, and polonides, rather than oxides. Many metal ores exist as chalcogenides. Photoconductive chalcogenide glasses are used in xerography. Some pigments and catalysts are also based on chalcogenides. The metal dichalcogenide MoS2 is a common solid lubricant. Alkali metal and alkaline earth chalcogenides Alkali metal and alkaline earth monochalcogenides are salt-like, being colourless and often water-soluble. The sulfides tend to undergo hydrolysis to form derivatives containing bisulfide (SH−) anions. The alkali metal chalcogenides often crystallize with the antifluorite structure and the alkaline ear ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Tantalum Disulfide
Tantalum is a chemical element; it has symbol Ta and atomic number 73. It is named after Tantalus, a figure in Greek mythology. Tantalum is a very hard, ductile, lustrous, blue-gray transition metal that is highly corrosion-resistant. It is part of the refractory metals group, which are widely used as components of strong high-melting-point alloys. It is a group 5 element, along with vanadium and niobium, and it always occurs in geologic sources together with the chemically similar niobium, mainly in the mineral groups tantalite, columbite, and coltan. The chemical inertness and very high melting point of tantalum make it valuable for laboratory and industrial equipment such as reaction vessels and vacuum furnaces. It is used in tantalum capacitors for electronic equipment such as computers. It is being investigated for use as a material for high-quality superconducting resonators in quantum processors. History Tantalum was discovered in Sweden in 1802 by Anders Ekeberg, in t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Titanium Disulfide
Titanium disulfide is an inorganic compound with the formula Ti S2. A golden yellow solid with high electrical conductivity, it belongs to a group of compounds called transition metal di chalcogenides, which consist of the stoichiometry M E2. TiS2 has been employed as a cathode material in rechargeable batteries. Structure With a layered structure, TiS2 adopts a hexagonal close packed (hcp) structure, analogous to cadmium iodide (CdI2). In this motif, half of the octahedral holes are filled with a "cation", in this case Ti4+. Each Ti centre is surrounded by six sulfide ligands in an octahedral structure. Each sulfide is connected to three Ti centres, the geometry at S being pyramidal. Several metal di chalcogenides adopt similar structures, but some, notably MoS2, do not. The layers of TiS2 consist of covalent Ti-S bonds. The individual layers of TiS2 are bound together by van der Waals forces, which are relatively weak intermolecular forces. It crystallises in the space ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Graphite
Graphite () is a Crystallinity, crystalline allotrope (form) of the element carbon. It consists of many stacked Layered materials, layers of graphene, typically in excess of hundreds of layers. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on a large scale (1.3million metric tons per year in 2022) for uses in many critical industries including refractories (50%), lithium-ion batteries (18%), foundries (10%), and lubricants (5%), among others (17%). Graphite converts to diamond under extremely high pressure and temperature. Graphite's low cost, thermal and chemical inertness and characteristic conductivity of heat and electricity finds numerous applications in high energy and high temperature processes. Types and varieties Graphite can occur naturally or be produced synthetically. Natural graphite is obtained from naturally occurring geologic deposits and synthetic graphite is produced t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Iron Oxychloride
Iron oxychloride is the inorganic compound with the formula FeOCl. This purple solid adopts a layered structure, akin to that of cadmium chloride. The material slowly hydrolyses in moist air. The solid intercalates electron donors such as tetrathiafulvalene and even pyridine to give mixed valence charge-transfer salts. Intercalation is accompanied by a marked increase in electrical conductivity and a color change to black. Production FeOCl is prepared by heating iron(III) oxide with ferric chloride at over the course of several days: :Fe2O3 + FeCl3 → 3 FeOCl Alternatively, FeOCl may be prepared by the thermal decomposition Thermal decomposition, or thermolysis, is a chemical decomposition of a substance caused by heat. The decomposition temperature of a substance is the temperature at which the substance chemically decomposes. The reaction is usually endothermic ... of FeCl3⋅6H2O at over the course of one hour: :FeCl3 ⋅ 6H2O → FeOCl + 5 H2O + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |