|
Hexane-2,5-dione
2,5-Hexanedione (Acetonylacetone) is an aliphatic diketone. It is a colorless liquid. In humans, it is a toxic metabolite of hexane and of 2-hexanone. Symptoms of poisoning The chronic toxicity of hexane is attributed to hexane-2,5-dione. The symptoms are tingling and cramps in the arms and legs, followed by general muscular weakness. In severe cases, atrophy of the skeletal muscles is observed, along with a loss of coordination and vision problems. Similar symptoms are observed in animal models. They are associated with a degeneration of the peripheral nervous system (and eventually the central nervous system), starting with the distal portions of the longer and wider nerve axons. Mechanism of action It appears that the neurotoxicity of 2,5-hexanedione resides in its γ-diketone structure since 2,3-, 2,4-hexanedione and 2,6-heptanedione are not neurotoxic, whereas 2,5-heptanedione and 3,6-octanedione and other γ-diketones are. 2,5-Hexanedione reacts with lysine r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,5-Dimethylthiophene
2,5-Dimethylthiophene is an organosulfur compound with the formula C4H2(CH3)2S. It is one of four isomers of dimethylthiophene. A colorless liquid, it is prepared by sulfurization of hexane-2,5-dione. It is approved as a food flavouring additive in Europe. References [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,5-Dimethylfuran
2,5-Dimethylfuran is a heterocyclic compound with the formula (CH3)2C4H2O. Although often abbreviated DMF, it should not be confused with dimethylformamide. A derivative of furan, this simple compound is a potential biofuel, being derivable from cellulose. Production Fructose can be converted into 2,5-dimethylfuran in a catalytic biomass-to-liquid process. The conversion of fructose to DMF proceeds via hydroxymethylfurfural. Fructose is obtainable from glucose, a building block in cellulose. Potential as a biofuel DMF has a number of attractions as a biofuel. It has an energy density 40% greater than that of ethanol, making it comparable to gasoline (petrol). It is also chemically stable and, being insoluble in water, does not absorb moisture from the atmosphere. Evaporating dimethylfuran during the production process also requires around one third less energy than the evaporation of ethanol, although it has a boiling point some 14 °C higher, at 92 °C, compared to 78& ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Pentasulfide
Phosphorus pentasulfide is the inorganic compound with the formula (monomer) or (dimer). This yellow solid is the one of two phosphorus sulfides of commercial value. Samples often appear greenish-gray due to impurities. It is soluble in carbon disulfide but reacts with many other solvents such as alcohols, DMSO, and DMF. Structure and synthesis Its tetrahedral molecular structure is similar to that of adamantane and almost identical to the structure of phosphorus pentoxide. Phosphorus pentasulfide is obtained by the reaction of liquid white phosphorus () with sulfur above 300 °C. The first synthesis of by Berzelius in 1843 was by this method. Alternatively, can be formed by reacting elemental sulfur or pyrite, , with ferrophosphorus, a crude form of (a byproduct of white phosphorus () production from phosphate rock): : : Applications Approximately 150,000 tons of are produced annually. The compound is mainly converted to other derivatives for use as lubrication ad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isocarboxazid
Isocarboxazid (Marplan, Marplon, Enerzer) is a non-selective, irreversible monoamine oxidase inhibitor (MAOI) of the hydrazine class used as an antidepressant. Along with phenelzine and tranylcypromine, it is one of only three classical MAOIs still available for clinical use in the treatment of psychiatric disorders in the United States, though it is not as commonly employed in comparison to the others. Isocarboxazid is primarily used to treat mood and anxiety disorders. It has also been investigated in the treatment of schizophrenia, Parkinson's disease and other dementia-related disorders. Isocarboxazid, as well as other MAOIs, increase the levels of the monoamine neurotransmitters serotonin, dopamine, norepinephrine, epinephrine, melatonin, phenethylamine in the brain.Volz, Hanz-Peter. (November 1998) “Monoamine Oxidase Inhibitors A Perspective on Their Use in The Elderly” ''Biochemical Pharmacology'' (5) 341-352. Classical MAOIs, including isocarboxazid, are u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
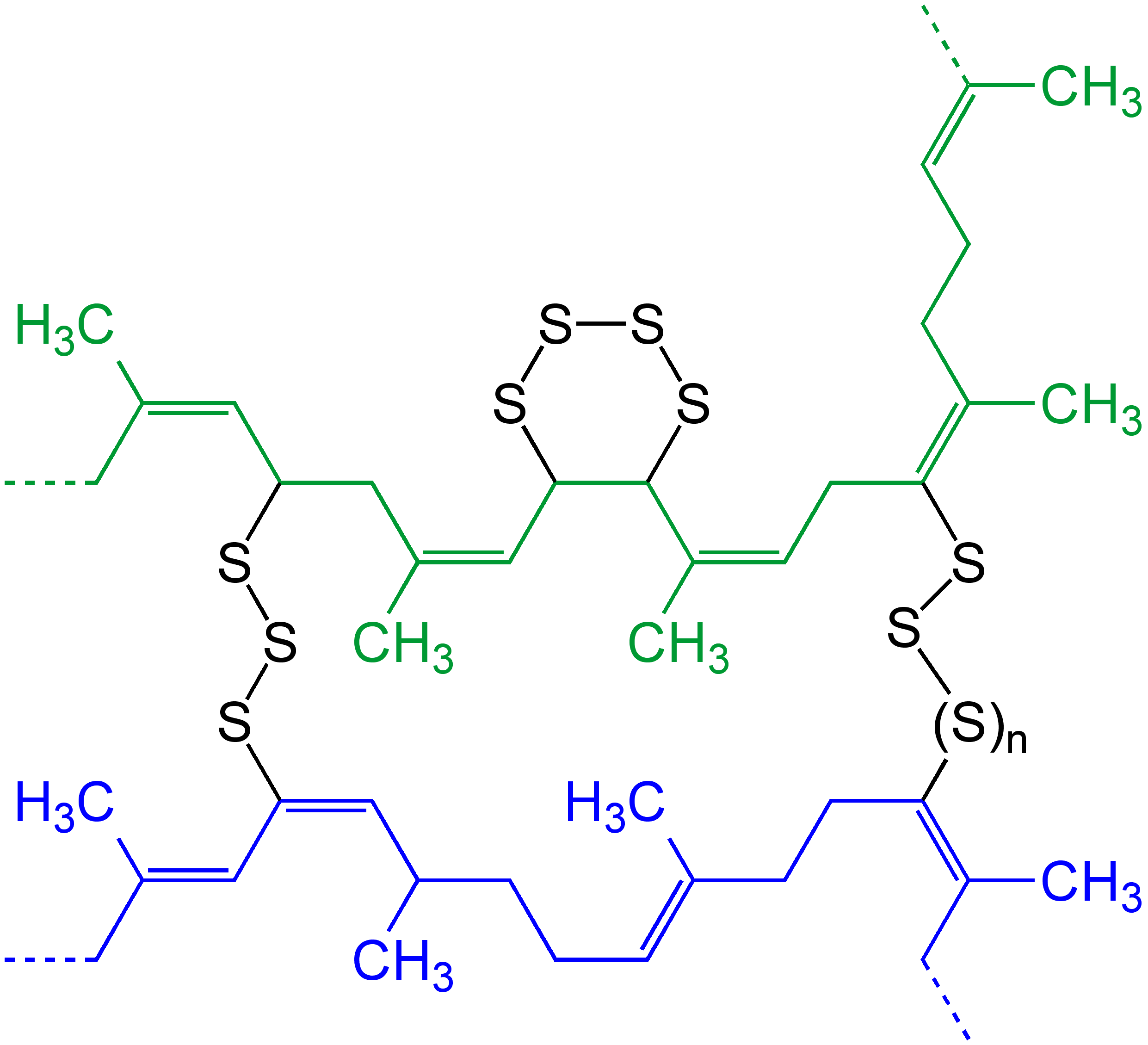
Cross-link
In chemistry and biology a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural polymers (such as proteins). In polymer chemistry "cross-linking" usually refers to the use of cross-links to promote a change in the polymers' physical properties. When "crosslinking" is used in the biological field, it refers to the use of a probe to link proteins together to check for protein–protein interactions, as well as other creative cross-linking methodologies. Although the term is used to refer to the "linking of polymer chains" for both sciences, the extent of crosslinking and specificities of the crosslinking agents vary greatly. As with all science, there are overlaps, and the following delineations are a starting point to understanding the subtleties. Polymer chemistry Crosslinking is the general term for the process of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogenation, C=C (and other) bonds ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrrole
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4 H4 NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., ''N''-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme. Pyrroles are components of more complex macrocycles, including the porphyrinogens and products derived therefrom, including porphyrins of heme, the chlorins, bacteriochlorins, and chlorophylls. Properties Pyrrole is a colorless volatile liquid that darkens readily upon exposure to air, and is usually purified by distillation immediately before use. Pyrrole has a nutty odor. Pyrrole is a 5-membered aromatic heterocycle, like furan and thiophene. Unlike furan and thiophene, it has a dipole in which the positive end lies on the side of the heteroatom, with a dipole moment of 1.58 D. In CDCl3, it has ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclization
A cyclic compound (or ring compound) is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where all the atoms are carbon (i.e., are carbocycles), none of the atoms are carbon (inorganic cyclic compounds), or where both carbon and non-carbon atoms are present (heterocyclic compounds). Depending on the ring size, the bond order of the individual links between ring atoms, and their arrangements within the rings, carbocyclic and heterocyclic compounds may be aromatic or non-aromatic; in the latter case, they may vary from being fully saturated to having varying numbers of multiple bonds between the ring atoms. Because of the tremendous diversity allowed, in combination, by the valences of common atoms and their ability to form rings, the number of possible cyclic structures, even of small size (e.g., < 17 total atoms) numbers in the many b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Schiff Base
In organic chemistry, a Schiff base (named after Hugo Schiff) is a compound with the general structure ( = alkyl or aryl, but not hydrogen). They can be considered a sub-class of imines, being either secondary ketimines or secondary aldimines depending on their structure. The term is often synonymous with azomethine which refers specifically to secondary aldimines (i.e. where R' ≠ H). A number of special naming systems exist for these compounds. For instance a Schiff base derived from an aniline, where is a phenyl or a substituted phenyl, can be called an ''anil'', while bis-compounds are often referred to as salen-type compounds. The term Schiff base is normally applied to these compounds when they are being used as ligands to form coordination complexes with metal ions. Such complexes occur naturally, for instance in corrin, but the majority of Schiff bases are artificial and are used to form many important catalysts, such as Jacobsen's catalyst. Synthesis Schiff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lysine
Lysine (symbol Lys or K) is an α-amino acid that is a precursor to many proteins. It contains an α-amino group (which is in the protonated form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −COO− form under biological conditions), and a side chain lysyl ((CH2)4NH2), classifying it as a basic, charged (at physiological pH), aliphatic amino acid. It is encoded by the codons AAA and AAG. Like almost all other amino acids, the α-carbon is chiral and lysine may refer to either enantiomer or a racemic mixture of both. For the purpose of this article, lysine will refer to the biologically active enantiomer L-lysine, where the α-carbon is in the ''S'' configuration. The human body cannot synthesize lysine. It is essential in humans and must therefore be obtained from the diet. In organisms that synthesise lysine, two main biosynthetic pathways exist, the diaminopimelate and α-aminoadipate pathways, which employ distinct e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |