|
Equivalent Potential Temperature
Equivalent potential temperature, commonly referred to as theta-e \left( \theta_e \right), is a quantity that is conserved during changes to an air parcel's pressure (that is, during vertical motions in the atmosphere), even if water vapor condenses during that pressure change. It is therefore more conserved than the ordinary potential temperature, which remains constant only for unsaturated vertical motions (pressure changes). \theta_e is the temperature a parcel of air would reach if all the water vapor in the parcel were to condense, releasing its latent heat, and the parcel was brought adiabatically to a standard reference pressure, usually 1000 hPa (1000 mbar) which is roughly equal to atmospheric pressure at sea level. Its use in estimating atmospheric stability Stability of incompressible fluid Like a ball balanced on top of a hill, denser fluid lying above less dense fluid would be dynamically unstable: overturning motions (convection) can lower the center of gravity, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Earth's Atmosphere
The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth protects life on Earth by creating pressure allowing for liquid water to exist on the Earth's surface, absorbing ultraviolet solar radiation, warming the surface through heat retention (greenhouse effect), and reducing temperature extremes between day and night (the diurnal temperature variation). By mole fraction (i.e., by number of molecules), dry air contains 78.08% nitrogen, 20.95% oxygen, 0.93% argon, 0.04% carbon dioxide, and small amounts of other gases. Air also contains a variable amount of water vapor, on average around 1% at sea level, and 0.4% over the entire atmosphere. Air composition, temperature, and atmospheric pressure vary with altitude. Within the atmosphere, air suitable for use in photosynthesis by terrestrial plants and breathing of terrestrial animals is found only in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Internal Energy
The internal energy of a thermodynamic system is the total energy contained within it. It is the energy necessary to create or prepare the system in its given internal state, and includes the contributions of potential energy and internal kinetic energy. It keeps account of the gains and losses of energy of the system that are due to changes in its internal state. It does not include the kinetic energy of motion of the system as a whole, or any external energies from surrounding force fields. The internal energy of an isolated system is constant, which is expressed as the law of conservation of energy, a foundation of the first law of thermodynamics. The internal energy is an extensive property. The internal energy cannot be measured directly and knowledge of all its components is rarely interesting, such as the static rest mass energy of its constituent matter. Thermodynamics is chiefly concerned only with ''changes'' in the internal energy, not with its absolute value. Instea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
AMDAR
Aircraft Meteorological Data Relay (AMDAR) is a program initiated by the World Meteorological Organization. AMDAR is used to collect meteorological data worldwide by using commercial aircraft. Data is collected by the aircraft navigation systems and the onboard standard temperature and static pressure probes. The data is then preprocessed before linking them down to the ground either via VHF communication (ACARS) or via satellite link ASDAR. A detailed description is given in the AMDAR Reference Manual (WMO-No 958) available from the World Meteorological Organization, Geneva, Switzerlandhttps://library.wmo.int/doc_num.php?explnum_id=9026 Usage AMDAR transmissions are most commonly used in forecast models as a supplement to radiosonde A radiosonde is a battery-powered telemetry instrument carried into the atmosphere usually by a weather balloon that measures various atmospheric parameters and transmits them by radio to a ground receiver. Modern radiosondes measure or cal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Relative Humidity
Humidity is the concentration of water vapor present in the air. Water vapor, the gaseous state of water, is generally invisible to the human eye. Humidity indicates the likelihood for precipitation, dew, or fog to be present. Humidity depends on the temperature and pressure of the system of interest. The same amount of water vapor results in higher relative humidity in cool air than warm air. A related parameter is the dew point. The amount of water vapor needed to achieve saturation increases as the temperature increases. As the temperature of a parcel of air decreases it will eventually reach the saturation point without adding or losing water mass. The amount of water vapor contained within a parcel of air can vary significantly. For example, a parcel of air near saturation may contain 28 g of water per cubic metre of air at , but only 8 g of water per cubic metre of air at . Three primary measurements of humidity are widely employed: absolute, relative, and specific. Ab ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mixing Ratio
In chemistry and physics, the dimensionless mixing ratio is the abundance of one component of a mixture relative to that of all other components. The term can refer either to mole ratio (see concentration) or mass ratio (see stoichiometry). In atmospheric chemistry and meteorology Mole ratio In atmospheric chemistry, mixing ratio usually refers to the mole ratio ''ri'', which is defined as the amount of a constituent ''ni'' divided by the total amount of all ''other'' constituents in a mixture: :r_i = \frac The mole ratio is also called amount ratio. If ''ni'' is much smaller than ''n''tot (which is the case for atmospheric trace constituents), the mole ratio is almost identical to the mole fraction. Mass ratio In meteorology, mixing ratio usually refers to the mass ratio of water \zeta, which is defined as the mass of water m_\mathrm divided by the mass of dry air (m_\mathrm-m_\mathrm) in a given air parcel: :\zeta = \frac The unit is typically given in \mathrm\,\mathrm^. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Specific Heat Capacity
In thermodynamics, the specific heat capacity (symbol ) of a substance is the heat capacity of a sample of the substance divided by the mass of the sample, also sometimes referred to as massic heat capacity. Informally, it is the amount of heat that must be added to one unit of mass of the substance in order to cause an increase of one unit in temperature. The SI unit of specific heat capacity is joule per kelvin per kilogram, J⋅kg−1⋅K−1. For example, the heat required to raise the temperature of of water by is , so the specific heat capacity of water is . Specific heat capacity often varies with temperature, and is different for each state of matter. Liquid water has one of the highest specific heat capacities among common substances, about at 20 °C; but that of ice, just below 0 °C, is only . The specific heat capacities of iron, granite, and hydrogen gas are about 449 J⋅kg−1⋅K−1, 790 J⋅kg−1⋅K−1, and 14300 J⋅kg−1⋅K−1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gas Constant
The molar gas constant (also known as the gas constant, universal gas constant, or ideal gas constant) is denoted by the symbol or . It is the molar equivalent to the Boltzmann constant, expressed in units of energy per temperature increment per amount of substance, i.e. the pressure–volume product, rather than energy per temperature increment per ''particle''. The constant is also a combination of the constants from Boyle's law, Charles's law, Avogadro's law, and Gay-Lussac's law. It is a physical constant that is featured in many fundamental equations in the physical sciences, such as the ideal gas law, the Arrhenius equation, and the Nernst equation. The gas constant is the constant of proportionality that relates the energy scale in physics to the temperature scale and the scale used for amount of substance. Thus, the value of the gas constant ultimately derives from historical decisions and accidents in the setting of units of energy, temperature and amount of substa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
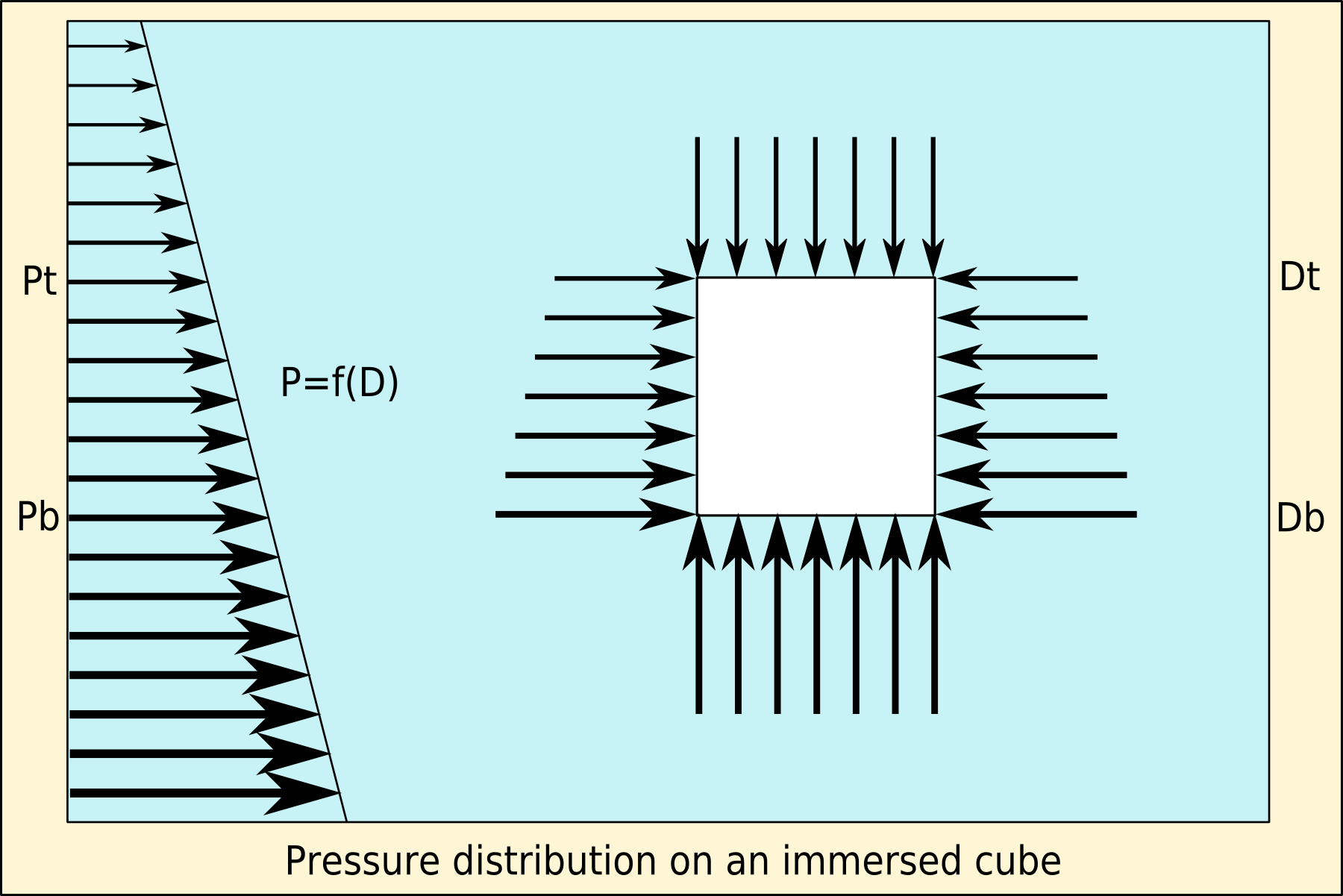
Buoyancy
Buoyancy (), or upthrust, is an upward force exerted by a fluid that opposes the weight of a partially or fully immersed object. In a column of fluid, pressure increases with depth as a result of the weight of the overlying fluid. Thus the pressure at the bottom of a column of fluid is greater than at the top of the column. Similarly, the pressure at the bottom of an object submerged in a fluid is greater than at the top of the object. The pressure difference results in a net upward force on the object. The magnitude of the force is proportional to the pressure difference, and (as explained by Archimedes' principle) is equivalent to the weight of the fluid that would otherwise occupy the submerged volume of the object, i.e. the displaced fluid. For this reason, an object whose average density is greater than that of the fluid in which it is submerged tends to sink. If the object is less dense than the liquid, the force can keep the object afloat. This can occur only in a no ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dry Adiabatic Lapse Rate
The lapse rate is the rate at which an atmospheric variable, normally temperature in Earth's atmosphere, falls with altitude. ''Lapse rate'' arises from the word ''lapse'', in the sense of a gradual fall. In dry air, the adiabatic lapse rate is 9.8 °C/km (5.4 °F per 1,000 ft). It corresponds to the vertical component of the spatial gradient of temperature. Although this concept is most often applied to the Earth's troposphere, it can be extended to any gravitationally supported parcel of gas. Definition A formal definition from the ''Glossary of Meteorology'' is: :The decrease of an atmospheric variable with height, the variable being temperature unless otherwise specified. Typically, the lapse rate is the negative of the rate of temperature change with altitude change: :\Gamma = -\frac where \Gamma (sometimes L) is the lapse rate given in units of temperature divided by units of altitude, ''T'' is temperature, and ''z'' is altitude. Convection and adiabatic expansion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Moist Adiabatic Lapse Rate
The lapse rate is the rate at which an atmospheric variable, normally temperature in Earth's atmosphere, falls with altitude. ''Lapse rate'' arises from the word ''lapse'', in the sense of a gradual fall. In dry air, the adiabatic lapse rate is 9.8 °C/km (5.4 °F per 1,000 ft). It corresponds to the vertical component of the spatial gradient of temperature. Although this concept is most often applied to the Earth's troposphere, it can be extended to any gravitationally supported parcel of gas. Definition A formal definition from the ''Glossary of Meteorology'' is: :The decrease of an atmospheric variable with height, the variable being temperature unless otherwise specified. Typically, the lapse rate is the negative of the rate of temperature change with altitude change: :\Gamma = -\frac where \Gamma (sometimes L) is the lapse rate given in units of temperature divided by units of altitude, ''T'' is temperature, and ''z'' is altitude. Convection and adiabatic expansion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Latent Heat Of Vaporisation
The enthalpy of vaporization (symbol ), also known as the (latent) heat of vaporization or heat of evaporation, is the amount of energy (enthalpy) that must be added to a liquid substance to transform a quantity of that substance into a gas. The enthalpy of vaporization is a function of the pressure at which that transformation takes place. The enthalpy of vaporization is often quoted for the normal boiling temperature of the substance. Although tabulated values are usually corrected to 298 K, that correction is often smaller than the uncertainty in the measured value. The heat of vaporization is temperature-dependent, though a constant heat of vaporization can be assumed for small temperature ranges and for reduced temperature T_r \ll 1. The heat of vaporization diminishes with increasing temperature and it vanishes completely at a certain point called the critical temperature (T_r = 1). Above the critical temperature, the liquid and vapor phases are indistinguishable ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clausius–Clapeyron Relation
The Clausius–Clapeyron relation, named after Rudolf Clausius and Benoît Paul Émile Clapeyron, specifies the temperature dependence of pressure, most importantly vapor pressure, at a discontinuous phase transition between two phases of matter of a single constituent. Its relevance to meteorology and climatology is the increase of the water-holding capacity of the atmosphere by about 7% for every 1 °C (1.8 °F) rise in temperature. Definition On a pressure–temperature (''P''–''T'') diagram, the line separating the two phases is known as the coexistence curve. The Clapeyron relation gives the slope of the tangents to this curve. Mathematically, :\frac = \frac=\frac, where \mathrmP/\mathrmT is the slope of the tangent to the coexistence curve at any point, L is the specific latent heat, T is the temperature, \Delta v is the specific volume change of the phase transition, and \Delta s is the specific entropy change of the phase transition. The Clausius–Clape ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |