|
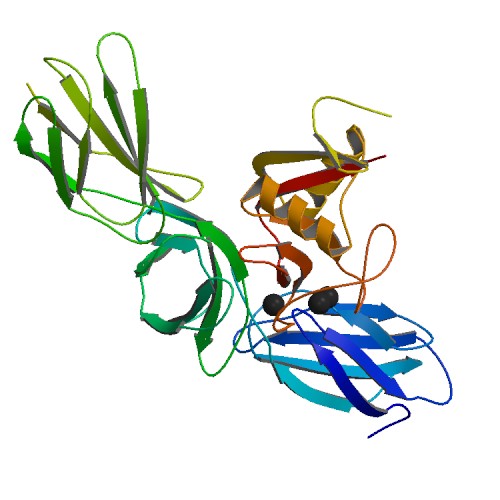
ADAMTS5
A disintegrin and metalloproteinase with thrombospondin motifs 5 also known as ADAMTS5 is an enzyme that in humans is encoded by the ''ADAMTS5'' gene. Function ADAMTS5 is a member of the ADAMTS (a disintegrin and metalloproteinase with thrombospondin motifs) protein family. Members of the family share several distinct protein modules, including a propeptide region, a metalloproteinase domain, a disintegrin-like domain, and a thrombospondin type 1 (TS) motif. Individual members of this family differ in the number of C-terminal TS motifs, and some have unique C-terminal domains. The enzyme encoded by this gene contains two C-terminal TS motifs and functions as aggrecanase to cleave aggrecan, a major proteoglycan of cartilage. ADAMTS5 may also have a role in the pathogenesis of human osteoarthritis. Animal studies Genetically modified mice in which the catalytic domain of ADAMTS5 was deleted are resistant to cartilage destruction in an experimental model of osteoarthritis. ADA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ADAMTS
ADAMTS (short for a disintegrin and metalloproteinase with thrombospondin motifs) is a family of multidomain extracellular protease enzymes. 19 members of this family have been identified in humans, the first of which, ADAMTS1, was described in 1997. Known functions of the ADAMTS proteases include processing of procollagens and von Willebrand factor as well as cleavage of aggrecan, versican, brevican and neurocan, making them key remodeling enzymes of the extracellular matrix. They have been demonstrated to have important roles in connective tissue organization, coagulation, inflammation, arthritis, angiogenesis and cell migration. Homologous subfamily of ADAMTSL (ADAMTS-like) proteins, which lack enzymatic activity, has also been described. Most cases of thrombotic thrombocytopenic purpura arise from autoantibody-mediated inhibition of ADAMTS13. Like ADAMs, the name of the ADAMTS family refers to its disintegrin and metalloproteinase activity, and in the case of ADAMTS, the prese ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thrombospondin
Thrombospondins (TSPs) are a family of secreted glycoproteins with antiangiogenic functions. Due to their dynamic role within the extracellular matrix they are considered matricellular proteins. The first member of the family, thrombospondin 1 (THBS1), was discovered in 1971 by Nancy L. Baenziger. Types The thrombospondins are a family of multifunctional proteins. The family consists of thrombospondins 1-5 and can be divided into 2 subgroups: A, which contains TSP-1 and TSP-2, and B, which contains TSP-3, TSP-4 and TSP-5 (also designated cartilage oligomeric protein or COMP). TSP-1 and TSP-2 are homotrimers, consisting of three identical subunits, whereas TSP-3, TSP-4 and TSP-5 are homopentamers. TSP-1 and TSP-2 are produced by immature astrocytes during brain development, which promotes the development of new synapses. Thrombospondin 1 Thrombospondin 1 (TSP-1) is encoded by THBS1. It was first isolated from platelets that had been stimulated with thrombin, and so was design ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aggrecanase
Aggrecanases are extracellular proteolytic enzymes that are members of the ADAMTS (A Disintegrin And Metalloprotease with Thrombospondin Motifs) family. Aggrecanases act on large proteoglycans known as aggrecans, which are components of connective tissues such as cartilage. The inappropriate activity of aggrecanase is a mechanism by which cartilage degradation occurs in diseases such as arthritis. At least two forms of aggrecanase exist in humans: ADAMTS4 or aggrecanase-1 and ADAMTS5 or aggrecanase-2. Both proteins contain thrombospondin Thrombospondins (TSPs) are a family of secreted glycoproteins with antiangiogenic functions. Due to their dynamic role within the extracellular matrix they are considered matricellular proteins. The first member of the family, thrombospondin 1 (T ... (TS) motifs required for proper recognition of substrates. Although both proteins can cleave the substrate aggrecan at the same position, they differ in kinetics and in secondary cleavage sites. Ref ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a basic unit of heredity and the molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protein-coding genes and noncoding genes. During gene expression, the DNA is first copied into RNA. The RNA can be directly functional or be the intermediate template for a protein that performs a function. The transmission of genes to an organism's offspring is the basis of the inheritance of phenotypic traits. These genes make up different DNA sequences called genotypes. Genotypes along with environmental and developmental factors determine what the phenotypes will be. Most biological traits are under the influence of polygenes (many different genes) as well as gen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metalloproteinase
A metalloproteinase, or metalloprotease, is any protease enzyme whose catalytic mechanism involves a metal. An example is ADAM12 which plays a significant role in the fusion of muscle cells during embryo development, in a process known as myogenesis. Most metalloproteases require zinc, but some use cobalt. The metal ion is coordinated to the protein via three ligands. The ligands coordinating the metal ion can vary with histidine, glutamate, aspartate, lysine, and arginine. The fourth coordination position is taken up by a labile water molecule. Treatment with chelating agents such as EDTA leads to complete inactivation. EDTA is a metal chelator that removes zinc, which is essential for activity. They are also inhibited by the chelator orthophenanthroline. Classification There are two subgroups of metalloproteinases: * Exopeptidases, metalloexopeptidases ( EC number: 3.4.17). * Endopeptidases, metalloendopeptidases (3.4.24). Well known metalloendopeptidases include ADAM pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disintegrin
Disintegrins are a family of small proteins (45–84 amino acids in length) from viper venoms that function as potent inhibitors of both platelet aggregation and integrin-dependent cell adhesion. Operation Disintegrins work by countering the blood clotting steps, inhibiting the clumping of platelets. They interact with the beta-1 and -3 families of integrins receptors. Integrins are cell receptors involved in cell–cell and cell–extracellular matrix interactions, serving as the final common pathway leading to aggregation via formation of platelet–platelet bridges, which are essential in thrombosis and haemostasis. Disintegrins contain an RGD (Arg-Gly-Asp) or KGD (Lys-Gly-Asp) sequence motif that binds specifically to integrin IIb-IIIa receptors on the platelet surface, thereby blocking the binding of fibrinogen to the receptor–glycoprotein complex of activated platelets. Disintegrins act as receptor antagonists, inhibiting aggregation induced by ADP, thrombin, platelet-a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aggrecan
Aggrecan (ACAN), also known as cartilage-specific proteoglycan core protein (CSPCP) or chondroitin sulfate proteoglycan 1, is a protein that in humans is encoded by the ''ACAN'' gene. This gene is a member of the lectican (chondroitin sulfate proteoglycan) family. The encoded protein is an integral part of the extracellular matrix in cartilagenous tissue and it withstands compression in cartilage. Aggrecan is a proteoglycan, or a protein modified with large carbohydrates; the human form of the protein is 2316 amino acids long and can be expressed in multiple isoforms due to alternative splicing. Aggrecan was named for its ability to form large aggregates in the cartilage tissue (a large aggregating proteoglycan). Structure Aggrecan is a high molecular weight (1x106 < M < 3x106) proteoglycan. It exhibits a bottlebrush structure, in which |
Proteoglycan
Proteoglycans are proteins that are heavily glycosylated. The basic proteoglycan unit consists of a "core protein" with one or more covalently attached glycosaminoglycan (GAG) chain(s). The point of attachment is a serine (Ser) residue to which the glycosaminoglycan is joined through a tetrasaccharide bridge (e.g. chondroitin sulfate- GlcA- Gal-Gal- Xyl-PROTEIN). The Ser residue is generally in the sequence -Ser-Gly-X-Gly- (where X can be any amino acid residue but proline), although not every protein with this sequence has an attached glycosaminoglycan. The chains are long, linear carbohydrate polymers that are negatively charged under physiological conditions due to the occurrence of sulfate and uronic acid groups. Proteoglycans occur in connective tissue. Types Proteoglycans are categorized by their relative size (large and small) and the nature of their glycosaminoglycan chains. Types include: Certain members are considered members of the "small leucine-rich proteoglyc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Osteoarthritis
Osteoarthritis (OA) is a type of degenerative joint disease that results from breakdown of joint cartilage and underlying bone which affects 1 in 7 adults in the United States. It is believed to be the fourth leading cause of disability in the world. The most common symptoms are joint pain and stiffness. Usually the symptoms progress slowly over years. Initially they may occur only after exercise but can become constant over time. Other symptoms may include joint swelling, decreased range of motion, and, when the back is affected, weakness or numbness of the arms and legs. The most commonly involved joints are the two near the ends of the fingers and the joint at the base of the thumbs; the knee and hip joints; and the joints of the neck and lower back. Joints on one side of the body are often more affected than those on the other. The symptoms can interfere with work and normal daily activities. Unlike some other types of arthritis, only the joints, not internal organs, are af ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MEROPS
MEROPS is an online database for peptidases (also known as proteases, proteinases and proteolytic enzymes) and their inhibitors. The classification scheme for peptidases was published by Rawlings & Barrett in 1993, and that for protein inhibitors by Rawlings ''et al.'' in 2004.Rawlings, N.D., Tolle, D.P. & Barrett, A.J. (2004) "Evolutionary families of peptidase inhibitors." ''Biochem J'' 378, 705-716. The most recent version, MEROPS 12.3, was released in September 2020. Overview The classification is based on similarities at the tertiary and primary structural levels. Comparisons are restricted to that part of the sequence directly involved in the reaction, which in the case of a peptidase must include the active site, and for a protein inhibitor the reactive site. The classification is hierarchical: sequences are assembled into families, and families are assembled into clans. Each peptidase, family, and clan has a unique identifier. Classification Family The families of pe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |