|
Carbamoyl Phosphate Synthetase III
Carbamoyl phosphate synthetase III (CPS III) is one of the three isoforms of the carbamoyl phosphate synthetase, an enzyme that catalyzes the active production of carbamoyl phosphate in many organisms. CPS III (EC 6.3.5.5.) is a ligase (3.) that forms carbon-nitrogen bonds (6.3.) with glutamine as amido-N-donor (6.3.5.) (see BRENDA). Context Many aquatic organisms, including most of the fish species, are ammoniotelic, which means they produce ammonia as metabolic waste, that they generally excrete by diffusion through their gills. Similar to terrestrial vertebrates, some fish species also significantly include urea as metabolic waste. This phenomenon concerns larvae stages since they do not have gills to excrete ammonia from, but also the adult stage in some species. Based on the proportion of metabolic waste represented by urea, these species are partial or fully ureotelic. Ureotelic species produce urea via the ornithine-urea cycle (OUC) in which CPS plays an important role. Ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbamoyl Phosphate Synthetase
Carbamoyl phosphate synthetase catalyzes the ATP-dependent synthesis of carbamoyl phosphate from glutamine () or ammonia () and bicarbonate. This enzyme catalyzes the reaction of ATP and bicarbonate to produce carboxy phosphate and ADP. Carboxy phosphate reacts with ammonia to give carbamic acid. In turn, carbamic acid reacts with a second ATP to give carbamoyl phosphate plus ADP. It represents the first committed step in pyrimidine and arginine biosynthesis in prokaryotes and eukaryotes, and in the urea cycle in most terrestrial vertebrates. Most prokaryotes carry one form of CPSase that participates in both arginine and pyrimidine biosynthesis, however certain bacteria can have separate forms. There are three different forms that serve very different functions: * Carbamoyl phosphate synthetase I (mitochondria, urea cycle) * Carbamoyl phosphate synthetase II (cytosol, pyrimidine metabolism). * Carbamoyl phosphate synthetase III (found in fish). Mechanism Carbamoyl phos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbamoyl Phosphate Synthase II
Carbamoyl phosphate synthetase (glutamine-hydrolysing) () is an enzyme that catalyzes the reactions that produce carbamoyl phosphate in the cytosol (as opposed to type I, which functions in the mitochondria). Its systemic name is ''hydrogen-carbonate:L-glutamine amido-ligase (ADP-forming, carbamate-phosphorylating)''. In pyrimidine biosynthesis, it serves as the rate-limiting enzyme and catalyzes the following reaction: : 2 ATP + L-glutamine + HCO3− + H2O \rightleftharpoons 2 ADP + phosphate + L-glutamate + carbamoyl phosphate (overall reaction) : (1a) L-glutamine + H2O \rightleftharpoons L-glutamate + NH3 : (1b) 2 ATP + HCO3− + NH3 \rightleftharpoons 2 ADP + phosphate + carbamoyl phosphate It is activated by ATP and PRPP and it is inhibited by UMP (Uridine monophosphate, the end product of the pyrimidine synthesis pathway). Neither CPSI nor CPSII require biotin as a coenzyme, as seen with most carboxylation reactions. It is one of the three enzyme functions coded by the CA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Effector (biology)
In biochemistry, an effector molecule is usually a small molecule that selectively binds to a protein and regulates its biological activity. In this manner, effector molecules act as ligands that can increase or decrease enzyme activity, gene expression, or cell signaling. Effector molecules can also directly regulate the activity of some mRNA molecules (riboswitches). Other examples of effector functions in biochemistry include hormone signaling and immune response. In some cases, specific proteins serve the same role as effector molecules (note: small molecules refers to organic compounds similar in size to amino acids or RNA strands. Most effector molecules are therefor much smaller than individual proteins, which consist of many amino acids). One example of this is in cellular signal transduction cascades. The term ''effector'' is used in other fields of biology. For instance, the effector end of a neuron is the terminus where an axon makes contact with the muscle or o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
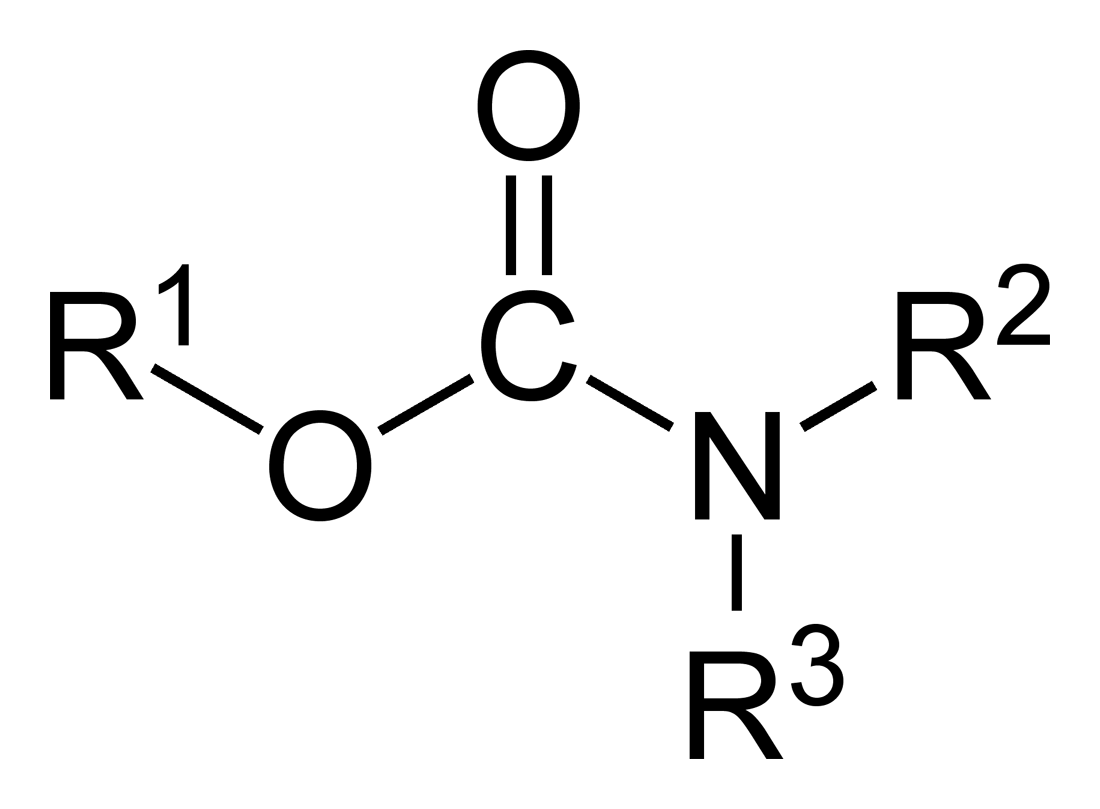
Carbamate
In organic chemistry, a carbamate is a category of organic compounds with the general formula and structure , which are formally derived from carbamic acid (). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion (e.g. ammonium carbamate). Polymers whose units are joined by carbamate groups are an important family of plastics, the polyurethanes. Properties While carbamic acids are unstable, many carbamate esters or ionic) are stable and well known. Equilibrium with carbonate and bicarbonate In water solutions, the carbamate anion slowly equilibrates with the ammonium cation and the carbonate or bicarbonate anions: : : Calcium carbamate is soluble in water, whereas calcium carbonate is not. Adding a calcium salt to an ammonium carbamate/carbonate solution will precipitate some calcium carbonate immediatel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
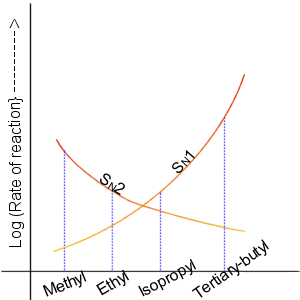
Nucleophilic Substitution
In chemistry, a nucleophilic substitution is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). The molecule that contains the electrophile and the leaving functional group is called the substrate. The most general form of the reaction may be given as the following: :\text\mathbf + \ce + \text\mathbf The electron pair (:) from the nucleophile (Nuc) attacks the substrate () and bonds with it. Simultaneously, the leaving group (LG) departs with an electron pair. The principal product in this case is . The nucleophile may be electrically neutral or negatively charged, whereas the substrate is typically neutral or positively charged. An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is hydroxyl () and the leaving group is bromide (). :R-Br + OH- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutamine
Glutamine (symbol Gln or Q) is an α-amino acid that is used in the biosynthesis of proteins. Its side chain is similar to that of glutamic acid, except the carboxylic acid group is replaced by an amide. It is classified as a charge-neutral, polar amino acid. It is non-essential and conditionally essential in humans, meaning the body can usually synthesize sufficient amounts of it, but in some instances of stress, the body's demand for glutamine increases, and glutamine must be obtained from the diet. It is encoded by the codons CAA and CAG. In human blood, glutamine is the most abundant free amino acid. The dietary sources of glutamine include especially the protein-rich foods like beef, chicken, fish, dairy products, eggs, vegetables like beans, beets, cabbage, spinach, carrots, parsley, vegetable juices and also in wheat, papaya, Brussels sprouts, celery, kale and fermented foods like miso. Functions Glutamine plays a role in a variety of biochemical functions: * Pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Triphosphate
Adenosine triphosphate (ATP) is an organic compound that provides energy to drive many processes in living cells, such as muscle contraction, nerve impulse propagation, condensate dissolution, and chemical synthesis. Found in all known forms of life, ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer. When consumed in metabolic processes, it converts either to adenosine diphosphate (ADP) or to adenosine monophosphate (AMP). Other processes regenerate ATP. The human body recycles its own body weight equivalent in ATP each day. It is also a precursor to DNA and RNA, and is used as a coenzyme. From the perspective of biochemistry, ATP is classified as a nucleoside triphosphate, which indicates that it consists of three components: a nitrogenous base (adenine), the sugar ribose, and the Polyphosphate, triphosphate. Structure ATP consists of an adenine attached by the 9-nitrogen atom to the 1′ carbon atom of a sugar (ribose), which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acid Synthesis
Amino acid synthesis is the set of biochemical processes (metabolic pathways) by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can synthesize 11 of the 20 standard amino acids. These 11 are called the non-essential amino acids). α-Ketoglutarates: glutamate, glutamine, proline, arginine Most amino acids are synthesized from α- ketoacids, and later transaminated from another amino acid, usually glutamate. The enzyme involved in this reaction is an aminotransferase. : α-ketoacid + glutamate ⇄ amino acid + α-ketoglutarate Glutamate itself is formed by amination of α-ketoglutarate: : α-ketoglutarate + ⇄ glutamate The α-ketoglutarate family of amino acid synthesis (synthesis of glutamate, glutamine, proline and arginine) begins with α-ketoglutarate, an intermediate in the Citric Acid Cycle. The concentr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
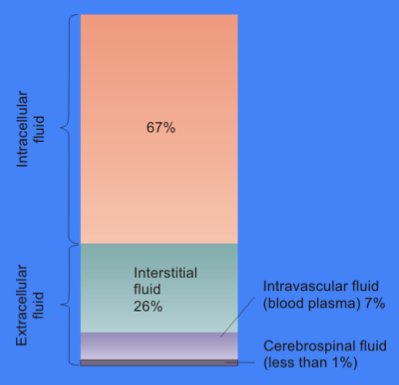
Cytosol
The cytosol, also known as cytoplasmic matrix or groundplasm, is one of the liquids found inside cells (intracellular fluid (ICF)). It is separated into compartments by membranes. For example, the mitochondrial matrix separates the mitochondrion into many compartments. In the eukaryotic cell, the cytosol is surrounded by the cell membrane and is part of the cytoplasm, which also comprises the mitochondria, plastids, and other organelles (but not their internal fluids and structures); the cell nucleus is separate. The cytosol is thus a liquid matrix around the organelles. In prokaryotes, most of the chemical reactions of metabolism take place in the cytosol, while a few take place in membranes or in the periplasmic space. In eukaryotes, while many metabolic pathways still occur in the cytosol, others take place within organelles. The cytosol is a complex mixture of substances dissolved in water. Although water forms the large majority of the cytosol, its structure and prope ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
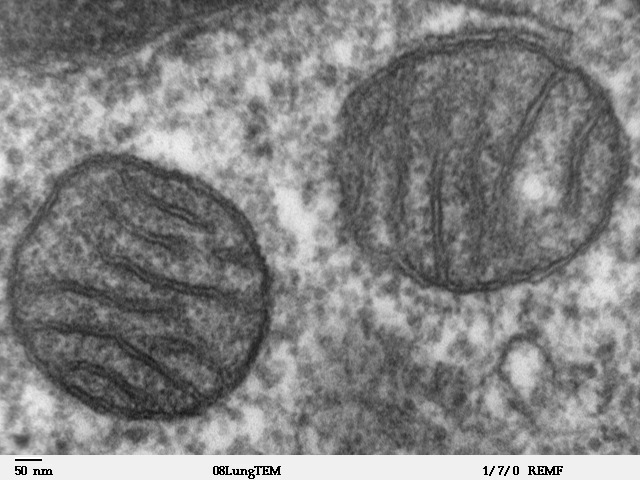
Mitochondrion
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitochondria, and one multicellular organism, '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |