|
Coble Creep
Coble creep, a form of diffusion creep, is a mechanism for deformation of crystalline solids. Contrasted with other diffusional creep mechanisms, Coble creep is similar to Nabarro–Herring creep in that it is dominant at lower stress levels and higher temperatures than creep mechanisms utilizing dislocation glide. Coble creep occurs through the diffusion of atoms in a material along grain boundaries. This mechanism is observed in polycrystals or along the surface in a single crystal, which produces a net flow of material and a sliding of the grain boundaries. Robert L. Coble first reported his theory of how materials creep across grain boundaries and at high temperatures in alumina. Here he famously noticed a different creep mechanism that was more dependent on the size of the grain. The strain rate in a material experiencing Coble creep is given by : \frac \equiv \dot_C = A_C\frac\fracD_0e^ = A_C\frac\fracD_, where : A_c is a geometric prefactor : \sigma is the applied stre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diffusion Creep
Diffusion creep refers to the deformation of crystalline solids by the diffusion of vacancies through their crystal lattice. Diffusion creep results in plastic deformation rather than brittle failure of the material. Diffusion creep is more sensitive to temperature than other deformation mechanisms. It usually takes place at high homologous temperatures (i.e. within about a tenth of its absolute melting temperature). Diffusion creep is caused by the migration of crystalline defects through the lattice of a crystal such that when a crystal is subjected to a greater degree of compression in one direction relative to another, defects migrate to the crystal faces along the direction of compression, causing a net mass transfer that shortens the crystal in the direction of maximum compression. The migration of defects is in part due to vacancies, whose migration is equal to a net mass transport in the opposite direction. Principle Crystalline materials are never perfect on a microsca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
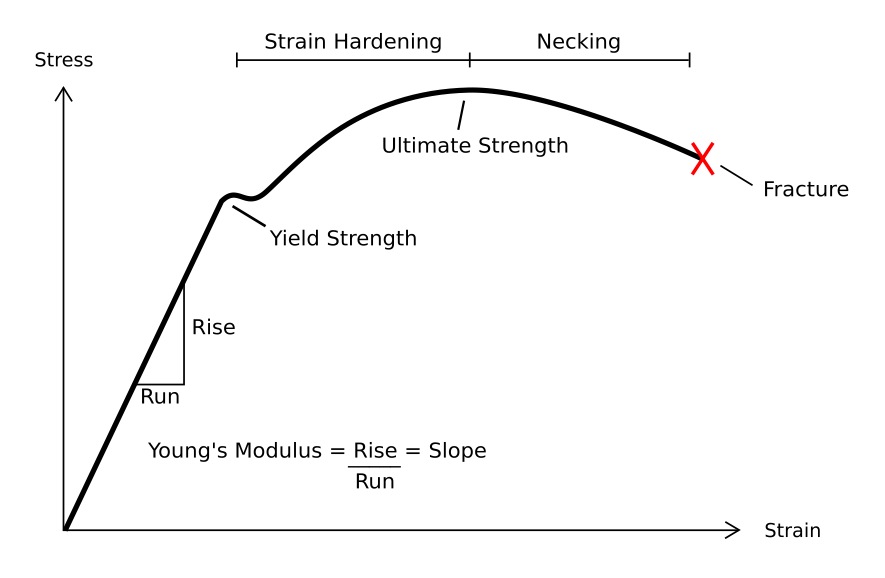
Deformation (engineering)
In engineering, deformation refers to the change in size or shape of an object. ''Displacements'' are the ''absolute'' change in position of a point on the object. Deflection is the relative change in external displacements on an object. Strain is the ''relative'' internal change in shape of an infinitesimally small cube of material and can be expressed as a non-dimensional change in length or angle of distortion of the cube. Strains are related to the forces acting on the cube, which are known as stress, by a stress-strain curve. The relationship between stress and strain is generally linear and reversible up until the yield point and the deformation is elastic. The linear relationship for a material is known as Young's modulus. Above the yield point, some degree of permanent distortion remains after unloading and is termed plastic deformation. The determination of the stress and strain throughout a solid object is given by the field of strength of materials and for a structu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystalline
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. The scientific study of crystals and crystal formation is known as crystallography. The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The word ''crystal'' derives from the Ancient Greek word (), meaning both " ice" and " rock crystal", from (), "icy cold, frost". Examples of large crystals include snowflakes, diamonds, and table salt. Most inorganic solids are not crystals but polycrystals, i.e. many microscopic crystals fused together into a single solid. Polycrystals include most metals, rocks, ceramics, and ice. A third catego ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solid
Solid is one of the four fundamental states of matter (the others being liquid, gas, and plasma). The molecules in a solid are closely packed together and contain the least amount of kinetic energy. A solid is characterized by structural rigidity and resistance to a force applied to the surface. Unlike a liquid, a solid object does not flow to take on the shape of its container, nor does it expand to fill the entire available volume like a gas. The atoms in a solid are bound to each other, either in a regular geometric lattice (crystalline solids, which include metals and ordinary ice), or irregularly (an amorphous solid such as common window glass). Solids cannot be compressed with little pressure whereas gases can be compressed with little pressure because the molecules in a gas are loosely packed. The branch of physics that deals with solids is called solid-state physics, and is the main branch of condensed matter physics (which also includes liquids). Material ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nabarro–Herring Creep
Nabarro–Herring creep is a mode of deformation of crystalline materials (and amorphous materials) that occurs at low stresses and held at elevated temperatures in fine-grained materials. In Nabarro–Herring creep (NH creep), atoms diffuse through the crystals, and the creep rate varies inversely with the square of the grain size so fine-grained materials creep faster than coarser-grained ones. NH creep is solely controlled by diffusional mass transport. This type of creep results from the diffusion of vacancies from regions of high chemical potential at grain boundaries subjected to normal tensile stresses to regions of lower chemical potential where the average tensile stresses across the grain boundaries are zero. Self-diffusion within the grains of a polycrystalline solid can cause the solid to yield to an applied shearing stress, the yielding being caused by a diffusional flow of matter within each crystal grain away from boundaries where there is a normal pressure and towa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grain Boundaries
In materials science, a grain boundary is the interface between two grains, or crystallites, in a polycrystalline material. Grain boundaries are two-dimensional defects in the crystal structure, and tend to decrease the electrical and thermal conductivity of the material. Most grain boundaries are preferred sites for the onset of corrosion and for the precipitation of new phases from the solid. They are also important to many of the mechanisms of creep. On the other hand, grain boundaries disrupt the motion of dislocations through a material, so reducing crystallite size is a common way to improve mechanical strength, as described by the Hall–Petch relationship. High and low angle boundaries It is convenient to categorize grain boundaries according to the extent of misorientation between the two grains. ''Low-angle grain boundaries'' (''LAGB'') or ''subgrain boundaries'' are those with a misorientation less than about 15 degrees. Generally speaking they are composed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Robert L
The name Robert is an ancient Germanic given name, from Proto-Germanic "fame" and "bright" (''Hrōþiberhtaz''). Compare Old Dutch ''Robrecht'' and Old High German ''Hrodebert'' (a compound of '' Hruod'' ( non, Hróðr) "fame, glory, honour, praise, renown" and ''berht'' "bright, light, shining"). It is the second most frequently used given name of ancient Germanic origin. It is also in use as a surname. Another commonly used form of the name is Rupert. After becoming widely used in Continental Europe it entered England in its Old French form ''Robert'', where an Old English cognate form (''Hrēodbēorht'', ''Hrodberht'', ''Hrēodbēorð'', ''Hrœdbœrð'', ''Hrœdberð'', ''Hrōðberχtŕ'') had existed before the Norman Conquest. The feminine version is Roberta. The Italian, Portuguese, and Spanish form is Roberto. Robert is also a common name in many Germanic languages, including English, German, Dutch, Norwegian, Swedish, Scots, Danish, and Icelandic. It can be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Creep (deformation)
In materials science, creep (sometimes called cold flow) is the tendency of a solid material to move slowly or deform permanently under the influence of persistent mechanical stresses. It can occur as a result of long-term exposure to high levels of stress that are still below the yield strength of the material. Creep is more severe in materials that are subjected to heat for long periods and generally increases as they near their melting point. The rate of deformation is a function of the material's properties, exposure time, exposure temperature and the applied structural load. Depending on the magnitude of the applied stress and its duration, the deformation may become so large that a component can no longer perform its function – for example creep of a turbine blade could cause the blade to contact the casing, resulting in the failure of the blade. Creep is usually of concern to engineers and metallurgists when evaluating components that operate under high stresses or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boltzmann's Constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin and the gas constant, and in Planck's law of black-body radiation and Boltzmann's entropy formula, and is used in calculating thermal noise in resistors. The Boltzmann constant has dimensions of energy divided by temperature, the same as entropy. It is named after the Austrian scientist Ludwig Boltzmann. As part of the 2019 redefinition of SI base units, the Boltzmann constant is one of the seven " defining constants" that have been given exact definitions. They are used in various combinations to define the seven SI base units. The Boltzmann constant is defined to be exactly . Roles of the Boltzmann constant Macroscopically, the ideal gas law states that, for an ideal gas, the product of pressure and volume is proportional to the product of amount of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kelvin
The kelvin, symbol K, is the primary unit of temperature in the International System of Units (SI), used alongside its prefixed forms and the degree Celsius. It is named after the Belfast-born and University of Glasgow-based engineer and physicist William Thomson, 1st Baron Kelvin (1824–1907). The Kelvin scale is an absolute thermodynamic temperature scale, meaning it uses absolute zero as its null (zero) point. Historically, the Kelvin scale was developed by shifting the starting point of the much-older Celsius scale down from the melting point of water to absolute zero, and its increments still closely approximate the historic definition of a degree Celsius, but since 2019 the scale has been defined by fixing the Boltzmann constant to be exactly . Hence, one kelvin is equal to a change in the thermodynamic temperature that results in a change of thermal energy by . The temperature in degree Celsius is now defined as the temperature in kelvins minus 273.15, mea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vacancy Defect
In crystallography, a vacancy is a type of point defect in a crystal where an atom is missing from one of the lattice sites.Ehrhart, P. (1991) "Properties and interactions of atomic defects in metals and alloys", chapter 2, p. 88 in ''Landolt-Börnstein, New Series III'', Vol. 25, Springer, Berlin Crystals inherently possess imperfections, sometimes referred to as crystallographic defects. Vacancies occur naturally in all crystalline materials. At any given temperature, up to the melting point of the material, there is an equilibrium concentration (ratio of vacant lattice sites to those containing atoms). At the melting point of some metals the ratio can be approximately 1:1000. This temperature dependence can be modelled by :N_ = N \exp\left(\frac\right) where is the vacancy concentration, is the energy required for vacancy formation, is the Boltzmann constant, is the absolute temperature, and is the concentration of atomic sites i.e. : N = \frac where is mass, the Av ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |