|
Corey–Seebach Reaction
The Corey–Seebach reaction, or Seebach Umpolung is a name reaction of organic chemistry that allows for acylation by converting aldehydes into lithiated dithiane#1,3-Dithianes, 1,3-dithianes. The lithiated 1,3-dithianes serves as an acyl anion equivalent, undergoing alkylation with electrophiles. The reaction is of pedagogical value, but it is cumbersome, so it is not widely used. Implementation The aldehyde is first converted into a dithiane, usually with 1,3-propanedithiol. The resulting 1,3-dithiane is then lithiated with the use of N-butyllithium, butyllithium. The 2-lithio-1,3-dithiane reacts with electrophiles to give a 2-alkyl-1,3-dithiane. Finally, the 2-alkyl-1,3-dithiane can be converted to a carbonyl by hydrolysis, usually with the use of mercury(II) oxide. Alternatively the 2-alkyl-1,3-dithiane can be reduced to an alkane. : Scope The lithiated 1,3-dithiane can react with alkyl halides, epoxides, ketones, acyl halides, and iminium salts, which after hydrolysis o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elias James Corey
Elias James Corey (born July 12, 1928) is an American organic chemistry, organic chemist. In 1990, he won the Nobel Prize in Chemistry "for his development of the theory and methodology of organic synthesis", specifically retrosynthetic analysis. Regarded by many as one of the greatest living chemists, he has developed numerous synthetic reagents, methodologies and total syntheses and has advanced the science of organic synthesis considerably. Biography E.J. Corey (the surname was anglicized from Levantine Arabic ''Khoury'', meaning ''priest'') was born to Lebanese Greek Orthodox Christians, Lebanese Greek Orthodox Christian immigrants Fatima (née Hasham) and Elias Corey in Methuen, Massachusetts, north of Boston. His mother changed his name from William to "Elias" to honor his father, who died eighteen months after Corey's birth. His widowed mother, brother, two sisters, aunt and uncle all lived together in a spacious house, struggling through the Great Depression. As a young b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
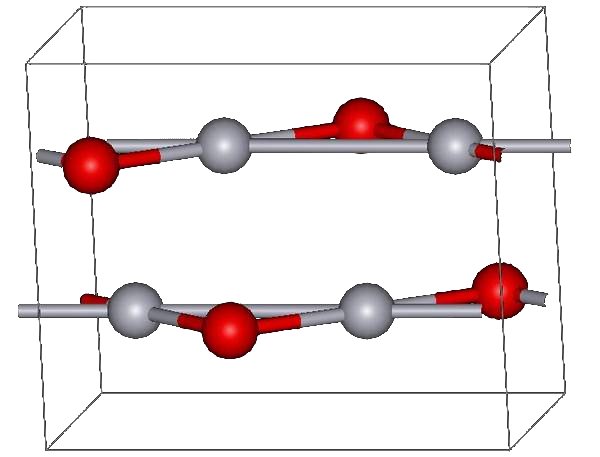
Dithiane Chemistry
A dithiane is a heterocyclic compound composed of a cyclohexane core structure wherein two methylene bridges (-- units) are replaced by sulfur centres. The three isomeric parent heterocycles are 1,2-dithiane, 1,3-dithiane and 1,4-dithiane. 1,3-Dithianes 1,3-Dithianes are protecting group of some carbonyl-containing compounds due to their inertness to many conditions. They form by treatment of the carbonyl compound with 1,3-Propanedithiol, 1,3-propanedithiol under conditions that remove water from the system. The protecting group can be removed with mercuric reagents, a process that exploits the high affinity of Hg(II) for thiolates. 1,3-Dithianes are also employed in umpolung#Carbonyl umpolung, umpolung reactions, such as the Corey–Seebach reaction:T. W. Green, P. G. M. Wuts, "Protective Groups in Organic Synthesis" Wiley-Interscience, New York, 1999. . : Typically, in organic synthesis, ketones and aldehydes are protected as their dioxolanes instead of dithianes. Refer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amides
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, such as in the amino acids asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid () with the hydroxyl group () replaced by an amine group (); or, equivalently, an acyl (alkanoyl) group () joined to an amine group. Common examples of amides are acetamide (), benzamide (), and dimethylformamide (). Amides are qualified as primary, secondary, and tertiary according to whether the amine subgroup has the form , , or , where R and R' are groups other than hydrogen. The core of amides is called the amide group (specifically, carboxamide group). Amides are pervasive in nature and technology. Proteins and important plastics ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry. Discovered independently by the Russian chemist Alexander Borodin in 1869 and by the French chemist Charles-Adolphe Wurtz in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as ''aldols'', from the ''ald''ehyde + alcoh''ol'', a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic. For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritol and the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt). The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the Alpha carbon, α- and β-carbon of the aldol adduct, mar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iminium
In organic chemistry, an iminium cation is a polyatomic ion with the general structure . They are common in synthetic chemistry and biology. Structure Iminium cations adopt alkene-like geometries. The central C=N unit is nearly coplanar with all four substituents. The C=N distances, which are near 129 picometers in length, are shorter than C-N single bonds. Cis/trans isomers are observed. Formation Iminium cations are obtained by protonation and alkylation of imines: :RN=CR'_2 + H+ -> NH=CR'_2 :RN=CR'_2 + R''+ -> R''N=CR'_2 They also are generated by the condensation of secondary amines with ketones or aldehydes: :O=CR'_2 + R2NH + H+ 2N=CR'_2 + H2O This rapid, reversible reaction is one step in "iminium catalysis". More exotic routes to iminium cations are known, e.g. from ring-opening reactions of pyridine. Occurrence Iminium derivatives are common in biology. Pyridoxal phosphate reacts with amino acids to give iminium derivatives. Many iminium salts are en ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acyl Halides
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid by replacing a hydroxyl group () with a halide group (, where X is a halogen). If the acid is a carboxylic acid (), the compound contains a functional group, which consists of a carbonyl group () singly bonded to a halogen atom. The general formula for such an acyl halide can be written RCOX, where R may be, for example, an alkyl group, CO is the carbonyl group, and X represents the halide, such as chloride. Acyl chlorides are the most commonly encountered acyl halides, but acetyl iodide is the one produced (transiently) on the largest scale. Billions of kilograms are generated annually in the production of acetic acid. Preparation Aliphatic acyl halides On an industrial scale, the reaction of acetic anhydride with hydrogen chloride produces a mixture of acetyl chloride and acetic acid: :(CH3CO)2O + HCl -> CH3COCl + CH3CO2H Common syntheses of acyl chlorid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketones
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' is methyl), with the formula . Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered ret ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxides
In organic chemistry, an epoxide is a cyclic ether () with a three-atom ring. This ring approximates an equilateral triangle, which makes it strained, and hence highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is called an ''epoxy'', but such materials do not contain epoxide groups (or contain only a few residual epoxy grou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyl Halides
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes that contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mercury(II) Oxide
Mercury(II) oxide, also called mercuric oxide or simply mercury oxide, is the inorganic compound with the formula Hg O. It has a red or orange color. Mercury(II) oxide is a solid at room temperature and pressure. The mineral form montroydite is very rarely found. History An experiment for the preparation of mercuric oxide was first described by 11th century Arab-Spanish alchemist, Maslama al-Majriti, in ''Rutbat al-hakim.'' In 1774, Joseph Priestley discovered that oxygen was released by heating mercuric oxide, although he did not identify the gas as oxygen (rather, Priestley called it " dephlogisticated air," as that was the paradigm that he was working under at the time). Synthesis The red form of HgO can be made by heating Hg in oxygen at roughly 350 °C, or by pyrolysis of Hg(NO3)2. The yellow form can be obtained by precipitation of aqueous Hg2+ with alkali. The difference in color is due to particle size; both forms have the same structure consisting of near lin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dieter Seebach
Dieter Seebach is a German chemist known for his synthesis of biopolymers and dendrimers, and for his contributions to stereochemistry. He was born on 31 October 1937 in Karlsruhe. He studied chemistry at the University of Karlsruhe (TH) under the supervision of Rudolf Criegee and at Harvard University with Elias Corey finishing in 1969. After his habilitation he became professor for organic chemistry at the University of Giessen. After six years he was appointed professor at the ETH Zurich where he worked until he retired in 2003. Work Seebach worked on dendrimer chemistry and on the synthesis of beta-peptides. The development of the umpolung, a polarity inversion of the carbonyl group, with 1,3-propanedithiol together with Corey had a big influence on organic synthesis, and subsequently the Corey-Seebach reaction was named after them. The Fráter–Seebach alkylation, a diastereoselective reaction of beta-hydroxy esters, is named after him. Awards *2000 Marcel Benoist Pri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-butyllithium
''n''-Butyllithium C4H9Li (abbreviated ''n''-BuLi) is an organolithium reagent. It is widely used as a polymerization initiator in the production of elastomers such as polybutadiene or styrene-butadiene-styrene (SBS). Also, it is broadly employed as a strong base (superbase) in the synthesis of organic compounds as in the pharmaceutical industry. Butyllithium is commercially available as solutions (15%, 25%, 1.5 M, 2 M, 2.5 M, 10 M, etc.) in alkanes such as pentane, hexanes, and heptanes. Solutions in diethyl ether and THF can be prepared, but are not stable enough for storage. Annual worldwide production and consumption of butyllithium and other organolithium compounds is estimated at 2000 to 3000 tonnes. Although butyllithium is colorless, ''n''-butyllithium is usually encountered as a pale yellow solution in alkanes. Such solutions are stable indefinitely if properly stored,. but in practice, they degrade upon aging. Fine white precipitate (l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |