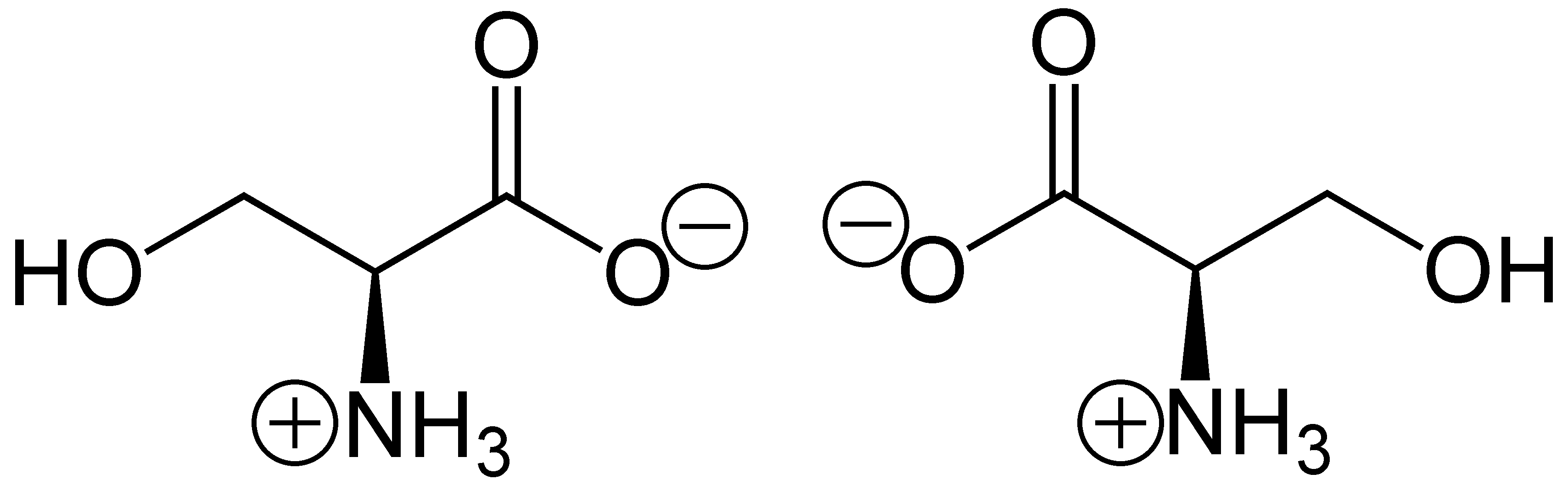|
DUSP28
Dual-specificity phosphatase (DUSP; DSP) is a form of phosphatase that can act upon tyrosine or serine/threonine residues. There are several families of dual-specificity phosphatase enzymes in mammals. All share a similar catalytic mechanism, by which a conserved cysteine residue forms a covalent intermediate with the phosphate group to be eliminated. The residues surrounding their catalytic core obey a rather strict consensus: His-Cys-x-x-x-x-x-Arg-Ser. The serine side chain and an additional conserved aspartate play a central role in the elimination of the Cys-linked intermediate, thus completing their enzymatic cycle. The main difference between tyrosine-specific phosphatases and dual-specificity phosphatases lies in the width of the latter enzymes' catalytic pocket: thus they can accommodate phosphorylated serine or threonine side chains as well as phosphorylated tyrosines. Classification The human genome encodes at least 61 different DUSP proteins. The following major groups ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphatase
In biochemistry, a phosphatase is an enzyme that uses water to cleave a phosphoric acid Ester, monoester into a phosphate ion and an Alcohol (chemistry), alcohol. Because a phosphatase enzyme catalysis, catalyzes the hydrolysis of its Substrate (chemistry), substrate, it is a subcategory of hydrolases. Phosphatase enzymes are essential to many biological functions, because phosphorylation (e.g. by protein kinases) and dephosphorylation (by phosphatases) serve diverse roles in cell growth, cellular regulation and cell signaling, signaling. Whereas phosphatases remove phosphate groups from molecules, kinases catalyze the transfer of phosphate groups to molecules from Adenosine triphosphate, ATP. Together, kinases and phosphatases direct a form of post-translational modification that is essential to the cell's regulatory network. Phosphatase enzymes are not to be confused with phosphorylase enzymes, which catalyze the transfer of a phosphate group from hydrogen phosphate to an acce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek ''tyrós'', meaning ''cheese'', as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a Hydrophobe, hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is Genetic code, encoded by the Genetic code#Codons, codons UAC and UAU in messenger RNA. Functions Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases. Phosphorylation of the hyd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serine
Serine (symbol Ser or S) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated − form under biological conditions), a carboxyl group (which is in the deprotonated − form under biological conditions), and a side chain consisting of a hydroxymethyl group, classifying it as a polar amino acid. It can be synthesized in the human body under normal physiological circumstances, making it a nonessential amino acid. It is encoded by the codons UCU, UCC, UCA, UCG, AGU and AGC. Occurrence This compound is one of the naturally occurring proteinogenic amino acids. Only the L-stereoisomer appears naturally in proteins. It is not essential to the human diet, since it is synthesized in the body from other metabolites, including glycine. Serine was first obtained from silk protein, a particularly rich source, in 1865 by Emil Cramer. Its name is derived from the Latin for silk, ''sericum''. Serine's structure was estab ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Threonine
Threonine (symbol Thr or T) is an amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form under biological conditions), a carboxyl group (which is in the deprotonated −COO− form under biological conditions), and a side chain containing a hydroxyl group, making it a polar, uncharged amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Threonine is synthesized from aspartate in bacteria such as ''E. coli''. It is encoded by all the codons starting AC (ACU, ACC, ACA, and ACG). Threonine sidechains are often hydrogen bonded; the most common small motifs formed are based on interactions with serine: ST turns, ST motifs (often at the beginning of alpha helices) and ST staples (usually at the middle of alpha helices). Modifications The threonine residue is susceptible to numerous posttranslational modifications. The hydroxyl side-chain can unde ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SSH1
''For the SSH-1 protocol, see Secure Shell#Version 1'' Protein phosphatase Slingshot homolog 1 is an enzyme that in humans is encoded by the ''SSH1'' gene In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba .... The ADF (actin-depolymerizing factor)/cofilin family (see MIM 601442) is composed of stimulus-responsive mediators of actin dynamics. ADF/cofilin proteins are inactivated by kinases such as LIM domain kinase-1 (LIMK1; MIM 601329). The SSH family appears to play a role in actin dynamics by reactivating ADF/cofilin proteins in vivo (Niwa et al., 2002). upplied by OMIMref name="entrez"/> References Further reading * * * * * * * * * * * * * * * * * {{gene-12-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SSH2
Protein phosphatase Slingshot homolog 2 is an enzyme that in humans is encoded by the ''SSH2'' gene In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba .... The ADF (actin-depolymerizing factor)/cofilin family (see MIM 601442) is composed of stimulus-responsive mediators of actin dynamics. ADF/cofilin proteins are inactivated by kinases such as LIM domain kinase-1 (LIMK1; MIM 601329). The SSH family appears to play a role in actin dynamics by reactivating ADF/cofilin proteins in vivo (Niwa et al., 2002). upplied by OMIMref name="entrez"> References Further reading * * * * * * * * * * External links * {{gene-17-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SSH3
Protein phosphatase Slingshot homolog 3 is an enzyme that in humans is encoded by the ''SSH3'' gene. Function The ADF (actin-depolymerizing factor)/cofilin family (see MIM 601442) is composed of stimulus-responsive mediators of actin dynamics. ADF/cofilin proteins are inactivated by kinases such as LIM domain kinase-1 (LIMK1 LIM domain kinase 1 is an enzyme that in humans is encoded by the ''LIMK1'' gene. Function There are approximately 40 known eukaryotic LIM proteins, so named for the LIM domains they contain. LIM domains are highly conserved cysteine-rich struc ...; MIM 601329). The SSH family appears to play a role in actin dynamics by reactivating ADF/cofilin proteins in vivo (Niwa et al., 2002). upplied by OMIMref name="entrez"> References Further reading * * * * {{gene-11-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cdc14
''Cdc14'' and Cdc14 are a gene and its protein product respectively. Cdc14 is found in most of the eukaryotes. Cdc14 was defined by Hartwell in his famous screen for loci that control the cell cycle of Saccharomyces cerevisiae. Cdc14 was later shown to encode a protein phosphatase. Cdc14 is dual-specificity, which means it has serine/threonine and tyrosine-directed activity. A preference for serines next to proline is reported. Many early studies, especially in the budding yeast Saccharomyces cerevisiae, demonstrated that the protein plays a key role in regulating late mitotic processes. However, more recent work in a range of systems suggests that its cellular function is more complex. Cellular function In Saccharomyces cerevisiae, the species in which Cdc14 activity is best understood and most-studied, the activity of Cdc14 (ScCdc14) leads to mitotic exit by dephosphorylating targets of Cdk1, a well-studied cyclin-dependent protein kinase. Cdc14 antagonizes Cdk1 by stimula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PTEN (gene)
Phosphatase and tensin homolog (PTEN) is a phosphatase in humans and is encoded by the ''PTEN'' gene. Mutations of this gene are a step in the development of many cancers, specifically glioblastoma, lung cancer, breast cancer, and prostate cancer. Genes corresponding to PTEN (orthologs) have been identified in most mammals for which complete genome data are available. ''PTEN'' acts as a tumor suppressor gene through the action of its phosphatase protein product. This phosphatase is involved in the regulation of the cell cycle, preventing cells from growing and dividing too rapidly. It is a target of many anticancer drugs. The protein encoded by this gene is a phosphatidylinositol-3,4,5-trisphosphate 3-phosphatase. It contains a tensin-like domain as well as a catalytic domain similar to that of the dual specificity protein tyrosine phosphatases. Unlike most of the protein tyrosine phosphatases, this protein preferentially dephosphorylates phosphoinositide substrates. It nega ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
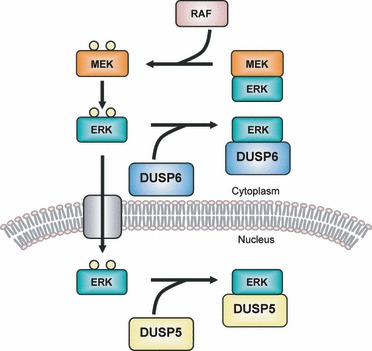
MAPK Phosphatase
MAPK phosphatases (MKPs) are the largest class of phosphatases involved in down-regulating Mitogen-activated protein kinases (MAPK) signaling. MAPK signalling pathways regulate multiple features of development and homeostasis. This can involve gene regulation, cell proliferation, programmed cell death and stress responses. MAPK phosphatases are therefore important regulator components of these pathways. Function MAPK phosphatases are only found in eukaryotes and negatively regulate MAP kinases to act as negative feedback. MKPs are also known as dual-specificity phosphatases (DUSPs) because they deactivate MAPK by dephosphorylating the Threonine and the Tyrosine residues residing in MAPKs activation site. MKPs have a catalytic region at their C-terminus and a regulatory region at their N-terminus. The position where the MAPK binds to MKP is found near the N-terminus of MKP. The binding is due to the electrostatic interactions of the positively charged residues on the MKP ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |