|
Bailey Peptide Synthesis
The Bailey peptide synthesis is a name reaction in organic chemistry developed 1949 by J. L. Bailey. It is a method for the synthesis of a peptide from ''α''-amino acid-''N''-carboxylic acid anhydrides (NCAs) and amino acids or peptide esters.J. Leggett Bailey: ''A new Peptide Synthesis.'' In: ''Nature.'' Band 164, Nr. 4177, 1949, S. 889, DOI:10.1038/164889a0. The reaction is characterized by short reaction times and a high yield of the target peptide. The reaction can be carried out at low temperatures in organic solvents. The residues R1 and R2 can be organic groups or hydrogen atoms, R3 is the used amino acid or peptide ester: Reaction mechanism The reaction mechanism is not known in detail. Supposedly, the reaction begins with a nucleophilic attack of the amino group on the carbonyl carbon of the anhydride group of the N-carboxylic acid anhydride (1). After an intramolecular proton migration, a 1,4-proton shift and the cleavage of carbon dioxide follows, resulting in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Name Reaction
A name reaction is a chemical reaction named after its discoverers or developers. Among the tens of thousands of organic reactions that are known, hundreds of such reactions are well-known enough to be named after people. Well-known examples include the Grignard reaction, the Sabatier reaction, the Wittig reaction, the Claisen condensation, the Friedel-Crafts acylation, and the Diels-Alder reaction. Books have been published devoted exclusively to name reactions;Alfred Hassner, C. Stumer. ''Organic syntheses based on name reactions''. Elsevier, 2002. Li, Jie Jack. ''Name Reactions: A Collection of Detailed Reaction Mechanisms''. Springer, 2003. the Merck Index, a chemical encyclopedia, also includes an appendix on name reactions. As organic chemistry developed during the 20th century, chemists started associating synthetically useful reactions with the names of the discoverers or developers; in many cases, the name is merely a mnemonic. Some cases of reactions that were not real ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzyl Group
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure . Benzyl features a benzene ring () attached to a methylene group () group. Nomenclature In IUPAC nomenclature, the prefix benzyl refers to a substituent, for example benzyl chloride or benzyl benzoate. Benzyl is not to be confused with phenyl with the formula . The term benzylic is used to describe the position of the first carbon bonded to a benzene or other aromatic ring. For example, is referred to as a "benzylic" carbocation. The benzyl free radical has the formula . The benzyl cation or phenylcarbenium ion is the carbocation with formula ; the benzyl anion or phenylmethanide ion is the carbanion with the formula . None of these species can be formed in significant amounts in the solution phase under normal conditions, but they are useful referents for discussion of reaction mechanisms and may exist as reactive intermediates. Abbreviations The abbreviation "Bn" denotes benzyl. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogenic amino acids. It is encoded by all the codons starting with GG (GGU, GGC, GGA, GGG). Glycine is integral to the formation of alpha-helices in secondary protein structure due to its compact form. For the same reason, it is the most abundant amino acid in collagen triple-helices. Glycine is also an inhibitory neurotransmitter – interference with its release within the spinal cord (such as during a '' Clostridium tetani'' infection) can cause spastic paralysis due to uninhibited muscle contraction. It is the only achiral proteinogenic amino acid. It can fit into hydrophilic or hydrophobic environments, due to its minimal side chain of only one hydrogen atom. History and etymology Glycine was discovered in 1820 by the French ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tyrosin
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek ''tyrós'', meaning ''cheese'', as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is encoded by the codons UAC and UAU in messenger RNA. Functions Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases. Phosphorylation of the hydroxyl group can change the activity of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycin
Glycin, or N-(4-hydroxyphenyl)glycine, is N-substituted p-aminophenol. It is a photographic developing agent used in classic black-and-white developer solutions. It is unrelated to the amino acid glycine. It is typically characterized as thin plates of white or silvery powder, although aged samples appear brown. It is sparingly soluble in water and most organic solvents; it is readily soluble in alkalies and acids.{{cite encyclopedia, last1=Mitchell, first1=Stephen C., last2=Waring, first2=Rosemary H., encyclopedia=Ullmann's Encyclopedia of Industrial Chemistry, publisher=Wiley-VCH, year=2000, doi=10.1002/14356007.a02_099, isbn=978-3527306732, chapter=Aminophenols Glycin is structurally related to 4-aminophenol and Metol. Decarboxylation of glycin gives Metol. Glycin has a milder reduction potential than Metol. The two developers have markedly different character. Glycin is slower-acting, but much longer-lasting in solution. Glycin is rarely used as a developing agent, primarily ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
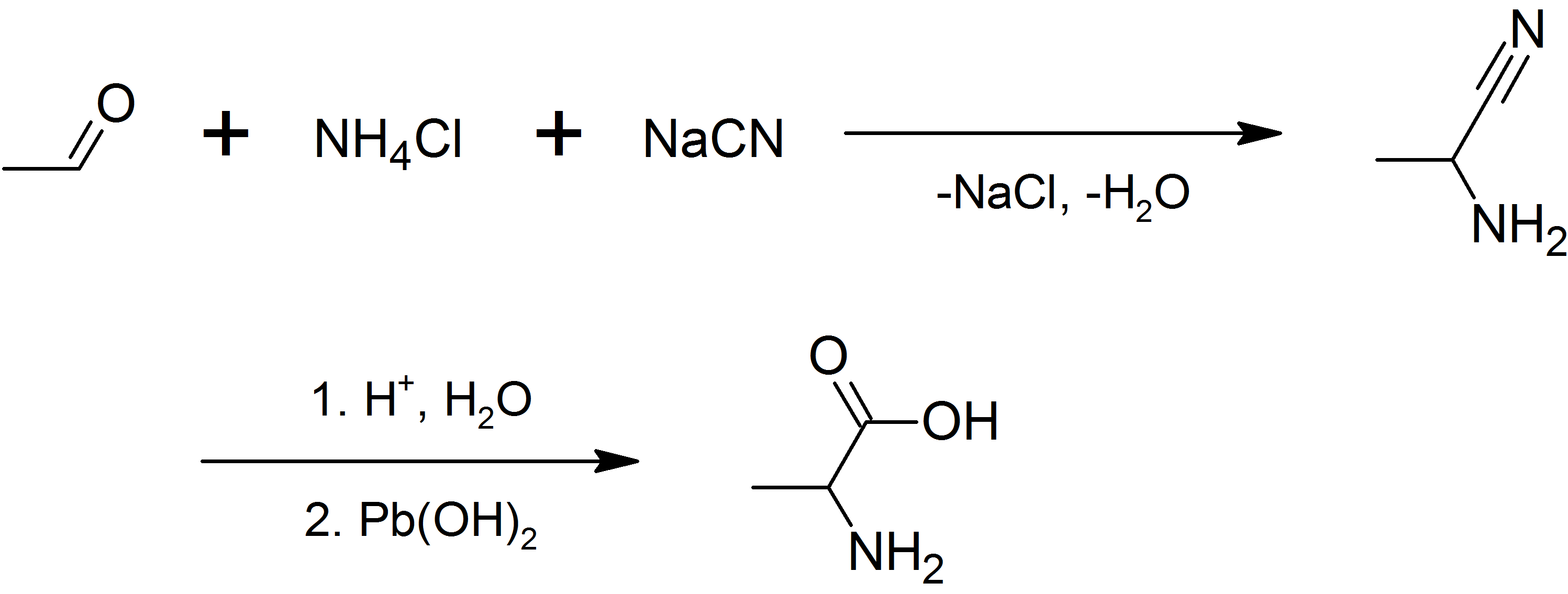
Alanine
Alanine (symbol Ala or A), or α-alanine, is an α-amino acid that is used in the biosynthesis of proteins. It contains an amine group and a carboxylic acid group, both attached to the central carbon atom which also carries a methyl group side chain. Consequently, its IUPAC systematic name is 2-aminopropanoic acid, and it is classified as a nonpolar, aliphatic α-amino acid. Under biological conditions, it exists in its zwitterionic form with its amine group protonated (as −NH3+) and its carboxyl group deprotonated (as −CO2−). It is non-essential to humans as it can be synthesised metabolically and does not need to be present in the diet. It is encoded by all codons starting with GC (GCU, GCC, GCA, and GCG). The L-isomer of alanine (left-handed) is the one that is incorporated into proteins. L-alanine is second only to leucine in rate of occurrence, accounting for 7.8% of the primary structure in a sample of 1,150 proteins. The right-handed form, D-alanine, occurs ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxalyl Chloride
Oxalyl chloride is an organic chemical compound with the formula (COCl)2. This colorless, sharp-smelling liquid, the diacyl chloride of oxalic acid, is a useful reagent in organic synthesis. Preparation Oxalyl chloride was first prepared in 1892 by the French chemist Adrien Fauconnier, who reacted diethyl oxalate with phosphorus pentachloride. It can also be prepared by treating oxalic acid with phosphorus pentachloride. Oxalyl chloride is produced commercially from ethylene carbonate. Photochlorination gives the tetrachloride, which is subsequently degraded: :C2H4O2CO + 4 Cl2 → C2Cl4O2CO + 4 HCl :C2Cl4O2CO → C2O2Cl2 + COCl2 Reactions Oxalyl chloride reacts with water giving off gaseous products only: hydrogen chloride (HCl), carbon dioxide (CO2), and carbon monoxide (CO). : In this, it is quite different from other acyl chlorides which hydrolyze with formation of hydrogen chloride and the original carboxylic acid. Applications in organic synthesis Oxidation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
By-product
A by-product or byproduct is a secondary product derived from a production process, manufacturing process or chemical reaction; it is not the primary product or service being produced. A by-product can be useful and marketable or it can be considered waste: for example, bran, which is a byproduct of the milling of wheat into refined flour, is sometimes composted or burned for disposal, but in other cases, it can be used as a nutritious ingredient in human food or animal feed. Gasoline was once a byproduct of oil refining that later became a desirable commodity as motor fuel. The plastic used in plastic shopping bags also started as a by-product of oil refining. In economics In the context of production, a by-product is the "output from a joint production process that is minor in quantity and/or net realizable value (NRV) when compared with the main products". Because they are deemed to have no influence on reported financial results, by-products do not receive allocations ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protecting Group
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis. In many preparations of delicate organic compounds, some specific parts of their molecules cannot survive the required reagents or chemical environments. Then, these parts, or groups, must be protected. For example, lithium aluminium hydride is a highly reactive but useful reagent capable of reducing esters to alcohols. It will always react with carbonyl groups, and this cannot be discouraged by any means. When a reduction of an ester is required in the presence of a carbonyl, the attack of the hydride on the carbonyl has to be prevented. For example, the carbonyl is converted into an acetal, which does not react with hydrides. The acetal is then called a protecting group for the carbonyl. After the step involving the hydride is complete, the a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tert-Butyloxycarbonyl Protecting Group
The ''tert''-butyloxycarbonyl protecting group or ''tert''-butoxycarbonyl protecting group (BOC group) is a protecting group used in organic synthesis. The BOC group can be added to the amine under aqueous conditions using di-''tert''-butyl dicarbonate in the presence of a base such as sodium carbonate: Protection of the amine can also be accomplished in acetonitrile solution using 4-dimethylaminopyridine (DMAP) as the base. Removal of the BOC in amino acids can be accomplished with strong acids such as trifluoroacetic acid in dichloromethane, or with HCl in methanol. A complication may be the tendency of the ''t''-butyl cation intermediate to alkylate other nucleophiles; scavengers such as anisole or thioanisole may be used. Selective cleavage of the ''N''-Boc group in the presence of other protecting groups is possible when using AlCl3. Sequential treatment with trimethylsilyl iodide then methanol can also be used for Boc deprotection, especially where other deprotec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |