|
Ion-selective Electrode
An ion-selective electrode (ISE), also known as a specific ion electrode (SIE), is a transducer (or sensor) that converts the activity of a specific ion dissolved in a solution into an electrical potential. The voltage is theoretically dependent on the logarithm of the ionic activity, according to the Nernst equation. Ion-selective electrodes are used in analytical chemistry and biochemical/biophysical research, where measurements of ionic concentration in an aqueous solution are required. Types of ion-selective membrane There are four main types of ion-selective membrane used in ion-selective electrodes (ISEs): glass, solid state, liquid based, and compound electrode. Glass membranes Glass membranes are made from an ion-exchange type of glass ( silicate or chalcogenide). This type of ISE has good selectivity, but only for several single-charged cations; mainly H+, Na+, and Ag+. Chalcogenide glass also has selectivity for double-charged metal ions, such as Pb2+, and Cd2+. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
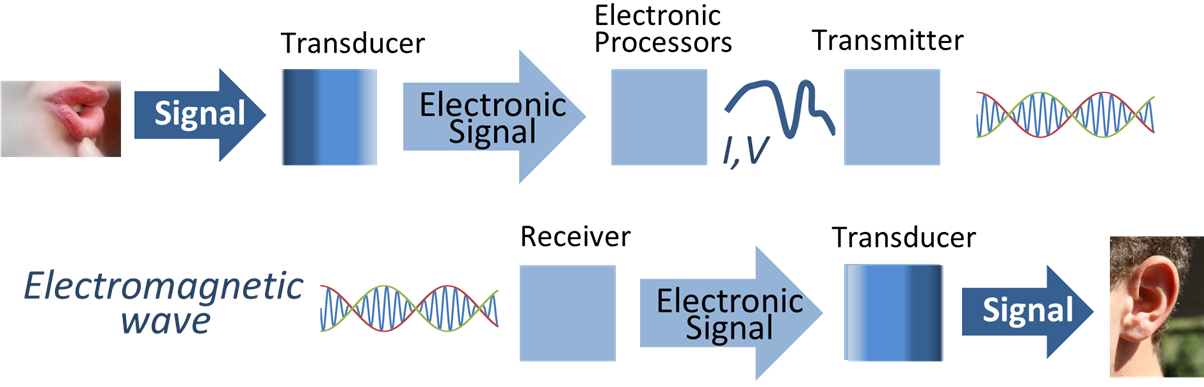
Transducer
A transducer is a device that converts energy from one form to another. Usually a transducer converts a signal in one form of energy to a signal in another. Transducers are often employed at the boundaries of automation, measurement, and control systems, where electrical signals are converted to and from other physical quantities (energy, force, torque, light, motion, position, etc.). The process of converting one form of energy to another is known as transduction. Types * Mechanical transducers, so-called as they convert physical quantities into mechanical outputs or vice versa; * Electrical transducers however convert physical quantities into electrical outputs or signals. Examples of these are: ** a thermocouple that changes temperature differences into a small voltage; ** a linear variable differential transformer (LVDT), used to measure displacement (position) changes by means of electrical signals. Sensors, actuators and transceivers Transducers can be categorized by wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cation
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, using energy from sunlight, where it is used to make cellulose in cell walls, the most abundant carbohydrate in the world. In energy metabolism, glucose is the most important source of energy in all organisms. Glucose for metabolism is stored as a polymer, in plants mainly as starch and amylopectin, and in animals as glycogen. Glucose circulates in the blood of animals as blood sugar. The naturally occurring form of glucose is -glucose, while -glucose is produced synthetically in comparatively small amounts and is less biologically active. Glucose is a monosaccharide containing six carbon atoms and an aldehyde group, and is therefore an aldohexose. The glucose molecule can exist in an open-chain (acyclic) as well as ring (cyclic) form. Gluco ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Valinomycin
Valinomycin is a naturally occurring dodecadepsipeptide used in the transport of potassium and as an antibiotic. Valinomycin is obtained from the cells of several '' Streptomyces'' species, '' S. fulvissimus'' being a notable one. It is a member of the group of natural neutral ionophores because it does not have a residual charge. It consists of enantiomers D- and L-valine (Val), D- alpha-hydroxyisovaleric acid, and L-lactic acid. Structures are alternately bound via amide and ester bridges. Valinomycin is highly selective for potassium ions over sodium ions within the cell membrane. It functions as a potassium-specific transporter and facilitates the movement of potassium ions through lipid membranes "down" the electrochemical potential gradient. The stability constant K for the potassium-valinomycin complex is nearly 100,000 times larger than that of the sodium-valinomycin complex. This difference is important for maintaining the selectivity of valinomycin for the transport of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Selective Electrode
Potassium selective electrodes are a type of ion selective electrode used in biochemical and biophysical research, where measurements of potassium concentration in an aqueous solution are required, usually on a real time basis. These electrodes are typical ion exchange resin membrane electrodes, using valinomycin, a potassium ionophore, as the ion carrier in the membrane to provide the potassium specificity. This type of ion-selective electrode is subject to interference from (in declining order of magnitude) rubidium, caesium, ammonium, sodium, calcium, magnesium, and lithium Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid el .... The most significant interference with measurement of potassium concentration is from the ammonium ion, which in practice is a problem where the ammoniu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ion-exchange Resin
An ion-exchange resin or ion-exchange polymer is a resin or polymer that acts as a medium for ion exchange. It is an insoluble matrix (or support structure) normally in the form of small (0.25–1.43 mm radius) microbeads, usually white or yellowish, fabricated from an organic polymer substrate. The beads are typically porous, providing a large surface area on and inside them where the trapping of ions occurs along with the accompanying release of other ions, and thus the process is called ion exchange. There are multiple types of ion-exchange resin. Most commercial resins are made of polystyrene sulfonate.François Dardel and Thomas V. Arden "Ion Exchangers" in Ullmann's Encyclopedia of Industrial Chemistry, 2008, Wiley-VCH, Weinheim. . Ion-exchange resins are widely used in different separation, purification, and decontamination processes. The most common examples are water softening and water purification. In many cases ion-exchange resins were introduced in such proce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lanthanum Trifluoride
Lanthanum trifluoride is a refractory ionic compound of lanthanum and fluorine. The LaF3 structure Bonding is ionic with lanthanum highly coordinated. The cation sits at the center of a trigonal prism. Nine fluorine atoms are close: three at the bottom corners of the trigonal prism, three in the faces of the trigonal prism, and three at top corners of the trigonal prism. There are also two fluorides a little further away above and below the prism. The cation can be considered 9-coordinate or 11-coordinate. At 300 K, the structure allows the formation of Schottky defects with an activation energy of 0.07 eV, and free flow of fluoride ions with an activation energy of 0.45 eV, making the crystal unusually electrically conductive. The larger sized rare earth elements (lanthanides), which are those with smaller atomic number, also form trifluorides with the LaF3 structure. Some actinides do as well. Applications This white salt is sometimes used as the "high-ind ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluoride Selective Electrode
A fluoride selective electrode is a type of ion selective electrode sensitive to the concentration of the fluoride ion. A common example is the lanthanum fluoride electrode. Lanthanum fluoride electrode In the lanthanum fluoride electrode, the sensing element is a crystal of lanthanum fluoride (LaF3), doped with europium(II) fluoride (EuF2) to create lattice vacancies. Such a crystal is an ionic conductor by virtue of the mobility of fluoride ions which jump between lattice vacancies. An electrochemical cell may be constructed using such a crystal as a membrane separating two fluoride solutions. This cell acts as a concentration cell with transference where the fluoride transport number is 1. As transference of charge through the crystal is almost exclusively due to fluoride, the electrode is highly specific to fluoride. The only ion which significantly interferes is hydroxide (OH−). Generally such "alkaline error" can be avoided by buffering the sample to a pH below 7. Cell ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). Electrodes are essential parts of batteries that can consist of a variety of materials depending on the type of battery. The electrophore, invented by Johan Wilcke, was an early version of an electrode used to study static electricity. Anode and cathode in electrochemical cells Electrodes are an essential part of any battery. The first electrochemical battery made was devised by Alessandro Volta and was aptly named the Voltaic cell. This battery consisted of a stack of copper and zinc electrodes separated by brine-soaked paper disks. Due to fluctuation in the voltage provided by the voltaic cell it wasn't very practical. The first practical battery was invented in 1839 and named the Daniell cell after John Frederic Daniell. Still making use of the zinc–copper electrode combination. Since then many more batteries have be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |