|
Cyanometalate
Cyanometallates or cyanometalates are a class of coordination compounds, most often consisting only of cyanide ligands. Most are anions. Cyanide is a highly basic and small ligand, hence it readily saturates the coordination sphere of metal ions. The resulting cyanometallate anions are often used as building blocks for more complex structures called coordination polymers, the best known example of which is Prussian blue, a common dyestuff.*Dunbar, K. R. and Heintz, R. A., "Chemistry of Transition Metal Cyanide Compounds: Modern Perspectives", Progress in Inorganic Chemistry, 1997, 45, 283-391. Examples Homoleptic cyanometallates ''Homoleptic'' cyanometallates are complexes where the only ligand is cyanide. For transition metals, well known homoleptic cyanometallates are the hexacyanides. Hexacyanometalates are known for Ti(III), V(III), Cr(III), Cr(II), Mn(IV), Mn(III), Mn(II), Fe(II), Fe(III), Co(III), Ru(III), Ru(II), Os(III), and Os(II). Other more labile derivatives are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyano Complexes
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms. In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom. In Inorganic compound, inorganic cyanides, the cyanide group is present as the anion . Soluble Salt (chemistry), salts such as sodium cyanide (NaCN) and potassium cyanide (KCN) are highly toxic. Hydrocyanic acid, also known as hydrogen cyanide, or HCN, is a highly Volatility (chemistry), volatile liquid that is produced on a large scale industrially. It is obtained by acidification of cyanide salts. Organic compound, Organic cyanides are usually called nitriles. In nitriles, the group is linked by a covalent bond to carbon. For example, in acetonitrile (), the cyanide group is bonded to methyl (). Although nitriles generally do not release cyanide ions, the cyanohydrins do and are thus rather ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metal Nitrile Complexes
Transition metal nitrile complexes are coordination compounds containing nitrile ligands. Because nitriles are weakly basic, the nitrile ligands in these complexes are often labile. Scope of nitriles Typical nitrile ligands are acetonitrile, propionitrile, and benzonitrile. The structures of u(NH3)5(NCPh)sup>n+ have been determined for the 2+ and 3+ oxidation states. Upon oxidation the Ru-NH3 distances contract and the Ru-NCPh distances elongate, consistent with amines serving as pure-sigma donor ligands and nitriles functioning as pi-acceptors. Synthesis and reactions Acetonitrile, propionitrile and benzonitrile are also popular solvents. Because nitrile solvents have high dielectric constants, cationic complexes containing a nitrile ligand are often soluble in a solution of that nitrile. Some complexes can be prepared by dissolving an anhydrous metal salt in the nitrile. In other cases, a suspension of the metal is oxidized with a solution of NOBF4 in the nitrile: :Ni + 6 Me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
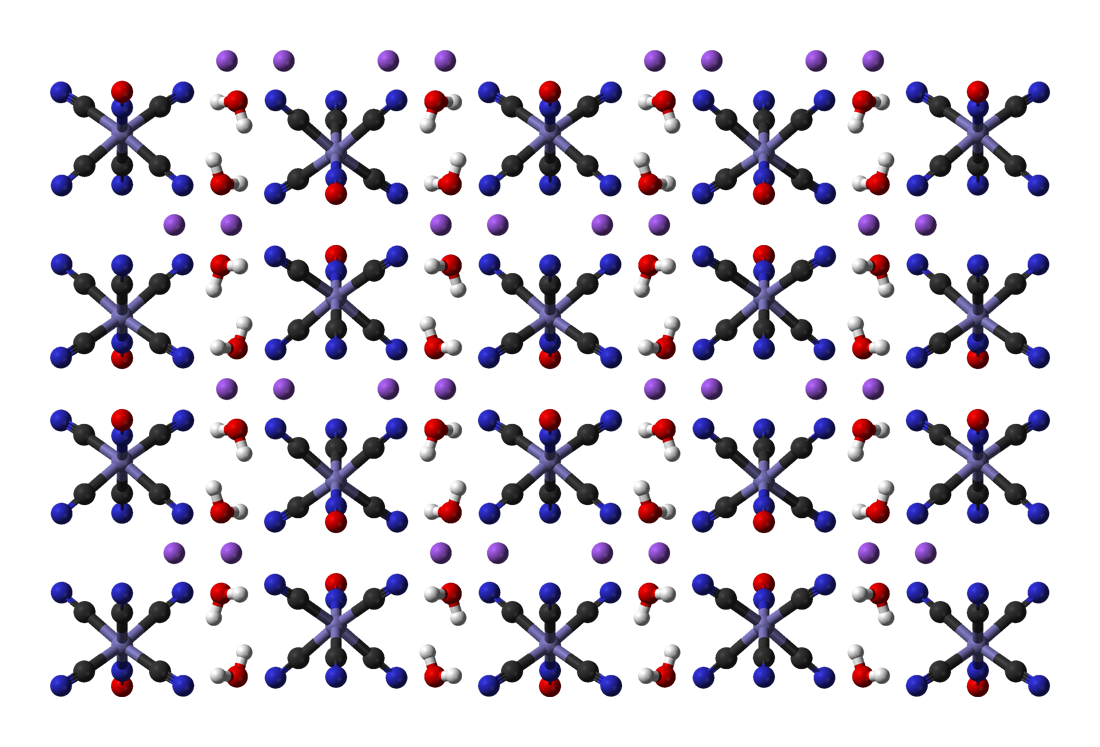
Prussian Blue
Prussian blue (also known as Berlin blue, Brandenburg blue or, in painting, Parisian or Paris blue) is a dark blue pigment produced by oxidation of ferrous ferrocyanide salts. It has the chemical formula Fe CN)">Cyanide.html" ;"title="e(Cyanide">CN) Turnbull's blue is chemically identical, but is made from different reagents, and its slightly different color stems from different impurities and particle sizes. Prussian blue was the first modern synthetic pigment. It is prepared as a very fine colloidal dispersion, because the compound is not soluble in water. It contains variable amounts of other ions and its appearance depends sensitively on the size of the colloidal particles. The pigment is used in paints, and it is the traditional "blue" in blueprints, and became prominent in 19th-century () Japanese woodblock prints. In medicine, orally administered Prussian blue is used as an antidote for certain kinds of heavy metal poisoning, e.g., by thallium(I) and radioactive is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyanide
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms. In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom. In inorganic cyanides, the cyanide group is present as the anion . Soluble salts such as sodium cyanide (NaCN) and potassium cyanide (KCN) are highly toxic. Hydrocyanic acid, also known as hydrogen cyanide, or HCN, is a highly volatile liquid that is produced on a large scale industrially. It is obtained by acidification of cyanide salts. Organic cyanides are usually called nitriles. In nitriles, the group is linked by a covalent bond to carbon. For example, in acetonitrile (), the cyanide group is bonded to methyl (). Although nitriles generally do not release cyanide ions, the cyanohydrins do and are thus rather toxic. Bonding The cyanide ion is isoelectronic with carbon monoxide a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Octacyanomolybdate(IV)
Potassium octacyanomolybdate(IV) is the inorganic salt with the formula K4 o(CN)8 A yellow light-sensitive solid, it is the potassium salt of the cyanometalate with the coordination number eight. The complex anion consists of a Mo(IV) center bound to eight cyanide ligands resulting in an overall charge of −4, which is balanced with four potassium cations. The salt is often prepared as its dihydrate K4 o(CN)8sup>.(H2O)2. Preparation The dihydrate K4 o(CN)8· 2 H2O can be prepared by the reduction of molybdate (MoO42-) with potassium borohydride (KBH4) in a solution with potassium cyanide and acetic acid. Yields of 70% are typical and the method is suited for scale-up. :4MoO42- + 32CN− + BH4− + 25H+ → 4 o(CN)8sup>4- + 13H2O + H3BO3 An alternative route starts from MoCl4(Et2O)2 avoiding the need for reductants. The yield of this route is typically around 70%. This synthesis is convenient for lower batch sizes than the earlier method but the MoCl4(Et2O)2 is typically less ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metals
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can use d orbitals as valence orbitals to form chemical bonds. The lanthanide and actinide elements (the f-block) are called inner transition metals and are sometimes considered to be transition metals as well. Since they are metals, they are lustrous and have good electrical and thermal conductivity. Most (with the exception of group 11 and group 12) are hard and strong, and have high melting and boiling temperatures. They form compounds in any of two or more different oxidation states and bind to a variety of ligands to form coordination complexes that are often coloured. They form many useful alloys and are often employed as catalysts in elemental form or in compounds such as coordination complexes and oxides. Most are strongly paramagn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solvatochromic
In chemistry, solvatochromism is the phenomenon observed when the colour due to a solute is different when that solute is dissolved in different solvents. The solvatochromic effect is the way the spectrum of a substance (the solute) varies when the substance is dissolved in a variety of solvents. In this context, the dielectric constant and hydrogen bonding capacity are the most important properties of the solvent. With various solvents there is a different effect on the electronic ground state and excited state of the solute, so that the size of energy gap between them changes as the solvent changes. This is reflected in the absorption or emission spectrum of the solute as differences in the position, intensity, and shape of the spectroscopic bands. When the spectroscopic band occurs in the visible part of the spectrum solvatochromism is observed as a change of colour. This is illustrated by Reichardt's dye, as shown in the image. Negative solvatochromism corresponds to a hypsoc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isocyanide
An isocyanide (also called isonitrile or carbylamine) is an organic compound with the functional group –. It is the isomer of the related nitrile (–C≡N), hence the prefix is ''isocyano''.IUPAC Goldboo''isocyanides''/ref> The organic fragment is connected to the isocyanide group through the nitrogen atom, not via the carbon. They are used as building blocks for the synthesis of other compounds. Properties Structure and bonding The C-N distance in isocyanides is 115.8 pm in methyl isocyanide. The C-N-C angles are near 180°. Akin to carbon monoxide, isocyanides are described by two resonance structures, one with a triple bond between the nitrogen and the carbon and one with a double bond between. The π lone pair of the nitrogen stabilizes the structure and is responsible of the linearity of isocyanides, although the reactivity of isocyanides reflects some carbene character, at least in a formal sense. Thus, both resonance structures are useful representations. They are sus ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ambidentate Ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chem ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetracyanonickelate
The cyanonickelates are a class of chemical compound containing anions consisting of nickel atoms, and cyanide groups. The most important of these are the tetracyanonickelates containing four cyanide groups per nickel. The tetracyanonickelates contain the [Ni(CN)4]2− anion. This can exist in solution or in solid salts. The ion has cyanide groups arranged in a square around the central nickel ion. The symmetry of the ion is Dihedral symmetry in three dimensions, D4''h''. The distance from the nickel atom to the carbon is 1.87 Å, and the carbon-nitrogen distance is 1.16 Å. Tetracyanonickelate(II) can be oxidised electrochemically in solution to yield tetracyanonickelate(III) [Ni(CN)4]−. [Ni(CN)4]− is unstable and Ni(III) oxidises the cyanide to cyanate OCN−. Tetracyanonickelate(III) can add two more cyanide groups to form hexacyanonickelate(III). In combination with alkyldiamines, and other metal ions, tetracyanonickelate ions can form cage structure that can ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases. ''Nucleophilic'' describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. History The terms ''nucleophile'' and ''electrophile'' were introduced by Christopher Kelk Ingold in 1933, replacing the terms ''anionoid'' and ''cationoid' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Nitroprusside
Sodium nitroprusside (SNP), sold under the brand name Nitropress among others, is a medication used to lower blood pressure. This may be done if the blood pressure is very high and resulting in symptoms, in certain types of heart failure, and during surgery to decrease bleeding. It is used by continuous injection into a vein. Onset is nearly immediate and effects last for up to ten minutes. It is available as a generic medication. Side effects and mechanism Common side effects include low blood pressure and cyanide toxicity. Other serious side effects include methemoglobinemia. It is not generally recommended during pregnancy due to concerns of side effects. High doses are not recommended for more than ten minutes. It works by increasing nitric oxide levels in the blood, which increases cGMP levels in cells, and causes dilation of blood vessels. History, society and culture Sodium nitroprusside was discovered as early as 1850 and found to be useful in medicine in 1928. It i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

copper(I)_hexafluorophosphate.png)




4-3D-balls.png)
-3D-balls.png)