|
Dianin's Compound
Dianin's compound (4-''p''-hydroxyphenyl-2,2,4-trimethylchroman) was first prepared by Aleksandr Dianin in 1914. This compound is a condensation isomer of bisphenol A and acetone and of special importance in host–guest chemistry because it can form a large variety of clathrates with suitable guest molecules. One example is the clathrate of Dianin's compound with morpholine. Slow evaporation of a solution containing both organic compounds yields crystals. Each asymmetric unit cell making up the crystal contains six chroman molecules of which two are deprotonated and two protonated morpholine molecules. The six chroman molecules are racemate In chemistry, a racemic mixture, or racemate (), is one that has equal amounts of left- and right-handed enantiomers of a chiral molecule or salt. Racemic mixtures are rare in nature, but many compounds are produced industrially as racemates. ... pairs. References {{Reflist Clathrates Phenols Russian inventions Chromanes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aleksandr Dianin
Aleksandr Pavlovich Dianin (russian: Александр Павлович Дианин; 20 April 1851 – 6 December 1918) was a Russian chemist from Saint Petersburg. He carried out studies on phenols and discovered a phenol derivative now known as bisphenol A and the accordingly named Dianin's compound. He was married to the adopted daughter of fellow chemist Alexander Borodin. In 1887, Dianin succeeded his father-in-law as chair of the Chemistry Department at the Imperial Medical-Surgical Academy in St. Petersburg (now the S.M. Kirov Military Medical Academy). Bisphenol A and Dianin's compound Dianin's method for preparing bisphenol A from 1891 remains the most widely-known approach to this important compound, though the method has been refined for industrial-scale synthesis. It involves the catalysed condensation of a 2:1 mixture of phenol and acetone in the presence of concentrated hydrochloric acid or sulfuric acid. The reaction proceeds readily at room temperature produci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Condensation Reaction
In organic chemistry, a condensation reaction is a type of chemical reaction in which two molecules are combined to form a single molecule, usually with the loss of a small molecule such as water. If water is lost, the reaction is also known as a dehydration synthesis. However other molecules can also be lost, such as ammonia, ethanol, acetic acid and hydrogen sulfide. The addition of the two molecules typically proceeds in a step-wise fashion to the addition product, usually in equilibrium, and with loss of a water molecule (hence the name condensation). The reaction may otherwise involve the functional groups of the molecule, and is a versatile class of reactions that can occur in acidic or basic conditions or in the presence of a catalyst. This class of reactions is a vital part of life as it is essential to the formation of peptide bonds between amino acids and to the biosynthesis of fatty acids. Many variations of condensation reactions exist. Common examples include the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomer
In chemistry, isomers are molecules or polyatomic ions with identical molecular formulae – that is, same number of atoms of each element – but distinct arrangements of atoms in space. Isomerism is existence or possibility of isomers. Isomers do not necessarily share similar chemical or physical properties. Two main forms of isomerism are structural or constitutional isomerism, in which ''bonds'' between the atoms differ; and stereoisomerism or spatial isomerism, in which the bonds are the same but the ''relative positions'' of the atoms differ. Isomeric relationships form a hierarchy. Two chemicals might be the same constitutional isomer, but upon deeper analysis be stereoisomers of each other. Two molecules that are the same stereoisomer as each other might be in different conformational forms or be different isotopologues. The depth of analysis depends on the field of study or the chemical and physical properties of interest. The English word "isomer" () is a back-for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bisphenol A
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water. BPA is produced on an industrial scale by the condensation of phenol and acetone, and has a global production scale which is expected to reach 10 million tonnes in 2022. BPA's largest single application is as a co-monomer in the production of polycarbonates, which accounts for 65–70% of all BPA production. The manufacturing of epoxy resins and vinyl ester resins account for 25–30% of BPA use. The remaining 5% is used as a major component of several high-performance plastics, and as a minor additive in PVC, polyurethane, thermal paper, and several other materials. It is not a plasticizer, although it is often wrongly labelled as such. The health effects of BPA have been the subject of prolonged public and scientific debate. BPA is a xenoestrogen, exhibiting hormone ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetone
Acetone (2-propanone or dimethyl ketone), is an organic compound with the formula . It is the simplest and smallest ketone (). It is a colorless, highly volatile and flammable liquid with a characteristic pungent odour. Acetone is miscible with water and serves as an important organic solvent in its own right, in industry, home, and laboratory. About 6.7 million tonnes were produced worldwide in 2010, mainly for use as a solvent and production of methyl methacrylate (and from that PMMA) as well as bisphenol A.Acetone World Petrochemicals report, January 2010Stylianos Sifniades, Alan B. Levy, "Acetone" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. It is a common building block in |
Host–guest Chemistry
In supramolecular chemistry, host–guest chemistry describes complexes that are composed of two or more molecules or ions that are held together in unique structural relationships by forces other than those of full covalent bonds. Host–guest chemistry encompasses the idea of molecular recognition and interactions through non-covalent bonding. Non-covalent bonding is critical in maintaining the 3D structure of large molecules, such as proteins and is involved in many biological processes in which large molecules bind specifically but transiently to one another. Although non-covalent interactions could be roughly divided into those with more electrostatic or dispersive contributions, there are few commonly mentioned types of non-covalent interactions: ionic bonding, hydrogen bonding, van der Waals forces and hydrophobic interactions. Overview Host–guest chemistry is a branch of supramolecular chemistry in which a host molecule forms a chemical compound with a guest molecul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Clathrate Compound
A clathrate is a chemical substance consisting of a lattice that traps or contains molecules. The word ''clathrate'' is derived from the Latin (), meaning ‘with bars, latticed’. Most clathrate compounds are polymeric and completely envelop the guest molecule, but in modern usage clathrates also include host–guest complexes and inclusion compounds.Atwood, J. L. (2012) "Inclusion Compounds" in ''Ullmann's Encyclopedia of Industrial Chemistry''. Wiley-VCH, Weinheim. According to IUPAC, clathrates are inclusion compounds "in which the guest molecule is in a cage formed by the host molecule or by a lattice of host molecules." The term refers to many molecular hosts, including calixarenes and cyclodextrins and even some inorganic polymers such as zeolites. Clathrates can be divided into two categories: clathrate hydrates and inorganic clathrates. Each clathrate is made up of a framework and guests that reside the framework. Most common clathrate crystal structures can be compos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Morpholine
Morpholine is an organic chemical compound having the chemical formula O( C H2CH2)2 NH. This heterocycle features both amine and ether functional groups. Because of the amine, morpholine is a base; its conjugate acid is called morpholinium. For example, treating morpholine with hydrochloric acid makes the salt morpholinium chloride. It is a colorless liquid with a weak, ammonia- or fish-like odor. The naming of morpholine is attributed to Ludwig Knorr, who incorrectly believed it to be part of the structure of morphine. Production Morpholine is often produced industrially by the dehydration of diethanolamine with sulfuric acid: : Uses Industrial applications Morpholine is a common additive, in parts per million concentrations, for pH adjustment in both fossil fuel and nuclear power plant steam systems. Morpholine is used because its volatility is about the same as water, so once it is added to the water, its concentration becomes distributed rather evenly in both the water a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Communications
''ChemComm'' (or ''Chemical Communications''), formerly known as ''Journal of the Chemical Society D: Chemical Communications'' (1969–1971), ''Journal of the Chemical Society, Chemical Communications'' (1972–1995), is a peer-reviewed scientific journal published by the Royal Society of Chemistry. It covers all aspects of chemistry. In January 2012, the journal moved to publishing 100 issues per year. The current chair of the Editorial Board is Douglas Stephan (University of Toronto, Canada), while the executive editor is Richard Kelly. Abstracting and indexing The journal is abstracted and indexed in: * Chemical Abstracts * Science Citation Index * Current Contents/Physical, Chemical & Earth Sciences * Scopus * Index Medicus/MEDLINE/PubMed According to the ''Journal Citation Reports'', the journal has a 2021 impact factor of 6.065. See also * ''New Journal of Chemistry'' * ''Chemical Society Reviews'' * ''Chemical Science'' * ''RSC Advances ''RSC Advances'' is an online ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Living t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
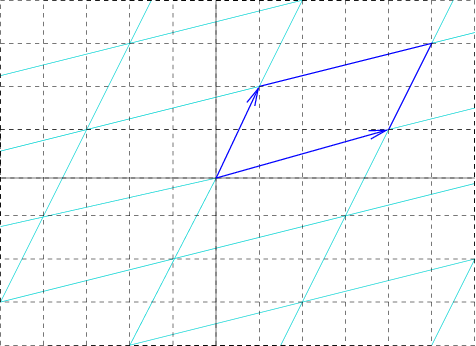
Unit Cell
In geometry, biology, mineralogy and solid state physics, a unit cell is a repeating unit formed by the vectors spanning the points of a lattice. Despite its suggestive name, the unit cell (unlike a unit vector, for example) does not necessarily have unit size, or even a particular size at all. Rather, the primitive cell is the closest analogy to a unit vector, since it has a determined size for a given lattice and is the basic building block from which larger cells are constructed. The concept is used particularly in describing crystal structure in two and three dimensions, though it makes sense in all dimensions. A lattice can be characterized by the geometry of its unit cell, which is a section of the tiling (a parallelogram or parallelepiped) that generates the whole tiling using only translations. There are two special cases of the unit cell: the primitive cell and the conventional cell. The primitive cell is a unit cell corresponding to a single lattice point, it is the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protonation
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), (H+) to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, is deprotonation.) Some examples include *The protonation of water by sulfuric acid: *:H2SO4 + H2O H3O+ + *The protonation of isobutene in the formation of a carbocation: *:(CH3)2C=CH2 + HBF4 (CH3)3C+ + *The protonation of ammonia in the formation of ammonium chloride from ammonia and hydrogen chloride: *:NH3( g) + HCl( g) → NH4Cl( s) Protonation is a fundamental chemical reaction and is a step in many stoichiometric and catalytic processes. Some ions and molecules can undergo more than one protonation and are labeled polybasic, which is true of many biological macromolecules. Protonation and deprotonation (removal of a proton) occur in most acid–base reactions; they are the core of most acid–base reaction theories. A Brønst ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |