|
Anodic Protection
Anodic protection (AP) otherwise refer as Anodic Control is a technique to control the corrosion of a metal surface by making it the anode of an electrochemical cell and controlling the electrode potential in an zone where the metal is passive. Anodic protection is used to protect metals that exhibit passivation in environments whereby the current density in the freely corroding state is significantly higher than the current density in the passive state over a wide range of potentials. Anodic protection is used for carbon steel storage tanks containing extreme pH environments including concentrated sulfuric acid and 50 percent caustic soda where cathodic protection is not suitable due to very high current requirements. In anodic protection potentiostat is used to maintain a metal at constant potential with respect to reference electrode. Out of three terminals of the potentiostat one is connected to tank to be protected, another to an auxiliary cathode(platinum) and the third to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion. In the most common use of the word, this means electrochemical oxidation of metal in reaction with an oxidant such as oxygen, hydrogen or hydroxide. Rusting, the formation of iron oxides, is a well-known example of electrochemical corrosion. This type of damage typically produces oxide(s) or salt(s) of the original metal and results in a distinctive orange colouration. Corrosion can also occur in materials other than metals, such as ceramics or polymers, although in this context, the term "degradation" is more common. Corrosion degrades the useful properties of materials and structures including strength, appearance and permeability to liquids and gases. Many structural ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
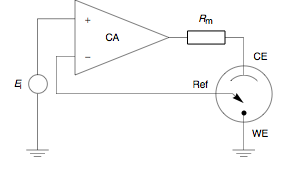
Potentiostat
A potentiostat is the electronic hardware required to control a three electrode cell and run most electroanalytical experiments. A ''Bipotentiostat'' and ''polypotentiostat'' are potentiostats capable of controlling two working electrodes and more than two working electrodes, respectively. The system functions by maintaining the potential of the working electrode at a constant level with respect to the reference electrode by adjusting the current at an auxiliary electrode. The heart of the different potentiostatic electronic circuits is an operational amplifier (op amp). It consists of an electric circuit which is usually described in terms of simple op amps. Primary use This equipment is fundamental to modern electrochemical studies using three electrode systems for investigations of reaction mechanisms related to redox chemistry and other chemical phenomena. The dimensions of the resulting data depend on the experiment. In voltammetry, electric current in amps is pl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wetting Voltage
In electrical and electronics engineering, wetting current is the minimum electric current needing to flow through a contact to break through the surface film resistance at a contact. It is typically far below the contact's nominal maximum current rating. A thin film of oxidation, or an otherwise passivated layer, tends to form in most environments, particularly those with high humidity, and, along with surface roughness, contributes to the contact resistance at an interface. Providing a sufficient amount of wetting current is a crucial step in designing Control engineering, systems that use delicate switches with small contact pressure as sensor inputs. Failing to do this might result in switches remaining electrically "open" when pressed, due to contact oxidation. Capacitor discharge solution In some low voltage applications, where switching current is below the manufacturer's wetting current specification, a capacitor discharge method may be employed by placing a small capaci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sacrificial Metal
A sacrificial metal is a metal used as a sacrificial anode in cathodic protection that corrodes to prevent a primary metal from corrosion or rusting. It may also be used for galvanization. Equation When two metals touch each other and water is present, electrolysis occurs. One well known example is the reaction between zinc (Zn) and iron (Fe). Zinc atoms will lose electrons in preference to the iron as they are more electropositive and therefore zinc is oxidized and corrodes. Zn(s)→(aq) +2e (oxidation) Capacity derivation from 1st Principles The capacity of a sacrificial metal may be calculated from first principle as follows: * 1 Kg Al = 1000/27 moles Al * 1 Kg Al = 3 x 1000/27 moles of electrons * 1 Kg Al = 3 x 1000/27 x 96494 coulombs of charge (by Faraday principles) * = 10.72 x 106 Amp.seconds of charge per Kg Al (1 Coulomb = 1 Amp.Second) * = 10.72 x 106/3600 = 2978 Amp.Hours per Kg By similar calculations Zinc and Magnesium have a capacity of 825 and 2206 Amp.Hou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion Engineering
Corrosion engineering is an engineering specialty that applies scientific, technical, engineering skills, and knowledge of natural laws and physical resources to design and implement materials, structures, devices, systems, and procedures to manage corrosion. From a holistic perspective, corrosion is the phenomenon of metals returning to the state they are found in nature. The driving force that causes metals to corrode is a consequence of their temporary existence in metallic form. To produce metals starting from naturally occurring minerals and ores, it is necessary to provide a certain amount of energy, e.g. Iron ore in a blast furnace. It is therefore thermodynamically inevitable that these metals when exposed to various environments would revert to their state found in nature. Corrosion and corrosion engineering thus involves a study of chemical kinetics, thermodynamics, electrochemistry and materials science. General background Generally related to metallurgy or material ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cathodic Protection
Cathodic protection (CP; ) is a technique used to control the corrosion of a metal surface by making it the cathode of an electrochemical cell. A simple method of protection connects the metal to be protected to a more easily corroded "sacrificial metal" to act as the anode. The sacrificial metal then corrodes instead of the protected metal. For structures such as long pipelines, where passive galvanic cathodic protection is not adequate, an external DC electrical power source is used to provide sufficient current. Cathodic protection systems protect a wide range of metallic structures in various environments. Common applications are: steel water or fuel pipelines and steel storage tanks such as home water heaters; steel pier piles; ship and boat hulls; offshore oil platforms and onshore oil well casings; offshore wind farm foundations and metal reinforcement bars in concrete buildings and structures. Another common application is in galvanized steel, in which a sacrif ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reference Electrode
A reference electrode is an electrode which has a stable and well-known electrode potential. The high stability of the electrode potential is usually reached by employing a redox system with constant (buffered or saturated) concentrations of each participant of the redox reaction. There are many ways reference electrodes are used. The simplest is when the reference electrode is used as a half-cell to build an electrochemical cell. This allows the reduction potential, potential of the other half cell to be determined. An accurate and practical method to measure an electrode's potential in isolation (absolute electrode potential) has yet to be developed. Aqueous reference electrodes Common reference electrodes and potential with respect to the standard hydrogen electrode (SHE): * Standard hydrogen electrode (SHE) (E = 0.000 V) activity of H+ = 1 Molar * Normal hydrogen electrode (NHE) (E ≈ 0.000 V) concentration H+ = 1 Molar * Reversible hydrogen electrode (RHE) (E = 0.000 V ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cathode
A cathode is the electrode from which a conventional current leaves a polarized electrical device. This definition can be recalled by using the mnemonic ''CCD'' for ''Cathode Current Departs''. A conventional current describes the direction in which positive charges move. Electrons have a negative electrical charge, so the movement of electrons is opposite to that of the conventional current flow. Consequently, the mnemonic ''cathode current departs'' also means that electrons flow ''into'' the device's cathode from the external circuit. For example, the end of a household battery marked with a + (plus) is the cathode. The electrode through which conventional current flows the other way, into the device, is termed an anode. Charge flow Conventional current flows from cathode to anode outside of the cell or device (with electrons moving in the opposite direction), regardless of the cell or device type and operating mode. Cathode polarity with respect to the anode can be positive ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Power Supply
A power supply is an electrical device that supplies electric power to an electrical load. The main purpose of a power supply is to convert electric current from a source to the correct voltage, current, and frequency to power the load. As a result, power supplies are sometimes referred to as electric power converters. Some power supplies are separate standalone pieces of equipment, while others are built into the load appliances that they power. Examples of the latter include power supplies found in desktop computers and consumer electronics devices. Other functions that power supplies may perform include limiting the current drawn by the load to safe levels, shutting off the current in the event of an electrical fault, power conditioning to prevent electronic noise or voltage surges on the input from reaching the load, power-factor correction, and storing energy so it can continue to power the load in the event of a temporary interruption in the source power (uninterruptible ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal. Its name originates from Spanish , a diminutive of "silver". Platinum is a member of the platinum group of elements and group 10 of the periodic table of elements. It has six naturally occurring isotopes. It is one of the rarer elements in Earth's crust, with an average abundance of approximately 5 μg/kg. It occurs in some nickel and copper ores along with some native deposits, mostly in South Africa, which accounts for ~80% of the world production. Because of its scarcity in Earth's crust, only a few hundred tonnes are produced annually, and given its important uses, it is highly valuable and is a major precious metal commodity. Platinum is one of the least reactive metals. It has remarkable resistance to corrosion, even at high temperatures, and is therefore considered a noble metal. Consequent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Current Density
In electromagnetism, current density is the amount of charge per unit time that flows through a unit area of a chosen cross section. The current density vector is defined as a vector whose magnitude is the electric current per cross-sectional area at a given point in space, its direction being that of the motion of the positive charges at this point. In SI base units, the electric current density is measured in amperes per square metre. Definition Assume that ''A'' (SI unit: m2) is a small surface centred at a given point ''M'' and orthogonal to the motion of the charges at ''M''. If ''I'' (SI unit: A) is the electric current flowing through ''A'', then electric current density ''j'' at ''M'' is given by the limit: :j = \lim_ \frac = \left.\frac \_, with surface ''A'' remaining centered at ''M'' and orthogonal to the motion of the charges during the limit process. The current density vector j is the vector whose magnitude is the electric current density, and whose dire ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ACID, for "anode current into device". The direction of conventional current (the flow of positive charges) in a circuit is opposite to the direction of electron flow, so (negatively charged) electrons flow out the anode of a galvanic cell, into an outside or external circuit connected to the cell. For example, the end of a household battery marked with a "-" (minus) is the anode. In both a galvanic cell and an electrolytic cell, the anode is the electrode at which the oxidation reaction occurs. In a galvanic cell the anode is the wire or plate having excess negative charge as a result of the oxidation reaction. In an electrolytic cell, the anode is the wire or plate upon which excess positive charge is imposed. As a result of this, anion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |