|
Dependence Receptor
In cellular biology, dependence receptors are proteins that mediate programmed cell death by monitoring the absence of certain trophic factors (or, equivalently, the presence of anti-trophic factors) that otherwise serve as ligands (interactors) for the dependence receptors. A trophic ligand is a molecule whose protein binding stimulates cell growth, differentiation, and/or survival. Cells depend for their survival on stimulation that is mediated by various receptors and sensors, and integrated via signaling within the cell and between cells. The withdrawal of such trophic support leads to a form of cellular suicide. Various dependence receptors are involved in a range of biological events: developmental cell death (naturally occurring cell death), trophic factor withdrawal-induced cell death, the spontaneous regression characteristic of type IV-S neuroblastoma, neurodegenerative cell death, inhibition of new tumor cells (tumorigenesis) and metastasis, and therapeutic antibody ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proteins
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid residues ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
NEO1
Neogenin is a protein that in humans is encoded by the ''NEO1'' gene. Interactions NEO1 has been shown to interact with PTK2 PTK2 protein tyrosine kinase 2 (PTK2), also known as focal adhesion kinase (FAK), is a protein that, in humans, is encoded by the ''PTK2'' gene. PTK2 is a focal adhesion-associated protein kinase involved in cellular adhesion (how cells stick .... Also Neogenin receptor is pointed as a component of the mechanisms that determine skeletal cell fusion via RGMa (repulsive guidance molecule a) binding. References Further reading * * * * * * * * * * Human proteins {{gene-15-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Integrins
Integrins are transmembrane receptors that facilitate cell-cell and cell-extracellular matrix (ECM) adhesion. Upon ligand binding, integrins activate signal transduction pathways that mediate cellular signals such as regulation of the cell cycle, organization of the intracellular cytoskeleton, and movement of new receptors to the cell membrane. The presence of integrins allows rapid and flexible responses to events at the cell surface (''e.g''. signal platelets to initiate an interaction with coagulation factors). Several types of integrins exist, and one cell generally has multiple different types on its surface. Integrins are found in all animals while integrin-like receptors are found in plant cells. Integrins work alongside other proteins such as cadherins, the immunoglobulin superfamily cell adhesion molecules, selectins and syndecans, to mediate cell–cell and cell–matrix interaction. Ligands for integrins include fibronectin, vitronectin, collagen and laminin. Structur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Androgen Receptor
The androgen receptor (AR), also known as NR3C4 (nuclear receptor subfamily 3, group C, member 4), is a type of nuclear receptor that is activated by binding any of the androgenic hormones, including testosterone and dihydrotestosterone in the cytoplasm and then translocating into the nucleus. The androgen receptor is most closely related to the progesterone receptor, and progestins in higher dosages can block the androgen receptor. The main function of the androgen receptor is as a DNA-binding transcription factor that regulates gene expression; however, the androgen receptor has other functions as well. Androgen-regulated genes are critical for the development and maintenance of the male sexual phenotype. Function Effect on development In some cell types, testosterone interacts directly with androgen receptors, whereas, in others, testosterone is converted by 5-alpha-reductase to dihydrotestosterone, an even more potent agonist for androgen receptor activation. T ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anaplastic Lymphoma Kinase
Anaplastic lymphoma kinase (ALK) also known as ALK tyrosine kinase receptor or CD246 (cluster of differentiation 246) is an enzyme that in humans is encoded by the ''ALK'' gene. Identification Anaplastic lymphoma kinase (ALK) was originally discovered in 1994 in anaplastic large-cell lymphoma (ALCL) cells. ALCL is caused by a (2;5)(p23:q35) chromosomal translocation that generates the fusion protein NPM-ALK, in which the kinase domain of ALK is fused to the amino-terminal part of the nucleophosmin (NPM) protein. Dimer (chemistry), Dimerization of NPM constitutively activates the ALK kinase domain. The full-length protein ALK was identified in 1997 by two groups. The deduced amino acid sequences revealed that ALK was a novel receptor tyrosine kinase (RTK), having an extracellular domain, extracellular ligand-binding domain, a transmembrane domain, and an intracellular tyrosine kinase domain. While the tyrosine kinase domain of human ALK shares a high degree of similarity with tha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Insulin-like Growth Factor 1 Receptor
The insulin-like growth factor 1 (IGF-1) receptor is a protein found on the surface of human cells. It is a transmembrane receptor that is activated by a hormone called insulin-like growth factor 1 ( IGF-1) and by a related hormone called IGF-2. It belongs to the large class of tyrosine kinase receptors. This receptor mediates the effects of IGF-1, which is a polypeptide protein hormone similar in molecular structure to insulin. IGF-1 plays an important role in growth and continues to have anabolic effects in adults – meaning that it can induce hypertrophy of skeletal muscle and other target tissues. Mice lacking the IGF-1 receptor die late in development, and show a dramatic reduction in body mass. This testifies to the strong growth-promoting effect of this receptor. Structure Two alpha subunits and two beta subunits make up the IGF-1 receptor. Both the α and β subunits are synthesized from a single mRNA precursor. The precursor is then glycosylated, proteolytically ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
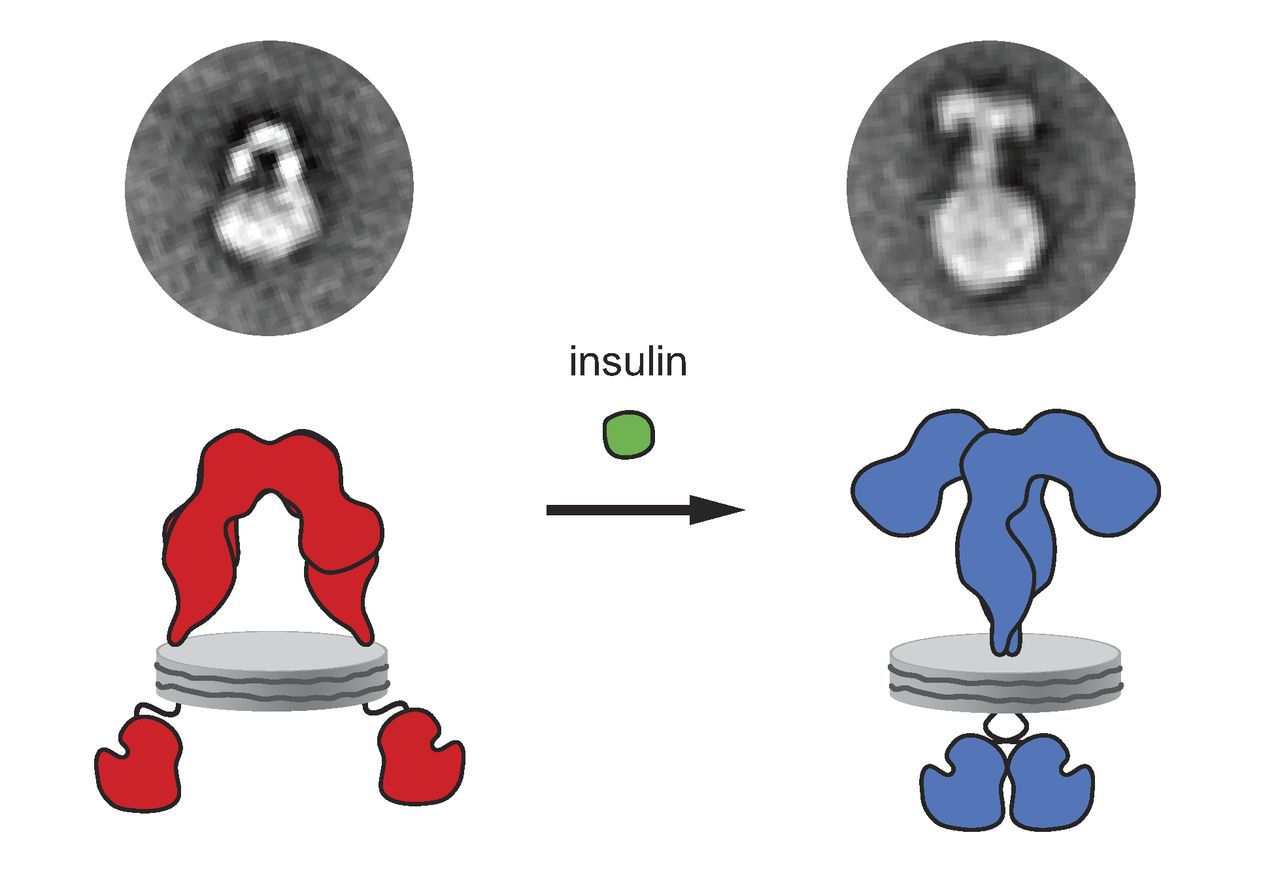
Insulin Receptor
The insulin receptor (IR) is a transmembrane receptor that is activated by insulin, IGF-I, IGF-II and belongs to the large class of receptor tyrosine kinase. Metabolically, the insulin receptor plays a key role in the regulation of glucose homeostasis, a functional process that under degenerate conditions may result in a range of clinical manifestations including diabetes and cancer. Insulin signalling controls access to blood glucose in body cells. When insulin falls, especially in those with high insulin sensitivity, body cells begin only to have access to lipids that do not require transport across the membrane. So, in this way, insulin is the key regulator of fat metabolism as well. Biochemically, the insulin receptor is encoded by a single gene , from which alternate splicing during transcription results in either IR-A or IR-B isoforms. Downstream post-translational events of either isoform result in the formation of a proteolytically cleaved α and β subunit, which upon com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
C-Met
c-Met, also called tyrosine-protein kinase Met or hepatocyte growth factor receptor (HGFR), is a protein that in humans is encoded by the ''MET'' gene. The protein possesses tyrosine kinase activity. The primary single chain precursor protein is post-translationally cleaved to produce the alpha and beta subunits, which are disulfide linked to form the mature receptor. MET is a single pass tyrosine kinase receptor essential for embryonic development, organogenesis and wound healing. Hepatocyte growth factor/Scatter Factor (HGF/SF) and its splicing isoform (NK1, NK2) are the only known ligands of the MET receptor. MET is normally expressed by cells of epithelial origin, while expression of HGF/SF is restricted to cells of mesenchymal origin. When HGF/SF binds its cognate receptor MET it induces its dimerization through a not yet completely understood mechanism leading to its activation. Abnormal MET activation in cancer correlates with poor prognosis, where aberrantly active MET t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
EPH Receptor A4
EPH receptor A4 (ephrin type-A receptor 4) is a protein that in humans is encoded by the ''EPHA4'' gene. This gene belongs to the ephrin receptor subfamily of the protein-tyrosine kinase family. EPH and EPH-related receptors have been implicated in mediating developmental events, particularly in the nervous system. Receptors in the EPH subfamily typically have a single kinase domain and an extracellular region containing a Cys-rich domain and 2 fibronectin type III repeats. The ephrin receptors are divided into 2 groups based on the similarity of their extracellular domain sequences and their affinities for binding ephrin-A and ephrin-B ligands. In 2012, a publication in ''Nature Medicine'' revealed a connection between EPHA4 and the neurodegenerative disease Amyotrophic lateral sclerosis Amyotrophic lateral sclerosis (ALS), also known as motor neuron disease (MND) or Lou Gehrig's disease, is a neurodegenerative disease that results in the progressive loss of motor neurons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TrkC Receptor
Tropomyosin receptor kinase C (TrkC), also known as NT-3 growth factor receptor, neurotrophic tyrosine kinase receptor type 3, or TrkC tyrosine kinase is a protein that in humans is encoded by the ''NTRK3'' gene. TrkC is the high affinity catalytic receptor for the neurotrophin NT-3 (neurotrophin-3). As such, TrkC mediates the multiple effects of this neurotrophic factor, which includes neuronal differentiation and survival. The TrkC receptor is part of the large family of receptor tyrosine kinases. A "tyrosine kinase" is an enzyme which is capable of adding a phosphate group to the certain tyrosines on target proteins, or "substrates". A receptor tyrosine kinase is a "tyrosine kinase" which is located at the cellular membrane, and is activated by binding of a ligand via its extracellular domain. Other example of tyrosine kinase receptors include the insulin receptor, the IGF-1 receptor, the MuSK protein receptor, the vascular endothelial growth factor (VEGF) receptor, etc. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TrkA Receptor
Tropomyosin receptor kinase A (TrkA), also known as high affinity nerve growth factor receptor, neurotrophic tyrosine kinase receptor type 1, or TRK1-transforming tyrosine kinase protein is a protein that in humans is encoded by the ''NTRK1'' gene. This gene encodes a member of the neurotrophic tyrosine kinase receptor (NTKR) family. This kinase is a membrane-bound receptor that, upon neurotrophin binding, phosphorylates itself (autophosphorylation) and members of the MAPK pathway. The presence of this kinase leads to cell differentiation and may play a role in specifying sensory neuron subtypes. Mutations in this gene have been associated with congenital insensitivity to pain with anhidrosis, self-mutilating behaviors, intellectual disability and/or cognitive impairment and certain cancers. Alternate transcriptional splice variants of this gene have been found, but only three have been characterized to date. Function and Interaction with NGF TrkA is the high affinity cataly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
RET Proto-oncogene
The ''RET'' proto-oncogene encodes a receptor tyrosine kinase for members of the glial cell line-derived neurotrophic factor (GDNF) family of extracellular signalling molecules. ''RET'' loss of function mutations are associated with the development of Hirschsprung's disease, while gain of function mutations are associated with the development of various types of human cancer, including medullary thyroid carcinoma, multiple endocrine neoplasias type 2A and 2B, pheochromocytoma and parathyroid hyperplasia. Structure ''RET'' is an abbreviation for "rearranged during transfection", as the DNA sequence of this gene was originally found to be rearranged within a 3T3 fibroblast cell line following its transfection with DNA taken from human lymphoma cells. The human gene ''RET'' is localized to chromosome 10 (10q11.2) and contains 21 exons. The natural alternative splicing of the ''RET'' gene results in the production of 3 different isoforms of the protein RET. RET51, RET43 and RET9 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |