|
Acidic Oxides
An acidic oxide is an oxide that either produces an acidic solution upon addition to water, or acts as an acceptor of hydroxide ions effectively functioning as a Lewis acid. Acidic oxides will typically have a low pKa and may be inorganic or organic. A commonly encountered acidic oxide, carbon dioxide produces an acidic solution (and the generation of carbonic acid) when dissolved. The acidity of an oxide can be reasonably assumed by its accompanying constituents. Less electronegative elements tend to form basic oxides such as sodium oxide and magnesium oxide, whereas more electronegative elements tend to produce acidic oxides as seen with carbon dioxide and phosphorus pentoxide. Some oxides like aluminium oxides are amphoteric. Acidic oxides are of environmental concern. Sulfur and nitrogen oxides are considered air pollutants as they react with atmospheric water vapour to produce acid rain. Examples Carbonic acid is an illustrative example of the Lewis acidity of an acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. It functions as a base, a ligand, a nucleophile, and a catalyst. The hydroxide ion forms salts, some of which dissociate in aqueous solution, liberating solvated hydroxide ions. Sodium hydroxide is a multi-million-ton per annum commodity chemical. The corresponding electrically neutral compound HO• is the hydroxyl radical. The corresponding covalently bound group –OH of atoms is the hydroxy group. Both the hydroxide ion and hydroxy group are nucleophiles and can act as catalysts in organic chemistry. Many inorganic substances which bear the word ''hydroxide'' in their names are not ionic compounds of the hydroxide ion, but covalent compounds which contain hydroxy groups. Hydroxide ion The hydroxide ion is a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
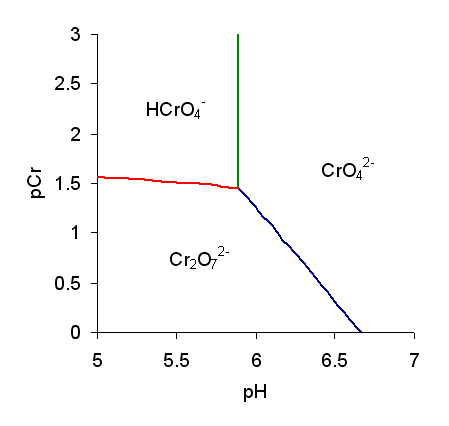
Chromic Acid
The term chromic acid is usually used for a mixture made by adding concentrated sulfuric acid to a dichromate, which may contain a variety of compounds, including solid chromium trioxide. This kind of chromic acid may be used as a cleaning mixture for glass. Chromic acid may also refer to the molecular species, H2CrO4 of which the trioxide is the anhydride. Chromic acid features chromium in an oxidation state of +6 (or VI). It is a strong and corrosive oxidising agent. Molecular chromic acid Molecular chromic acid, H2CrO4, has much in common with sulfuric acid, H2SO4. Only sulfuric acid can be classified as part of the 7 strong acids list. Due to the laws pertinent to the concept of "first order ionization energy", the first proton is lost most easily. It behaves extremely similar to sulfuric acid deprotonation. Since the process of polyvalent acid-base titrations have more than one proton (especially when the acid is starting substance and the base is the titrant), protons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Trioxide
Phosphorus trioxide is the chemical compound with the molecular formula P4O6. Although the molecular formula suggests the name tetraphosphorus hexaoxide, the name phosphorus trioxide preceded the knowledge of the compound's molecular structure, and its usage continues today. This colorless solid is structurally related to adamantane. It is formally the anhydride of phosphorous acid, H3PO3, but cannot be obtained by the dehydration of the acid. A white solid that melts at room temperature, it is waxy, crystalline and highly toxic, with garlic odour. Preparation It is obtained by the combustion of phosphorus in a limited supply of air at low temperatures. :P4 + 3 O2 → P4O6 By-products include red phosphorus suboxide. Chemical properties Phosphorus trioxide reacts with water to form phosphorous acid, reflecting the fact that it is the anhydride of that acid. : P4O6 + 6 H2O → 4 H3PO3 It reacts with hydrogen chloride to form H3PO3 and phosphorus trichloride. : P4 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus(V) Oxide
Phosphorus pentoxide is a chemical compound with molecular formula P4 O10 (with its common name derived from its empirical formula, P2O5). This white crystalline solid is the anhydride of phosphoric acid. It is a powerful desiccant and dehydrating agent. Structure Phosphorus pentoxide crystallizes in at least four forms or polymorphs. The most familiar one, a metastable form (shown in the figure), comprises molecules of P4O10. Weak van der Waals forces hold these molecules together in a hexagonal lattice (However, in spite of the high symmetry of the molecules, the crystal packing is not a close packing). The structure of the P4O10 cage is reminiscent of adamantane with ''T''d symmetry point group. It is closely related to the corresponding anhydride of phosphorous acid, P4O6. The latter lacks terminal oxo groups. Its density is 2.30 g/cm3. It boils at 423 °C under atmospheric pressure; if heated more rapidly it can sublimate. This form can be made by condensing the vap ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorous Acid
Phosphorous acid (or phosphonic acid (singular)) is the compound described by the formula H3PO3. This acid is diprotic (readily ionizes two protons), not triprotic as might be suggested by this formula. Phosphorous acid is an intermediate in the preparation of other phosphorus compounds. Organic derivatives of phosphorous acid, compounds with the formula RPO3H2, are called phosphonic acids. Nomenclature and tautomerism H3PO3 is more clearly described with the structural formula HPO(OH)2. In the solid state, has tetrahedral geometry about the central phosphorus atom, with a P–H bond of 132 pm, one P=O double bond of 148 pm and two longer P–O(H) single bonds of 154 pm. This species exists in equilibrium with an extremely minor tautomer . IUPAC recommends that the latter be called phosphorous acid, whereas the dihydroxy form is called phosphonic acid.. Only the reduced phosphorus compounds are spelled with an "ous" ending. Other important oxyacids of ph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus(III) Oxide
Phosphorus trioxide is the chemical compound with the molecular formula P4O6. Although the molecular formula suggests the name tetraphosphorus hexaoxide, the name phosphorus trioxide preceded the knowledge of the compound's molecular structure, and its usage continues today. This colorless solid is structurally related to adamantane. It is formally the anhydride of phosphorous acid, H3PO3, but cannot be obtained by the dehydration of the acid. A white solid that melts at room temperature, it is waxy, crystalline and highly toxic, with garlic odour. Preparation It is obtained by the combustion of phosphorus in a limited supply of air at low temperatures. :P4 + 3 O2 → P4O6 By-products include red phosphorus suboxide. Chemical properties Phosphorus trioxide reacts with water to form phosphorous acid, reflecting the fact that it is the anhydride of that acid. : P4O6 + 6 H2O → 4 H3PO3 It reacts with hydrogen chloride to form H3PO3 and phosphorus trichloride. : P4O6 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicic Acid
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and most abundant families of materials, existing as a compound of several minerals and as a synthetic product. Notable examples include fused quartz, fumed silica, silica gel, opal and aerogels. It is used in structural materials, microelectronics (as an electrical insulator), and as components in the food and pharmaceutical industries. Structure In the majority of silicates, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central Si atomsee 3-D Unit Cell. Thus, SiO2 forms 3-dimensional network solids in which each silicon atom is covalently bonded in a tetrahedral manner to 4 oxygen atoms. In contrast, CO2 is a linear molecule. The starkly different structures of the dioxide ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
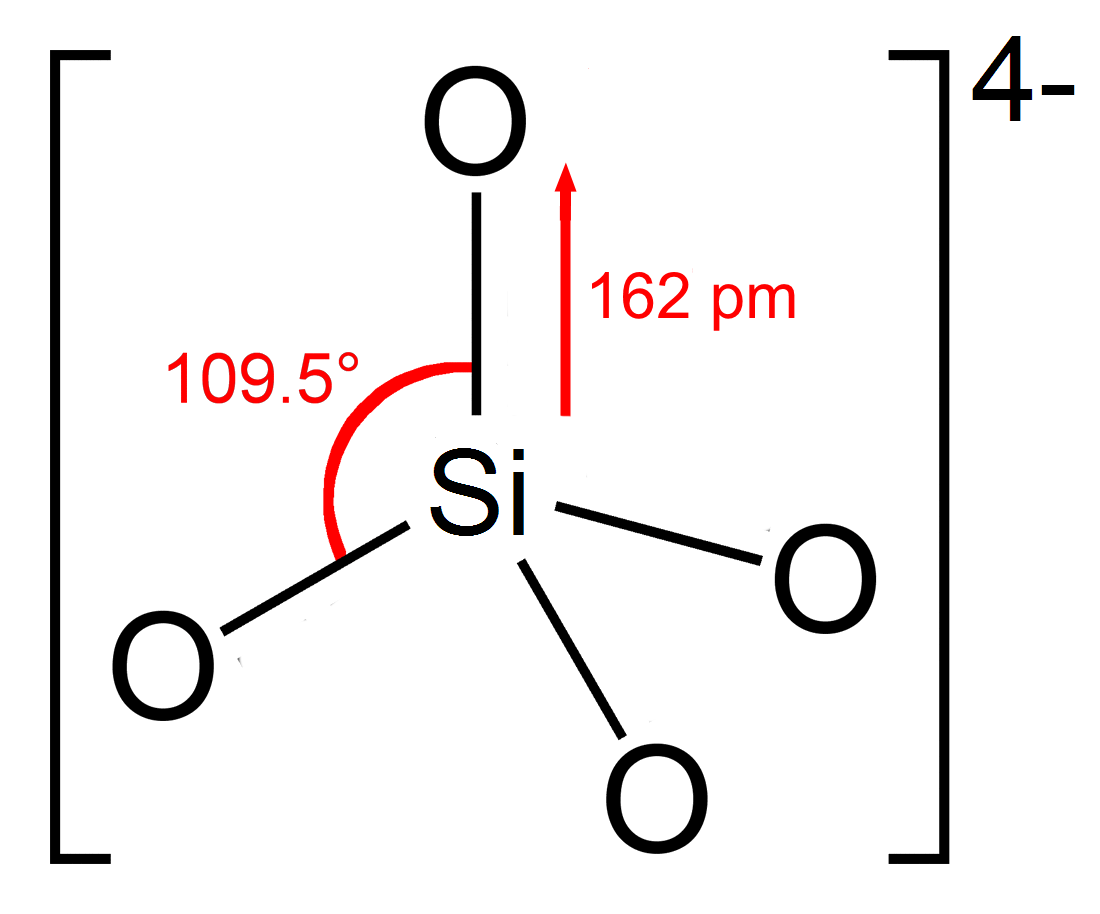
Silicate
In chemistry, a silicate is any member of a family of polyatomic anions consisting of silicon and oxygen, usually with the general formula , where . The family includes orthosilicate (), metasilicate (), and pyrosilicate (, ). The name is also used for any salt of such anions, such as sodium metasilicate; or any ester containing the corresponding chemical group, such as tetramethyl orthosilicate. The name "silicate" is sometimes extended to any anions containing silicon, even if they do not fit the general formula or contain other atoms besides oxygen; such as hexafluorosilicate .Most commonly, silicates are encountered as silicate minerals. For diverse manufacturing, technological, and artistic needs, silicates are versatile materials, both natural (such as granite, gravel, and garnet) and artificial (such as Portland cement, ceramics, glass, and waterglass). Structural principles In all silicates, silicon atom occupies the center of an idealized tetrahedron whose c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Dioxide
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and most abundant families of materials, existing as a compound of several minerals and as a synthetic product. Notable examples include fused quartz, fumed silica, silica gel, opal and aerogels. It is used in structural materials, microelectronics (as an electrical insulator), and as components in the food and pharmaceutical industries. Structure In the majority of silicates, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central Si atomsee 3-D Unit Cell. Thus, SiO2 forms 3-dimensional network solids in which each silicon atom is covalently bonded in a tetrahedral manner to 4 oxygen atoms. In contrast, CO2 is a linear molecule. The starkly different structures of the d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminate
In chemistry, an aluminate is a compound containing an oxyanion of aluminium, such as sodium aluminate. In the naming of inorganic compounds, it is a suffix that indicates a polyatomic anion with a central aluminum atom. Aluminate oxyanions Aluminium oxide (alumina) is amphoteric: it dissolves in both bases and acids. When dissolved in bases it forms hydroxyaluminate ions in the same way as aluminium hydroxide or aluminium salts. The hydroxyaluminate or hydrated aluminate can be precipitated and then calcined to produce anhydrous aluminates. Aluminates are often formulated as a combination of basic oxide and aluminium oxide, for example the formula of anhydrous sodium aluminate NaAlO2 would be shown as Na2O·Al2O3. A number of aluminate oxyanions are known: * The simplest is the approximately tetrahedral found in the compound Na5AlO4, * framework ions in anhydrous sodium aluminate NaAlO2 and monocalcium aluminate, CaAl2O4 made up of corner-sharing tetrahedra. * A ring anion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Permanganic Acid
Permanganic acid (or manganic(VII) acid) is the inorganic compound with the formula H MnO4. This strong oxoacid has been isolated as its dihydrate. It is the conjugate acid of permanganate salts. It is the subject of few publications and its characterization as well as its uses are very limited. Preparation and structure Permanganic acid is most often prepared by the reaction of dilute sulfuric acid with a solution of barium permanganate, the insoluble barium sulfate byproduct being removed by filtering: :Ba(MnO4)2 + H2SO4 → 2 HMnO4 + BaSO4↓ The sulfuric acid used must be dilute; reactions of permanganates with concentrated sulfuric acid yield the anhydride, manganese heptoxide. Permanganic acid has also been prepared through the reaction of hydrofluorosilicic acid with potassium permanganate, through electrolysis, and through hydrolysis of manganese heptoxide, though the last route often results in explosions. Crystalline permanganic acid has been prepared at low temp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |