|
Calcitonin Receptor
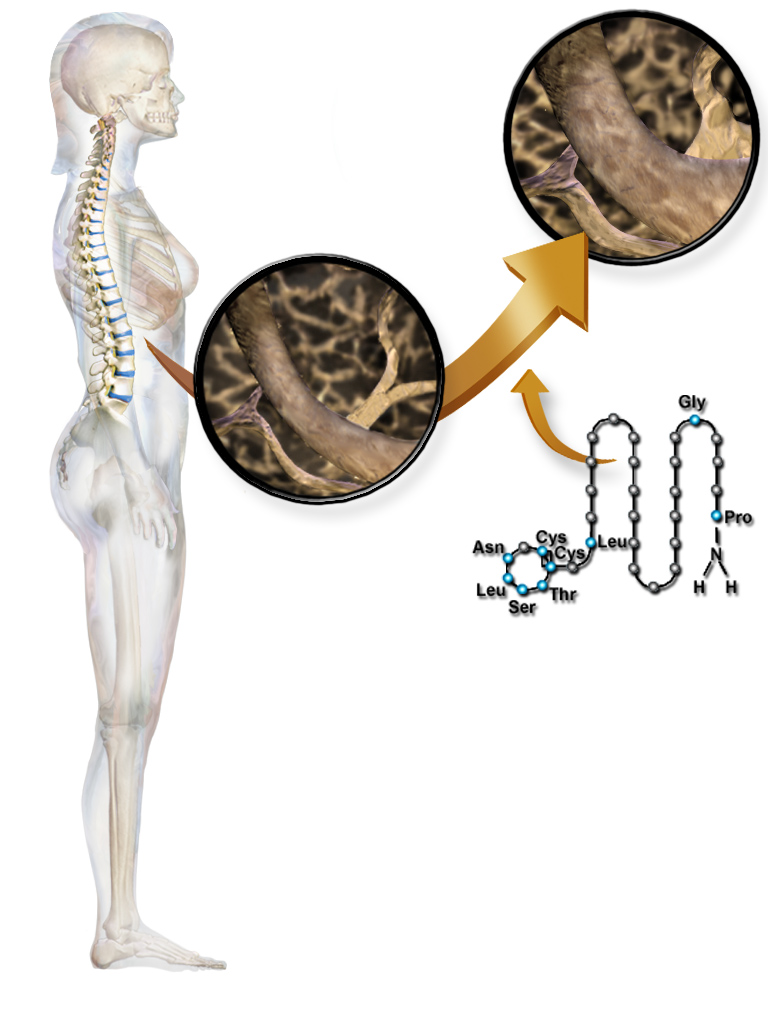
The calcitonin receptor (CT) is a G protein-coupled receptor that binds the peptide hormone calcitonin and is involved in maintenance of calcium homeostasis, particularly with respect to bone formation and metabolism. CT works by activating the G-proteins Gs and Gq often found on osteoclasts, on cells in the kidney, and on cells in a number of regions of the brain. It may also affect the ovaries in women and the testes in men. The function of the CT receptor protein is modified through its interaction with s (RAMPs), forming the multimeric |
G Protein-coupled Receptor
G protein-coupled receptors (GPCRs), also known as seven-(pass)-transmembrane domain receptors, 7TM receptors, heptahelical receptors, serpentine receptors, and G protein-linked receptors (GPLR), form a large group of evolutionarily-related proteins that are cell surface receptors that detect molecules outside the cell and activate cellular responses. Coupling with G proteins, they are called seven-transmembrane receptors because they pass through the cell membrane seven times. Text was copied from this source, which is available under Attribution 2.5 Generic (CC BY 2.5) license. Ligands can bind either to extracellular N-terminus and loops (e.g. glutamate receptors) or to the binding site within transmembrane helices (Rhodopsin-like family). They are all activated by agonists although a spontaneous auto-activation of an empty receptor can also be observed. G protein-coupled receptors are found only in eukaryotes, including yeast, choanoflagellates, and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcitonin
Calcitonin is a 32 amino acid peptide hormone secreted by parafollicular cells (also known as C cells) of the thyroid (or endostyle) in humans and other chordates. in the ultimopharyngeal body. It acts to reduce blood calcium (Ca2+), opposing the effects of parathyroid hormone (PTH). Its importance in humans has not been as well established as its importance in other animals, as its function is usually not significant in the regulation of normal calcium homeostasis. It belongs to the calcitonin-like protein family. Historically calcitonin has also been called thyrocalcitonin. Biosynthesis and regulation Calcitonin is formed by the proteolytic cleavage of a larger prepropeptide, which is the product of the CALC1 gene (). It is functionally an antagonist with PTH and Vitamin D3. The CALC1 gene belongs to a superfamily of related protein hormone precursors including islet amyloid precursor protein, calcitonin gene-related peptide, and the precursor of adrenomedullin. Secretion of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. It is the fifth most abundant element in Earth's crust, and the third most abundant metal, after iron and aluminium. The most common calcium compound on Earth is calcium carbonate, found in limestone and the fossilised remnants of early sea life; gypsum, anhydrite, fluorite, and apatite are also sources of calcium. The name derives from Latin ''calx'' "lime", which was obtained from heating limestone. Some calcium compounds were known to the ancients, though their chemistry was unknown until the seventeenth century. Pure calcium was isolated in 1808 via electrolysis of its oxide by Humphry Davy, who named the element. Calcium compounds are widely used in many industries: in foods and pharma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homeostasis
In biology, homeostasis (British English, British also homoeostasis) Help:IPA/English, (/hɒmɪə(ʊ)ˈsteɪsɪs/) is the state of steady internal, physics, physical, and chemistry, chemical conditions maintained by organism, living systems. This is the condition of optimal functioning for the organism and includes many variables, such as body temperature and fluid balance, being kept within certain pre-set limits (homeostatic range). Other variables include the pH of extracellular fluid, the concentrations of sodium, potassium and calcium ions, as well as that of the blood sugar level, and these need to be regulated despite changes in the environment, diet, or level of activity. Each of these variables is controlled by one or more regulators or homeostatic mechanisms, which together maintain life. Homeostasis is brought about by a natural resistance to change when already in the optimal conditions, and equilibrium is maintained by many regulatory mechanisms: it is thought to be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
G-protein
G proteins, also known as guanine nucleotide-binding proteins, are a family of proteins that act as molecular switches inside cells, and are involved in transmitting signals from a variety of stimuli outside a cell to its interior. Their activity is regulated by factors that control their ability to bind to and hydrolyze guanosine triphosphate (GTP) to guanosine diphosphate (GDP). When they are bound to GTP, they are 'on', and, when they are bound to GDP, they are 'off'. G proteins belong to the larger group of enzymes called GTPases. There are two classes of G proteins. The first function as monomeric small GTPases (small G-proteins), while the second function as heterotrimeric G protein complexes. The latter class of complexes is made up of ''alpha'' (α), ''beta'' (β) and ''gamma'' (γ) subunits. In addition, the beta and gamma subunits can form a stable dimeric complex referred to as the beta-gamma complex . Heterotrimeric G proteins located within the cell are activa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gs Alpha Subunit
The Gs alpha subunit (Gαs, Gsα) is a subunit of the heterotrimeric G protein Gs that stimulates the cAMP-dependent pathway by activating adenylyl cyclase. Gsα is a GTPase that functions as a cellular signaling protein. Gsα is the founding member of one of the four families of heterotrimeric G proteins, defined by the alpha subunits they contain: the Gαs family, Gαi/Gαo family, Gαq family, and Gα12/Gα13 family. The Gs-family has only two members: the other member is Golf, named for its predominant expression in the olfactory system. In humans, Gsα is encoded by the GNAS complex locus, while Golfα is encoded by the GNAL gene. Function The general function of Gs is to activate intracellular signaling pathways in response to activation of cell surface G protein-coupled receptors (GPCRs). GPCRs function as part of a three-component system of receptor-transducer-effector. The transducer in this system is a heterotrimeric G protein, composed of three subunits: a Gα ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gq Alpha Subunit
Gq protein alpha subunit is a family of heterotrimeric G protein alpha subunits. This family is also commonly called the Gq/11 (Gq/G11) family or Gq/11/14/15 family to include closely related family members. G alpha subunits may be referred to as Gq alpha, Gαq, or Gqα. Gq proteins couple to G protein-coupled receptors to activate beta-type phospholipase C (PLC-β) enzymes. PLC-β in turn hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) to diacyl glycerol (DAG) and inositol trisphosphate (IP3). IP3 acts as a second messenger to release stored calcium into the cytoplasm, while DAG acts as a second messenger that activates protein kinase C (PKC). Family members In humans, there are four distinct proteins in the Gq alpha subunit family: * Gαq is encoded by the gene GNAQ. * Gα11 is encoded by the gene GNA11. * Gα14 is encoded by the gene GNA14. * Gα15 is encoded by the gene GNA15. Function The general function of Gq is to activate intracellular signaling p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Receptor Activity-modifying Protein
Receptor activity-modifying proteins (RAMPs) are a class of protein that interact with and modulate the activities of several Class B G protein-coupled receptors including the receptors for secretin, calcitonin (CT), glucagon, and vasoactive intestinal peptide (VIP). There are three distinct types of RAMPs in mammals (though more in fish), designated RAMP1, RAMP2, and RAMP3, each encoded by a separate gene. Function Currently, the function of RAMPs is divided into classes of activities. When associated with the Calcitonin receptor (CTR) or Calcitonin receptor-like (CALCRL) (below), RAMPs can change the selectivity of the receptor for a specific hormone. In the cases of the other receptors mentioned, however, there is no evidence that they can do this, but instead function to regulate trafficking of receptors from the ER / golgi to the membrane. These functions appear to be ones where there is redundancy, as neither RAMP1 nor RAMP3 knockout mice (KO) have grossly abnorma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amylin
Amylin, or islet amyloid polypeptide (IAPP), is a 37-residue peptide hormone Peptide hormones or protein hormones are hormones whose molecules are peptide, or proteins, respectively. The latter have longer amino acid chain lengths than the former. These hormones have an effect on the endocrine system of animals, including h .... It is co-secreted with insulin from the pancreatic beta cell, β-cells in the ratio of approximately 100:1 (insulin:amylin). Amylin plays a role in Blood sugar level, glycemic regulation by slowing gastric emptying and promoting satiety, thereby preventing Postprandial, post-prandial spikes in blood glucose levels. IAPP is processed from an 89-residue coding sequence. Proislet amyloid polypeptide (proIAPP, proamylin, proislet protein) is produced in the pancreatic beta cells (β-cells) as a 67 amino acid, 7404 Dalton pro-peptide and undergoes post-translational modifications including protease cleavage to produce amylin. Synthesis ProIAPP consists ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Apolipoprotein B
Apolipoprotein B (ApoB) is a protein that in humans is encoded by the gene. Function Apolipoprotein B is the primary apolipoprotein of chylomicrons, VLDL, Lp(a), IDL, and LDL particles (LDL—commonly known as "bad cholesterol" when in reference to both heart disease and vascular disease in general), which is responsible for carrying fat molecules (lipids), including cholesterol, around the body to all cells within all tissues. While all the functional roles of ApoB within the LDL (and all larger) particles remain somewhat unclear, it is the primary organizing protein (of the entire complex shell enclosing/carrying fat molecules within) component of the particles and is absolutely required for the formation of these particles. What is also clear is that the ApoB on the LDL particle acts as a ligand for LDL receptors in various cells throughout the body (i.e., less formally, ApoB indicates fat carrying particles are ready to enter any cells with ApoB receptors and deliver fat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
LRP1
Low density lipoprotein receptor-related protein 1 (LRP1), also known as alpha-2-macroglobulin receptor (A2MR), apolipoprotein E receptor (APOER) or cluster of differentiation 91 (CD91), is a protein forming a receptor found in the plasma membrane of cells involved in receptor-mediated endocytosis. In humans, the LRP1 protein is encoded by the ''LRP1'' gene. LRP1 is also a key signalling protein and, thus, involved in various biological processes, such as lipoprotein metabolism and cell motility, and diseases, such as neurodegenerative diseases, atherosclerosis, and cancer. Structure The ''LRP1'' gene encodes a 600 kDa precursor protein that is processed by furin in the trans-Golgi complex, resulting in a 515 kDa alpha-chain and an 85 kDa beta-chain associated noncovalently. As a member of the LDLR family, LRP1 contains cysteine-rich complement-type repeats, EGF (gene) repeats, β-propeller domains, a transmembrane domain, and a cytoplasmic domain. The extracellular domain of L ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |