|
Chlorobis(ethylene)rhodium Dimer
Chlorobis(ethylene)rhodium dimer is an organorhodium compound with the formula Rh2Cl2(C2H4)4. It is a red-orange solid that is soluble in nonpolar organic solvents. The molecule consists of two bridging chloride ligands and four ethylene ligands. The ethylene ligands are labile and readily displaced even by other alkenes. A variety of homogeneous catalysts have been prepared from this complex. Preparation and reactions The complex is prepared by treating an aqueous methanolic solution of hydrated rhodium trichloride with ethylene at room temperature. Rh(III) is reduced with oxidation of ethylene to acetaldehyde: :2 RhCl3(H2O)3 + 6 C2H4 → Rh2Cl2(C2H4)4 + 2 CH3CHO + 4 HCl + 4 H2O Reflecting the lability of its ligands, the complex does not tolerate recrystallization.Richard Cramer "Di-μ-chlorotetrakis(ethylene)dirhodium(I), 2,4-pentanedionatobis(ethylene)rhodium(I), and di-μ-chlorotetracarbonyldirhodium(I)" Inorganic Syntheses 1974, vol. 15, pp. 14-18.. Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organorhodium Compound
Organorhodium chemistry is the chemistry of organometallic compounds containing a rhodium-carbon chemical bond, and the study of rhodium and rhodium compounds as catalysts in organic reactions. Stable organorhodium compounds and transient organorhodium intermediates are used as catalyst such as in olefin hydroformylation, olefin hydrogenation, olefin isomerization and the Monsanto process Classification based on principal oxidation states Organometallic rhodium compounds share many characteristics with those of iridium, but less so with cobalt. Rhodium can exist in oxidation states of -III to +V, but rhodium(I) and rhodium(III) are the more common. Rhodium(I) compounds (d8 configuration) usually occur with square planar or trigonal bipyramidal geometries, while rhodium (III) compounds (d6 configuration) typically have an octahedral geometry. Rhodium(0) Rhodium(0) complexes are binary carbonyls, the principal examples being tetrarhodium dodecacarbonyl, Rh4(CO)12, and hexadecacarb ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
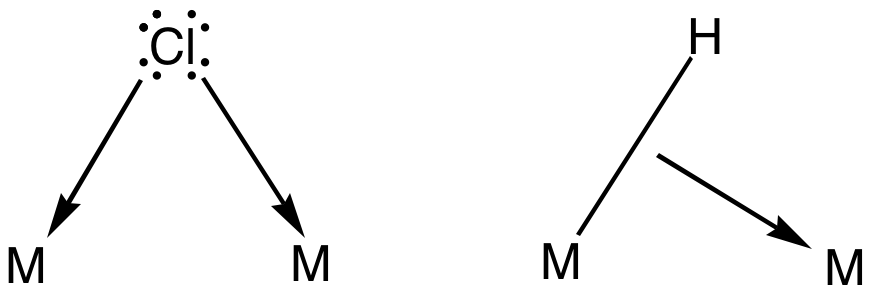
Ethylene Dimerization
In organic chemistry, hydrovinylation is the formal insertion of an alkene into the C-H bond of ethylene (). The more general reaction, hydroalkenylation, is the formal insertion of an alkene into the C-H bond of any terminal alkene. The reaction is catalyzed by metal complexes. A representative reaction is the conversion of styrene and ethylene to 3-phenybutene: :\ce + \longrightarrow \ce Ethylene dimerization The dimerization of ethylene gives 1-butene is another example of a hydrovinylation. In the Dimersol and Alphabutol Processes, alkenes are dimerized for the production of gasoline and for comonomers such as 1-butene. These processes operate at several refineries across the world at the scales of about 400,000 tons/year (2006 report). 1-Butene is amenable to isomerization to 2-butenes, which is used in Olefin conversion technology to give propylene. Hydroarylation Hydroarylation is again a special case of hydrovinylation. Hydroarylation has been demonstrated for al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimers (chemistry)
Dimer may refer to: * Dimer (chemistry), a chemical structure formed from two similar sub-units ** Protein dimer, a protein quaternary structure ** d-dimer * Dimer model, an item in statistical mechanics, based on ''domino tiling'' * Julius Dimer Julius Dimer (1 August 1871 – 20 October 1945) was a German chess master. At the beginning of his career, he played in several mini tournaments (''Quadrangular'') in Germany; at Altona 1897, Elmshorn 1898, Munich 1900, Kiel 1901, Hamburg 1903, ... (1871–1945), German chess master See also * Dimery (botany), having two parts in a distinct whorl of a plant structure * Di (other), a prefix * Dymer (other) * -mer, a suffix * Oligomer * Peierls transition, sometimes called dimerization {{Disambiguation, surname ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene Complexes
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond. Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, and Biological Chemistry'. 1232 pages. Two general types of monoalkenes are distinguished: terminal and internal. Also called α-olefins, terminal alkenes are more useful. However, the International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homogeneous Catalysis
In chemistry, homogeneous catalysis is catalysis by a soluble catalyst in a solution. Homogeneous catalysis refers to reactions where the catalyst is in the same phase as the reactants, principally in solution. In contrast, heterogeneous catalysis describes processes where the catalysts and substrate are in distinct phases, typically solid-gas, respectively. The term is used almost exclusively to describe solutions and implies catalysis by organometallic compounds. Homogeneous catalysis is an established technology that continues to evolve. An illustrative major application is the production of acetic acid. Enzymes are examples of homogeneous catalysts. Examples Acid catalysis The proton is a pervasive homogeneous catalyst because water is the most common solvent. Water forms protons by the process of self-ionization of water. In an illustrative case, acids accelerate (catalyze) the hydrolysis of esters: :CH3CO2CH3 + H2O CH3CO2H + CH3OH At neutral pH, aqueous solutions of most e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhodium Carbonyl Chloride
Rhodium carbonyl chloride is an organorhodium compound with the formula Rh2Cl2(CO)4. It is a red-brown volatile solid that is soluble in nonpolar organic solvents. It is a precursor to other rhodium carbonyl complexes, some of which are useful in homogeneous catalysis. Structure The molecule consists of two planar Rh(I) centers linked by two bridging chloride ligands and four CO ligands. X-ray crystallography shows that the two Rh(I) centers are square planar with the dihedral angle of 53° between the two RhCl2 planes. The metals are nonbonding. Synthesis and reactions First prepared by Walter Hieber, it is typically prepared by treating hydrated rhodium trichloride with flowing carbon monoxide, according to this idealized redox equation: :2 RhCl3(H2O)3 + 6 CO → Rh2Cl2(CO)4 + 2 COCl2 + 6 H2O.McCleverty, J. A.; Wilkinson, G. "Dichlorotetracarbonyldirhodium (rhodium carbonyl chloride)" Inorganic Syntheses 1966, volume 8, pp. 211-14. The complex reacts with triph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1-butene
1-Butene (or 1-Butylene) is the organic compound with the formula CH3CH2CH=CH2. It is a colorless gas that is easily condensed to give a colorless liquid. It is classified as a linear alpha-olefin. It is one of the isomers of butene (butylene). It is a precursor to diverse products. Reactions Polymerization of 1-butene give polybutene, which is used to make piping for domestic plumbing. Its main application is as a comonomer in the production of certain kinds of polyethylene, such as linear low-density polyethylene (LLDPE). It has also been used as a precursor to polypropylene resins, butylene oxide, and butanone. Manufacturing 1-Butene is produced by separation from crude C4 refinery streams and by ethylene dimerization. The former affords a mixture of 1-and 2-butenes, while the latter affords only the terminal alkene. It is distilled to give a very high purity product. An estimated 12 billion kilograms were produced in 2011. See also *Butene *Dimer (chemistry) *Octene Oct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetaldehyde
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3 CHO, sometimes abbreviated by chemists as MeCHO (Me = methyl). It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body. The International Agency for Research on Cancer (IARC) has listed acetaldehyde as a Group 1 carcinogen. Acetaldehyde is "one of the mos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bridging Ligand
In coordination chemistry, a bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually restricted to small ligands such as pseudohalides or to ligands that are specifically designed to link two metals. In naming a complex wherein a single atom bridges two metals, the bridging ligand is preceded by the Greek letter mu, μ, with a subscript number denoting the number of metals bound to the bridging ligand. μ2 is often denoted simply as μ. When describing coordination complexes care should be taken not to confuse μ with η ('eta'), which relates to hapticity. Ligands that are not bridging are called terminal ligands. List of bridging ligands Virtually all ligands are known to bridge, with the exception of amines and ammonia. Common bridging ligands include most of the common anions. Many simple organic ligands form str ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rhodium Trichloride
Rhodium(III) chloride refers to inorganic compounds with the formula RhCl3(H2O)''n'', where ''n'' varies from 0 to 3. These are diamagnetic solids featuring octahedral Rh(III) centres. Depending on the value of ''n'', the material is either a dense brown solid or a soluble reddish salt. The soluble trihydrated (n = 3) salt is widely used to prepare compounds used in homogeneous catalysis, notably for the industrial production of acetic acid and hydroformylation. Structures Aqueous solutions of RhCl3(H2O)3 have been characterized by 103Rh NMR spectroscopy, which shows the presence of several species. The proportions of which change with time and depend on the concentration of chloride. The relative distribution of these species determines the colour of the solutions, which can range from yellow (the hexaaquo ion) to "raspberry-red". Some of these species are h(H2O)6sup>3+, hCl(H2O)5sup>2+, ''cis''- and ''trans''- hCl2(H2O)4sup>+, and hCl3(H2O)3 Individual ions have been sep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Of Crystallization
In chemistry, water(s) of crystallization or water(s) of hydration are water molecules that are present inside crystals. Water is often incorporated in the formation of crystals from aqueous solutions. In some contexts, water of crystallization is the total mass of water in a substance at a given temperature and is mostly present in a definite (stoichiometric) ratio. Classically, "water of crystallization" refers to water that is found in the crystalline framework of a metal complex or a salt, which is not directly bonded to the metal cation. Upon crystallization from water, or water-containing solvents, many compounds incorporate water molecules in their crystalline frameworks. Water of crystallization can generally be removed by heating a sample but the crystalline properties are often lost. Compared to inorganic salts, proteins crystallize with large amounts of water in the crystal lattice. A water content of 50% is not uncommon for proteins. Applications Knowledge of hyd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |