|
Hydroxyacid Oxidase (glycolate Oxidase) 1
Hydroxyacid oxidase (glycolate oxidase) 1 is a protein that in humans is encoded by the HAO1 gene. Function This gene is one of three related genes that have 2-hydroxyacid oxidase activity yet differ in encoded protein amino acid sequence, tissue expression and substrate preference. Subcellular location of the encoded protein is the peroxisome. Specifically, this gene is expressed primarily in liver and pancreas and the encoded protein is most active on glycolate, a two-carbon substrate. Glycolate oxidase oxidizes glycolic acid to glyoxylate, and can also oxidize glyoxylate into oxalate. These reactions are central to the toxicity of ethylene glycol poisoning. The protein is also active on 2-hydroxy fatty acids. The transcript detected at high levels in pancreas may represent an alternatively spliced form or the use of a multiple near-consensus upstream polyadenylation Polyadenylation is the addition of a poly(A) tail to an RNA transcript, typically a messenger RNA (mRNA) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid resid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a basic unit of heredity and the molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protein-coding genes and noncoding genes. During gene expression, the DNA is first copied into RNA. The RNA can be directly functional or be the intermediate template for a protein that performs a function. The transmission of genes to an organism's offspring is the basis of the inheritance of phenotypic traits. These genes make up different DNA sequences called genotypes. Genotypes along with environmental and developmental factors determine what the phenotypes will be. Most biological traits are under the influence of polygenes (many different genes) as well as g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2-hydroxyacid
α-Hydroxy acids, or alpha hydroxy acids (AHAs), are a class of chemical compounds that consist of a carboxylic acid with a hydroxyl group substituent on the adjacent (alpha) carbon. Prominent examples are glycolic acid, lactic acid, mandelic acid and citric acid. Although these compounds are related to the ordinary carboxylic acids and are therefore weak acids, their chemical structure allows for the formation of an internal hydrogen bond between the hydrogen at the hydroxyl group and one of the oxygen atoms of the carboxylic group. The net effect is an increase in acidity. For example, the pKa of lactic acid is 3.86, while that of the unsubstituted propionic acid is 4.87; a full pKa unit difference means that lactic acid is ten times stronger than propionic acid. Industrial applications Feed additives 2-Hydroxy-4-(methylthio)butyric acid is produced commercially as a racemic mixture to substitute for methionine in animal feed. In nature, the same compound is an intermediate in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidase
In biochemistry, an oxidase is an enzyme that catalyzes oxidation-reduction reactions, especially one involving dioxygen (O2) as the electron acceptor. In reactions involving donation of a hydrogen atom, oxygen is reduced to water (H2O) or hydrogen peroxide (H2O2). Some oxidation reactions, such as those involving monoamine oxidase or xanthine oxidase, typically do not involve free molecular oxygen. The oxidases are a subclass of the oxidoreductases. Examples An important example is cytochrome c oxidase, the key enzyme that allows the body to employ oxygen in the generation of energy and the final component of the electron transfer chain. Other examples are: * Glucose oxidase * Monoamine oxidase * Cytochrome P450 oxidase * NADPH oxidase * Xanthine oxidase * L-gulonolactone oxidase * Laccase * Lysyl oxidase * Polyphenol oxidase * Sulfhydryl oxidase. This enzyme oxidises thiol groups. Oxidase test In microbiology, the oxidase test is used as a phenotypic characteristic for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxisome
A peroxisome () is a membrane-bound organelle, a type of microbody, found in the cytoplasm of virtually all eukaryotic cells. Peroxisomes are oxidative organelles. Frequently, molecular oxygen serves as a co-substrate, from which hydrogen peroxide (H2O2) is then formed. Peroxisomes owe their name to hydrogen peroxide generating and scavenging activities. They perform key roles in lipid metabolism and the conversion of reactive oxygen species. Peroxisomes are involved in the catabolism of very long chain fatty acids, branched chain fatty acids, bile acid intermediates (in the liver), D-amino acids, and polyamines, the reduction of reactive oxygen species – specifically hydrogen peroxide – and the biosynthesis of plasmalogens, i.e., ether phospholipids critical for the normal function of mammalian brains and lungs. They also contain approximately 10% of the total activity of two enzymes ( Glucose-6-phosphate dehydrogenase and 6-Phosphogluconate dehydrogenase) in t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycolate
Glycolic acid (or hydroxyacetic acid; chemical formula HOCH2CO2H) is a colorless, odorless and hygroscopic crystalline solid, highly soluble in water. It is used in various skin-care products. Glycolic acid is widespread in nature. A glycolate (sometimes spelled "glycollate") is a salt or ester of glycolic acid. History The name "glycolic acid" was coined in 1848 by French chemist Auguste Laurent (1807–1853). He proposed that the amino acid glycine—which was then called ''glycocolle''—might be the amine of a hypothetical acid, which he called "glycolic acid" (''acide glycolique''). Glycolic acid was first prepared in 1851 by German chemist Adolph Strecker (1822–1871) and Russian chemist Nikolai Nikolaevich Sokolov (1826–1877). They produced it by treating hippuric acid with nitric acid and nitrogen dioxide to form an ester of benzoic acid and glycolic acid (C6H5C(=O)OCH2COOH), which they called "benzoglycolic acid" (''Benzoglykolsäure''; also benzoyl glycolic aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glyoxylate
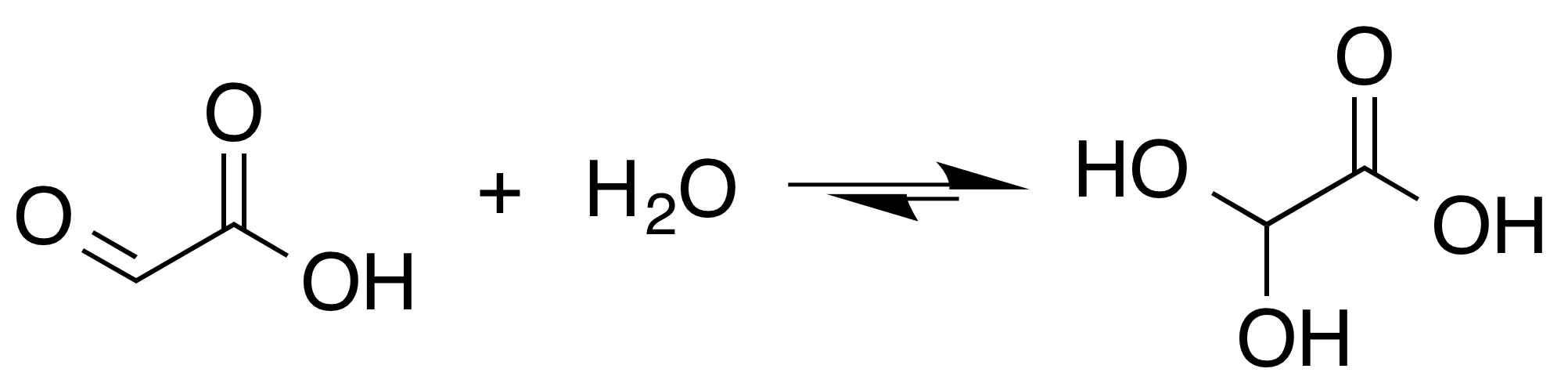
Glyoxylic acid or oxoacetic acid is an organic compound. Together with acetic acid, glycolic acid, and oxalic acid, glyoxylic acid is one of the C2 carboxylic acids. It is a colourless solid that occurs naturally and is useful industrially. Structure and nomenclature Although the structure of glyoxylic acid is described as having an aldehyde functional group, the aldehyde is only a minor component of the form most prevalent in some situations. Instead, it often exists as a hydrate or a cyclic dimer. For example, in the presence of water, the carbonyl rapidly converts to a geminal diol (described as the "monohydrate"). The equilibrium constant (''K'') is 300 for the formation of dihydroxyacetic acid at room temperature: : In solution, the monohydrate exists in equilibrium with a hemiacylal dimer form:Georges Mattioda and Yani Christidis “Glyoxylic Acid” Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. : In isolation, the aldehyde structure has ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxalate
Oxalate (IUPAC: ethanedioate) is an anion with the formula C2O42−. This dianion is colorless. It occurs naturally, including in some foods. It forms a variety of salts, for example sodium oxalate (Na2C2O4), and several esters such as dimethyl oxalate (C2O4(CH3)2). It is a conjugate base of oxalic acid. At neutral pH in aqueous solution, oxalic acid converts completely to oxalate. Relationship to oxalic acid The dissociation of protons from oxalic acid proceeds in a stepwise manner; as for other polyprotic acids, loss of a single proton results in the monovalent hydrogenoxalate anion . A salt with this anion is sometimes called an acid oxalate, monobasic oxalate, or hydrogen oxalate. The equilibrium constant ( ''K''a) for loss of the first proton is (p''K''a = 1.27). The loss of the second proton, which yields the oxalate ion, has an equilibrium constant of (p''K''a = 4.28). These values imply, in solutions with neutral pH, no oxalic acid and only tra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylene Glycol Poisoning
Ethylene glycol poisoning is poisoning caused by drinking ethylene glycol. Early symptoms include intoxication, vomiting and abdominal pain. Later symptoms may include a decreased level of consciousness, headache, and seizures. Long term outcomes may include kidney failure and brain damage. Toxicity and death may occur after drinking even in a small amount as ethylene glycol is more toxic than other diols. Ethylene glycol is a colorless, odorless, sweet liquid, commonly found in antifreeze. It may be drunk accidentally or intentionally in a suicide attempt. When broken down by the body it results in glycolic acid and oxalic acid which cause most of the toxicity. The diagnosis may be suspected when calcium oxalate crystals are seen in the urine or when acidosis or an increased osmol gap is present in the blood. Diagnosis may be confirmed by measuring ethylene glycol levels in the blood; however, many hospitals do not have the ability to perform this test. Early treatment in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |